1. Introduction
Catechins are part of a large group of plant polyphenols with exceptional antioxidant properties [
1]. Interestingly, these compounds may have protective properties against obesity. A catechin-rich grape seed extract has been reported to significantly reduce body weight in mice with high-fat diet-induced obesity [
2], while green tea catechins have been shown to reduce BMI, body weight and waist circumference in humans [
3]. For (-)-epicatechin specifically (referred to as epicatechin throughout), murine studies have shown that epicatechin administration can reverse the negative effects of maternal obesity [
4]. In humans, it has been demonstrated that epicatechin administration before a meal increased satiety [
5], and further that epicatechin improved post-prandial fat and carbohydrate metabolism [
6]. Altogether, there is evidence to suggest that catechins, and specifically epicatechin, may be protective against obesity.
Globally, more than 39% of adults are overweight and more than 13% are obese [
7,
8,
9]. However, in Australia, more than 67% of adults are overweight [
7]. Interestingly, obesity rates are increasing regardless of geographic location or socioeconomic status [
8]. Importantly, obesity in the elderly is associated with earlier mortality relating to comorbidities such as hypertension, diabetes and heart disease [
10,
11,
12]. Obesity is a complex and multifaceted disease that is not fully understood. However, there have been advancements in the investigations into the genetics of obesity [
13], in particular the potential role of taste genetics on dietary intake and metabolism. This study explores the relationship between taste genetics, body mass index (BMI) and epicatechin intake.
Bitter taste receptors (T2Rs) are a family of receptors responsible for the detection of bitter compounds and potential toxins. Humans have 25 functional T2Rs which, when combined, are capable of detecting hundreds of bitter compounds [
14,
15,
16]. In the oral cavity, genetic variation in these receptors influences oral detection, food preference and intake [
17,
18,
19,
20]. Importantly, these receptors are also expressed throughout the gastrointestinal tract, where they are thought to be involved in the modulation of appetite and satiation [
17,
21,
22], gut motility [
21,
22,
23] and glucose homeostasis [
24]. In addition, functional T2R variants are associated with obesity in a porcine model [
25]. Overall, bitter taste genetics may be associated with obesity via the modulation of dietary intake and/or by the regulation of gastrointestinal hormones and gut function [
26].
TAS2R38 is a widely studied taste gene responsible for the detection of the bitter compounds phenylthiocarbamide (PTC) and 6-n-propyl-2-thiouracil (PROP) [
27]. These compounds are commonly used as tools to detect taste phenotype. Three single-nucleotide polymorphisms (SNPs) give rise to two common forms of the gene. These polymorphisms are part of a haploblock and result in the amino acid substitutions proline-alanine-valine (PAV; associated with tasting PTC and PROP) or alanine-valine-isoleucine (AVI; associated with not tasting PTC or PROP). From this, there are three genotypes associated with taste sensitivity. PAV homozygotes can detect and respond strongly to PTC and PROP and are classified as super-tasters, heterozygotes are classified as tasters and AVI homozygotes cannot detect these compounds and are classified as non-tasters. It is important to note that
TAS2R38 genotype alone does not determine the ability to taste PTC and PROP [
28]. However, it is still used as a general marker of taste acuity [
29].
The
TAS2R38 genotype associated with non-taster status has been linked to significantly higher BMIs and/or increased dietary intake [
30,
31,
32,
33,
34,
35,
36,
37]. However, some studies report no association [
38,
39,
40] and others report inverse associations [
41]. These results may also vary with age and sex [
37,
39,
42]. For example, in a study of 381 females and 348 males, the
TAS2R38-rs1726866 T allele (non-taster) was associated with eating disinhibition in adult women [
17]. Conversely, a study in 81 children found a significant relationship between tasters and high BMI, but reported no differences in energy intake [
41]. Another study in children (
n = 53) which compared taster status to weight-for-height percentiles, found that taster females had a significantly higher weight for height compared to non-taster females and, contrastingly, that non-taster males had a higher weight for height than male tasters [
42]. Furthermore, a study in 118 elderly Polish women found no significant correlation between
TAS2R38 genotype and BMI [
39]. Importantly, the relationship between bitter sensitivity and BMI is known to vary with age [
43]. In a cross-sectional study of 311 men and women, it was found that individuals under 65 with a higher BMI (>28) were less sensitive to bitter taste. However, in the over 65 group, overweight subjects were more sensitive to bitter taste [
43]. Overall, bitter sensitivity, and the relationship between
TAS2R38 genotype and BMI may vary with age and sex.
