Abstract
Purpose
Traditional treatment regimens for advanced prostate cancer, especially castration-resistant prostate cancer, result in low survival times with severe side effects. Therefore, new treatment options are required. Vitamin C (VC) has been identified as a promising anti-cancer agent of which the effects depend on the accumulation of H2O2 that is produced through autoxidation. Sulfasalazine (SAS), a cystine transporter (Xc−) inhibitor, is known to suppress cellular glutathione (GSH) biosynthesis. Here, we hypothesized that targeting the Xc− transporter via SAS may improve the anti-cancer activity of VC through regulating GSH biosynthesis, which in turn may result in the accumulation of reactive oxygen species (ROS).
Methods
The anti-cancer effect of VC and/or SAS on prostate cancer cells was assessed using WST-8, colony formation and annexin V-FITC/PI FACS assays. Changes in cellular ROS and GSH levels were determined to verify our hypothesis. Finally, BALB/c nude mice bearing prostate cancer xenografts were used to assess the anti-cancer effects of single or combined VC and SAS therapies.
Results
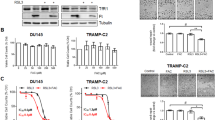
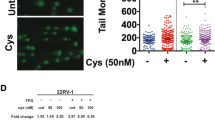
We found that SAS could potentiate the short- and long-term cytotoxicity of VC in prostate cancer cells. We also found that the synergistic effect of SAS and VC led to significant cellular GSH depletion, resulting in increased ROS accumulation. This synergistic effect could be reversed by the antioxidant N-acetyl-L-cysteine (NAC). The synergistic effect of SAS and VC was also noted in prostate cancer xenografts and correlated with immunohistochemistry results.
Conclusions
Our results strongly indicate that SAS, a relatively non-toxic drug that targets cystine transporters, in combination with VC may be superior to their single applications in the treatment of prostate cancer.






Similar content being viewed by others
Abbreviations
- VC:
-
Vitamin C
- ROS:
-
Reactive oxygen species
- SAS:
-
Sulfasalazine
- Xc− :
-
The xc− cystine/glutamate antiporter
- NAC:
-
N-Acetyl-L-cysteine
- SVCTs:
-
Sodium vitamin C co-transporters
- LIP:
-
Labile iron pool
- GLUTs:
-
Facilitative glucose transporters
- DHA:
-
Dehydroascorbate
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- CAM:
-
Complementary and alternative medicine
References
R. Chen, S. Ren, C. Chinese Prostate Cancer, M.K. Yiu, N.C. Fai, W.S. Cheng, L.H. Ian, S. Naito, T. Matsuda, E. Kehinde, A. Kural, J.Y. Chiu, R. Umbas, Q. Wei, X. Shi, L. Zhou, J. Huang, Y. Huang, L. Xie, L. Ma, C. Yin, D. Xu, K. Xu, Z. Ye, C. Liu, D. Ye, X. Gao, Q. Fu, J. Hou, J. Yuan, D. He, T. Pan, Q. Ding, F. Jin, B. Shi, G. Wang, X. Liu, D. Wang, Z. Shen, X. Kong, W. Xu, Y. Deng, H. Xia, A.N. Cohen, X. Gao, C. Xu, Y. Sun, Prostate cancer in Asia: A collaborative report. Asian J Urol 1, 15–29 (2014)
S.C. Ren, R. Chen, Y.H. Sun, Prostate cancer research in China. Asian J Androl 15, 350–353 (2013)
L. Emmett, K. Willowson, J. Violet, J. Shin, A. Blanksby, J. Lee, Lutetium (177) PSMA radionuclide therapy for men with prostate cancer: A review of the current literature and discussion of practical aspects of therapy. J Med Radiat Sci 64, 52–60 (2017)
J.D. Schoenfeld, Z.A. Sibenaller, K.A. Mapuskar, B.A. Wagner, K.L. Cramer-Morales, M. Furqan, S. Sandhu, T.L. Carlisle, M.C. Smith, T.A. Hejleh, D.J. Berg, J. Zhang, J. Keech, K.R. Parekh, S. Bhatia, V. Monga, K.L. Bodeker, L. Ahmann, S. Vollstedt, H. Brown, E.P.S. Kauffman, M.E. Schall, R.J. Hohl, G.H. Clamon, J.D. Greenlee, M.A. Howard, M.K. Schultz, B.J. Smith, D.P. Riley, F.E. Domann, J.J. Cullen, G.R. Buettner, J.M. Buatti, D.R. Spitz, B.G. Allen, O2(−) and H2O2-mediated disruption of Fe metabolism causes the differential susceptibility of NSCLC and GBM cancer cells to pharmacological ascorbate. Cancer Cell 32, 268 (2017)
L.J. Hoffer, L. Robitaille, R. Zakarian, D. Melnychuk, P. Kavan, J. Agulnik, V. Cohen, D. Small, W.H. Miller Jr., High-dose intravenous vitamin C combined with cytotoxic chemotherapy in patients with advanced cancer: A phase I-II clinical trial. PLoS One 10, e0120228 (2015)
Y. Ma, J. Chapman, M. Levine, K. Polireddy, J. Drisko, Q. Chen, High-dose parenteral ascorbate enhanced chemosensitivity of ovarian cancer and reduced toxicity of chemotherapy. Sci Transl Med 6, 222ra218 (2014)
J.L. Welsh, B.A. Wagner, T.J. van't Erve, P.S. Zehr, D.J. Berg, T.R. Halfdanarson, N.S. Yee, K.L. Bodeker, J. Du, L.J. Roberts, J. Drisko, M. Levine, G.R. Buettner, J.J. Cullen, Pharmacological ascorbate with gemcitabine for the control of metastatic and node-positive pancreatic cancer (PACMAN): Results from a phase I clinical trial. Cancer Chemoth Pharm 71, 765–775 (2013)
D.A. Monti, E. Mitchell, A.J. Bazzan, S. Littman, G. Zabrecky, C.J. Yeo, M.V. Pillai, A.B. Newberg, S. Deshmukh, M. Levine, Phase I evaluation of intravenous ascorbic acid in combination with gemcitabine and erlotinib in patients with metastatic pancreatic cancer. PLoS One 7, e29794 (2012)
S. Ohno, Y. Ohno, N. Suzuki, G. Soma, M. Inoue, High-dose vitamin C (ascorbic acid) therapy in the treatment of patients with advanced cancer. Anticancer Res 29, 809–815 (2009)
O.K. Serrano, N.L. Parrow, P.C. Violet, J. Yang, J. Zornjak, A. Basseville, M. Levine, Antitumor effect of pharmacologic ascorbate in the B16 murine melanoma model. Free Radic Biol Med 87, 193–203 (2015)
M. Fukui, N. Yamabe, H.J. Choi, K. Polireddy, Q. Chen, B.T. Zhu, Mechanism of ascorbate-induced cell death in human pancreatic cancer cells: Role of Bcl-2, Beclin 1 and autophagy. Planta Med 81, 838–846 (2015)
J. Du, S.M. Martin, M. Levine, B.A. Wagner, G.R. Buettner, S.H. Wang, A.F. Taghiyev, C. Du, C.M. Knudson, J.J. Cullen, Mechanisms of ascorbate-induced cytotoxicity in pancreatic cancer. Clin Cancer Res 16, 509–520 (2010)
Q. Chen, M.G. Espey, A.Y. Sun, C. Pooput, K.L. Kirk, M.C. Krishna, D.S. Khosh, J. Drisko, M. Levine, Pharmacologic doses of ascorbate act as a prooxidant and decrease growth of aggressive tumor xenografts in mice. Proc Natl Acad Sci U S A 105, 11105–11109 (2008)
H. Tsukaguchi, T. Tokui, B. Mackenzie, U.V. Berger, X.Z. Chen, Y. Wang, R.F. Brubaker, M.A. Hediger, A family of mammalian Na+−dependent L-ascorbic acid transporters. Nature 399, 70–75 (1999)
N.H. Riordan, H.D. Riordan, X. Meng, Y. Li, J.A. Jackson, Intravenous ascorbate as a tumor cytotoxic chemotherapeutic agent. Med Hypotheses 44, 207–213 (1995)