A group of bitter receptors, T2R4, T2R5 and T2R39, detect epicatechin [
44]. Therefore, we analysed three common
TAS2R polymorphisms that result in functional receptor changes. TAS2R39 was not analysed due to very low polymorphism frequency in this gene [
45]. Two common polymorphisms in the
TAS2R4 gene (rs2233998 and rs2234001) and one polymorphism in the
TAS2R5 gene (rs2227264) were assessed. These three SNPs are part of a haploblock on chromosome 7 and have previously been linked to perceived bitterness of coffee [
46]. This study explores the multidirectional interactions between
TAS2R genotype, epicatechin intake, and BMI together in an elderly cohort.
3. Results
A total of 563 participants were included in this study following exclusions (
Table 2). Of these, 254 were male and 309 were female. The average overall age was 77.4. In men, the average age was 77.4; and in women, the average age was 77.3 The average BMI for men was 28.5 and 28.6 for women, with an average of 28.5 overall. The average overall energy intake was 8223.5 kJ (8453.1–7993.9). There was a significant difference in average male daily energy intake (8656.2 kJ (8311.3–9001.1)) compared to females (7866.7 kJ (7563.1–8170.3)). The average number of serves of high-epicatechin foods per day was 5.2 for men, women and overall.
The genotype distributions are shown in
Table 3. The
TAS2R4 rs2233998 polymorphism has a minor allele frequency (MAF) of 0.42. 21% of participants were homozygous for the minor allele (CC), 25% were homozygous for the major allele (TT) and 54% of participants were heterozygotes. In the rs2234001 polymorphism, 20% of participants were homozygous for the minor allele (CC; MAF = 0.48), 25% were homozygous for the major allele (GG) and 55% were heterozygotes. In the
TAS2R5 rs2227264 polymorphism, 21% of participants were homozygous for the minor allele (TT; MAF = 0.44), 27% of participants were homozygous for the major allele and 62% were heterozygotes.
Homozygosity of the minor allele was associated with significantly lower BMI in both
TAS2R4 polymorphisms (
Figure 1). The
TAS2R4-rs2233998 CC genotype was associated with an average BMI of 27.7 (95% CI [26.8, 28.6]) and the presence of the G allele was associated with significantly larger average BMI of 28.7 (95% CI [28.2, 29.2];
p = 0.02). The
TAS2R4-rs2234001 CC genotype was similarly associated with a lower BMI (27.6 (95% CI [26.7, 28.5])) compared to the presence of the G allele (28.8 (95% CI [28.3, 29.2]);
p = 0.01). The presence of the major allele in the
TAS2R5 polymorphism (rs2227264) was not associated with a significant difference in BMI in this cohort.
The relationship between BMI and TASR genotype was sex specific (
Figure 2). For all three polymorphisms (rs2233998, rs2234001 and rs2227264), the presence of the major allele was associated with a significantly higher BMI than in males homozygous for the minor allele. The
TAS2R4-rs2233998 CC genotype was associated with a BMI of 26.8 (25.6–28.0) and the presence of the G allele was associated with a significantly higher BMI (28.9 (28.3–29.6);
p = 0.002). The
TAS2R4-s2234001 CC genotype was associated with a BMI of 26.9 (25.7–28.1), whereas the presence of the G allele was associated with a significantly higher BMI of 29.0 (28.4–29.7;
p = 0.003). Finally, the
TAS2R5-rs2227264 TT genotype was associated with a BMI of 26.8 (25.7–28.0) compared to 29.0 (28.4–29.6;
p = 0.001). In females, there was no significant difference in BMI of individuals homozygous for the minor allele, compared to the presence of the major allele. Additionally, these results remained significant when adjusted for daily energy intake.