J.C. Vera, C.I. Rivas, J. Fischbarg, D.W. Golde, Mammalian facilitative hexose transporters mediate the transport of dehydroascorbic acid. Nature 364, 79–82 (1993)
C.L. Linster, E. Van Schaftingen, Vitamin C - biosynthesis, recycling and degradation in mammals. FEBS J 274, 1–22 (2007)
S.J. Padayatty, H. Sun, Y.H. Wang, H.D. Riordan, S.M. Hewitt, A. Katz, R.A. Wesley, M. Levine, Vitamin C pharmacokinetics: Implications for oral and intravenous use. Ann Intern Med 140, 533–537 (2004)
E. Cameron, L. Pauling, Supplemental ascorbate in the supportive treatment of cancer: Prolongation of survival times in terminal human cancer. Proc Natl Acad Sci U S A 73, 3685–3689 (1976)
E. Cameron, L. Pauling, The orthomolecular treatment of cancer. I The role of ascorbic acid in host resistance. Chem Biol Interact 9, 273–283 (1974)
E. Cameron, A. Campbell, The orthomolecular treatment of cancer. II Clinical trial of high-dose ascorbic acid supplements in advanced human cancer. Chem Biol Interact 9, 285–315 (1974)
C.M. Stephenson, R.D. Levin, T. Spector, C.G. Lis, Phase I clinical trial to evaluate the safety, tolerability, and pharmacokinetics of high-dose intravenous ascorbic acid in patients with advanced cancer. Cancer Chemother Pharmacol 72, 139–146 (2013)
L.J. Hoffer, M. Levine, S. Assouline, D. Melnychuk, S.J. Padayatty, K. Rosadiuk, C. Rousseau, L. Robitaille, W.H. Miller, Phase I clinical trial of i.v. ascorbic acid in advanced malignancy. Ann Oncol 19, 1969–1974 (2008)
L. Pouillon, P. Bossuyt, J. Vanderstukken, D. Moulin, P. Netter, S. Danese, J.Y. Jouzeau, D. Loeuille, L. Peyrin-Biroulet, Management of patients with inflammatory bowel disease and spondyloarthritis. Expert Rev Clin Phar 10, 1363–1374 (2017)
J. Guan, M. Lo, P. Dockery, S. Mahon, C.M. Karp, A.R. Buckley, S. Lam, P.W. Gout, Y.Z. Wang, The x c − cystine/glutamate antiporter as a potential therapeutic target for small-cell lung cancer: Use of sulfasalazine. Cancer Chemother Pharmacol 64, 463–472 (2009)
J. Yun, E. Mullarky, C.Y. Lu, K.N. Bosch, A. Kavalier, K. Rivera, J. Roper, I.I.C. Chio, E.G. Giannopoulou, C. Rago, A. Muley, J.M. Asara, J. Paik, O. Elemento, Z.M. Chen, D.J. Pappin, L.E. Dow, N. Papadopoulos, S.S. Gross, L.C. Cantley, Vitamin C selectively kills KRAS and BRAF mutant colorectal cancer cells by targeting GAPDH. Science 350, 1391–1396 (2015)
S.J. Dixon, D. Patel, M. Welsch, R. Skouta, E. Lee, M. Hayano, A.G. Thomas, C. Gleason, N. Tatonetti, B.S. Slusher, B.R. Stockwell, Pharmacological inhibition of cystine-glutamate exchange induces endoplasmic reticulum stress and ferroptosis. Elife 3, e02523 (2014)
X.W. Wei, Y. Xu, F.F. Xu, L. Chaiswing, D. Schnell, T. Noel, C. Wang, J.F. Chen, D.K.S. Clair, W.H.S. Clair, RelB expression determines the differential effects of ascorbic acid in normal and cancer cells. Cancer Res 77, 1345–1356 (2017)
W.X. Zhong, H.L. Weiss, R.D. Jayswal, P.J. Hensley, L.M. Downes, D.K.S. Clair, L. Chaiswing, Extracellular redox state shift: A novel approach to target prostate cancer invasion. Free Rad Biol Med 117, 99–109 (2018)
D.W. Doxsee, P.W. Gout, T. Kurita, M. Lo, A.R. Buckley, Y.W. Wang, H. Xue, C.M. Karp, J.C. Cutz, G.R. Cunha, Y.Z. Wang, Sulfasalazine-induced cystine starvation: Potential use for prostate cancer therapy. Prostate 67, 162–171 (2007)