Due to the significant difference in energy intake between males and females (
Table 2), the data were analysed for a relationship between
TAS2R genotype and daily energy intake, this analysis was stratified by sex (
Table 4). The presence of the major allele was not associated with a significant different energy intake compared to homozygosity for the minor allele in any of the three polymorphisms. When stratified by sex, there was also no significant differences between the genotypes analysed.
There was a significant correlation between increased daily energy intake and increased epicatechin intake (
p < 0.0001; β = 0.5) (
Figure 3A). There was no significant relationship between BMI and serves of high-epicatechin foods per day (
p = 0.2; β = −0.06) (
Figure 3B). Additionally, there was no significant association between dietary energy intake per day and BMI (
p = 0.5; β = −0.03) in this cohort.
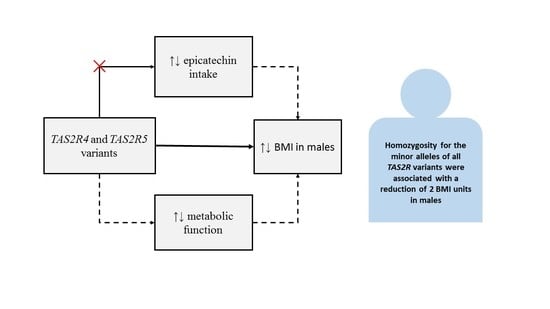
There was no significant association between
TAS2R genotype and the average number of serves of high-epicatechin foods (
Table 5). However, there was consistently higher epicatechin intake observed in males homozygous for the minor allele of all three polymorphisms (compared to male carriers of the major allele and both female groups). Interestingly, these are the same groups associated with significantly lower BMIs in
Figure 2.
4. Discussion
The secondary analysis presented here identifies potential associations between common
TAS2R4 polymorphisms and BMI. Homozygosity for the minor alleles of
TAS2R4-rs2233998 and
TAS2R4-rs2234001 was associated with significantly lower BMI compared to carriers of the major allele in this cohort. The three
TAS2RSNPs analysed in this study (
TAS2R4-rs2233998,
TAS2R4-rs2234001 and
TAS2R5-rs2227264) are part of a haploblock on chromosome 7 [
46]. Therefore, it was expected that the SNPs assessed may be associated with the same parameter (BMI). Importantly, the association between
TAS2R4 genotypes (
TAS2R4-rs2233998,
TAS2R4-rs2234001) and BMI could not be explained in this cohort by daily energy intake or by daily epicatechin intake. The lack of association between energy intake and BMI suggests
TAS2R4 genotypes do not modulate food intake. Alternatively, functional
TAS2R4 polymorphisms may affect the extra-oral roles of taste receptors in energy metabolism [
26].
Importantly, this study highlights a previously unexplored potential relationship between
TAS2R4 and
TAS2R5 genotypes and BMI. In males, homozygosity for the minor allele of all three polymorphisms corresponded to a lower BMI (>2 BMI units) in each instance, this equates to several kilograms of weight difference, depending on height. The risk for conditions associated with higher BMI such as hypertension, diabetes and cardiovascular disease increases with increased BMI [
8,
9,
60]. For example, each one-unit increase in BMI is significantly associated with a 4% risk of ischemic stroke and a 6% increase in risk of hemorrhagic stroke [
61]. Additionally, in adolescent men (
n = 37674), risk for diabetes increases by 9.8% and risk for heart disease increases by 12% per one BMI unit increase [
62]. The effects of increased BMI is particularly pronounced in the elderly where overweight and obese individuals experience earlier mortality than their normal weight counterparts [
10,
11,
12]. Overall, this study provides insight into the genetic risk factors for obesity in the elderly.