M. Lo, Y.Z. Wang, P.W. Gout, The x(c)(−) cystine/glutamate antiporter: A potential target for therapy of cancer and other diseases. J Cell Physiol 215, 593–602 (2008)
P. Chen, J. Yu, B. Chalmers, J. Drisko, J. Yang, B.Y. Li, Q. Chen, Pharmacological ascorbate induces cytotoxicity in prostate cancer cells through ATP depletion and induction of autophagy. Anti-Cancer Drug 23, 437–444 (2012)
C.M. Doskey, T.J. van't Erve, B.A. Wagner, G.R. Buettner, Moles of a substance per cell is a highly informative dosing metric in cell culture. PLoS One 10, e0132572 (2015)
D.R. Spitz, W.C. Dewey, G.C. Li, Hydrogen peroxide or heat shock induces resistance to hydrogen peroxide in Chinese hamster fibroblasts. J Cell Physiol 131, 364–373 (1987)
P.W. Gout, A.R. Buckley, C.R. Simms, N. Bruchovsky, Sulfasalazine, a potent suppressor of lymphoma growth by inhibition of the x(c)(−) cystine transporter: A new action for an old drug. Leukemia 15, 1633–1640 (2001)
W.J. Chung, H. Sontheimer, Sulfasalazine inhibits the growth of primary brain tumors independent of nuclear factor-kappa. J Neurochem 110, 182–193 (2009)
Q. Chen, M.G. Espey, A.Y. Sun, J.H. Lee, M.C. Krishna, E. Shacter, P.L. Choyke, C. Pooput, K.L. Kirk, G.R. Buettner, M. Levine, Ascorbate in pharmacologic concentrations selectively generates ascorbate radical and hydrogen peroxide in extracellular fluid in vivo. Proc Natl Acad Sci U S A 104, 8749–8754 (2007)
Q. Chen, M.G. Espey, M.C. Krishna, J.B. Mitchell, C.P. Corpe, G.R. Buettner, E. Shacter, M. Levine, Pharmacologic ascorbic acid concentrations selectively kill cancer cells: Action as a pro-drug to deliver hydrogen peroxide to tissues. Proc Natl Acad Sci U S A 102, 13604–13609 (2005)
C.G. Moertel, T.R. Fleming, E.T. Creagan, J. Rubin, M.J. O'Connell, M.M. Ames, High-dose vitamin C versus placebo in the treatment of patients with advanced cancer who have had no prior chemotherapy: A randomized double-blind comparison. New Eng J Med 312, 137–141 (1985)
E.T. Creagan, C.G. Moertel, J.R. O'Fallon, A.J. Schutt, M.J. O'Connell, J. Rubin, S. Frytak, Failure of high-dose vitamin C (ascorbic acid) therapy to benefit patients with advanced cancer: A controlled trial. New Eng J Med 301, 687–690 (1979)
E. Cameron, L. Pauling, Ascorbic acid and the glycosaminoglycans. An orthomolecular approach to cancer and other diseases. Oncology 27, 181–192 (1973)
E. Cameron, D. Rotman, Ascorbic acid, cell proliferation and cancer. Lancet 299, 542–542 (1972)
W. McCormick, Cancer: A collagen disease, secondary to a nutritional deficiency. Arch Pediat 76, 166–171 (1959)
W. McCormick, Cancer: The preconditioning factor in pathogenesis; a new etiologic approach. Arch Pediat 71, 313 (1954)
M.Z. Ma, G. Chen, P. Wang, W.H. Lu, C.F. Zhu, M. Song, J. Yang, S.J. Wen, R.H. Xu, Y.M. Hu, P. Huang, Xc(−) inhibitor sulfasalazine sensitizes colorectal cancer to cisplatin by a GSH-dependent mechanism. Cancer Lett 368, 88–96 (2015)
K. Aquilano, S. Baldelli, M.R. Ciriolo, Glutathione: New roles in redox signaling for an old antioxidant. Front Pharmacol 5, 196 (2014)
W.J. Wu, Y. Zhang, Z.L. Zeng, X.B. Li, K.S. Hu, H.Y. Luo, J. Yang, P. Huang, R.H. Xu, Beta-Phenylethyl isothiocyanate reverses platinum resistance by a GSH-dependent mechanism in cancer cells with epithelial-mesenchymal transition phenotype. Biochem Pharmacol 85, 486–496 (2013)
T. Ishimoto, O. Nagano, T. Yae, M. Tamada, T. Motohara, H. Oshima, M. Oshima, T. Ikeda, R. Asaba, H. Yagi, T. Masuko, T. Shimizu, T. Ishikawa, K. Kai, E. Takahashi, Y. Imamura, Y. Baba, M. Ohmura, M. Suematsu, H. Baba, H. Saya, CD44 variant regulates redox status in cancer cells by stabilizing the xCT subunit of system xc(−) and thereby promotes tumor growth. Cancer Cell 19, 387–400 (2011)
R.S. Chen, Y.M. Song, Z.Y. Zhou, T. Tong, Y. Li, M. Fu, X.L. Guo, L.J. Dong, X. He, H.X. Qiao, Q.M. Zhan, W. Li, Disruption of xCT inhibits cancer cell metastasis via the caveolin-1/beta-catenin pathway. Oncogene 28, 599–609 (2009)
L. Sleire, B.S. Skeie, I.A. Netland, H.E. Forde, E. Dodoo, F. Selheim, L. Leiss, J.I. Heggdal, P.H. Pedersen, J. Wang, P.O. Enger, Drug repurposing: Sulfasalazine sensitizes gliomas to gamma knife radiosurgery by blocking cystine uptake through system xc(−), leading to glutathione depletion. Oncogene 34, 5951–5959 (2015)
W. Zhang, D. Trachootham, J.Y. Liu, G. Chen, H. Pelicano, C. Garcia-Prieto, W.Q. Lu, J.A. Burger, C.M. Croce, W. Plunkett, M.J. Keating, P. Huang, Stromal control of cystine metabolism promotes cancer cell survival in chronic lymphocytic leukaemia. Nat Cell Biol 14, 276 (2012)
M. Lo, V. Ling, C. Low, Y.Z. Wang, P.W. Gout, Potential use of the anti-inflammatory drug, sulfasalazine, for targeted therapy of pancreatic cancer. Curr Oncol 17, 9–16 (2010)
V.S. Narang, G.M. Pauletti, P.W. Gout, D.J. Buckley, A.R. Buckley, Sulfasalazine-induced reduction of glutathione levels in breast cancer cells: Enhancement of growth-inhibitory activity of doxorubicin. Chemotherapy 53, 210–217 (2007)
Acknowledgments
We are grateful to all members of the Xiangsong Zhang group for contributions to this project. We thank QiaoSu, Gangyin Zhao and all members from the animal care facility at The First Affiliated Hospital of Sun Yat-sen University for animal management. We thank Yali Tang for technical supporting with FACS analysis.
This work is supported by the Science and Technology Planning Project of Guangdong Province grant 2017B020210001, the Science and Technology Program of Guangzhou grant 201607010353, the Training program of the Major Research Plan of Sun Yat-Sen University grant 17ykjc10 (to X.S.Z.), the Science and Technology Planning Project of Guangdong Province (Grant No. 2016B030307003), the Science and Technology Planning Project of Guangdong Province (Grant No. 2017B020210001) and the National Natural Science Foundation of China (Grant No.81501509).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All experiments were approved by the Institutional Animal Care and Use Committee of The First Affiliated Hospital of Sun Yat-sen University and conformed to the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zheng, Z., Luo, G., Shi, X. et al. The Xc− inhibitor sulfasalazine improves the anti-cancer effect of pharmacological vitamin C in prostate cancer cells via a glutathione-dependent mechanism. Cell Oncol. 43, 95–106 (2020). https://doi.org/10.1007/s13402-019-00474-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-019-00474-8