A potential role for other extra-oral bitter receptors genotypes in predicting BMI has previously been suggested [
35,
41]. A Korean study (
n = 3567) identified that the
TAS2R38-rs10246939 TT genotype (associated phenotypically with non-tasting) was associated with a significantly higher BMI in females. However, there was no association between genotype and energy intake, suggesting another biological mechanism [
35]. Additionally, a study in children (
n = 81) which found a significant association between tasters and high BMI, found no complementary relationship between taster status and energy intake [
41]. When taken together with the results presented here, a potential role for extra-oral T2Rs in predicting BMI, without modulating energy intake is suggested.
The extra-oral roles of T2R activation on appetite and gut motility may be a potential explanatory factor for these observations. Treatment with bitter taste receptor agonists has been shown to alter satiation, food intake and gastric emptying. Intra-gastric administration of 1 μmol/kg of the bitter taste receptor agonist, denatonium benzoate, significantly increased satiation in healthy volunteers (
n = 13) [
21]. Furthermore, a study in 16 women that examined the effects of chewing and then expectorating either a bitter bar or a pleasant-tasting bar determined that gastric emptying was significantly delayed in response to the bitter-tasting bar [
23]. In animal and cell models, it has been identified that intestinal taste receptors modulate the secretion of gastrointestinal hormones GLP-1, GIP, ghrelin, CCK and PYY [
21,
22,
23,
24,
26,
63,
64] involved in appetite, digestion and glucose homeostasis [
21,
22,
24,
63,
64,
65]. Therefore, functional extra-oral receptor changes related to
TAS2R genotype may influence the secretion of gastrointestinal hormones in response to bitter agonists and impact obesity risk. However, additional studies are needed to determine the causative mechanism(s).
Although there was no association between epicatechin intake and BMI in this cohort, the administration of epicatechin (detected by T2R4 and T2R5) has previously been associated with improved cardiometabolic function [
6,
66]. A study in 20 adults found that following 1 mg/kg epicatechin ingestion, lipid oxidation was significantly increased in overweight subjects and post-prandial triglyceridemia decreased in normal and overweight subjects [
6]. Another small study (12 males) reported significantly improved vascular function following 1–2 mg/kg body weight oral dose of epicatechin [
66]. However, results from the present study suggest that nutritive doses of epicatechin did not have an effect on BMI in elderly subjects. Similarly, a previous study identified that a nutritive dose of 25 mg/day had no effect on cardiometabolic factors (blood pressure, glucose, insulin, insulin resistance, triglycerides, or total LDL, or HDL cholesterol) [
67].
Interestingly, the number of serves of high-epicatechin foods per day was associated with increased daily energy intake in this study. It may be that higher epicatechin intake in this study is simply a function of higher overall food intake. By contrast, it has previously been demonstrated in humans that epicatechin administration before a meal increased satiety [
5]. Additionally, while
TAS2R38 genotypes have previously been associated with altered oral detection, food preference and intake [
17,
18,
19,
20], there was no significant association between
TAS2R4 and
TAS2R5 genotypes and epicatechin intake in this study. This suggests that these polymorphisms are not altering oral detection and modulating intake of epicatechin containing foods. However, functional receptor changes associated with these
TAS2Rpolymorphisms may alter extra-oral metabolic responses to epicatechin.
Associations between
TAS2R38 genotypes and BMI and associated taster status and BMI have previously been reported [
30,
31,
32,
33,
34,
35,
36,
37,
41]. However, this study is unique in examining the relationship between
TAS2R4 and
TAS2R5 genotypes and BMI and supports a role for
TAS2R genotypes in predicting BMI in males. The association between homozygosity for the minor alleles of
TAS2R4-rs2233998,
TAS2R4-rs2234001 and
TAS2R5-rs2227264 and lower BMI appears to be specific to males in this cohort. Sex specificity has been previously identified between
TAS2R genotypes and a variety of outcomes, including dietary intake [
17,
68], BMI, [
35,
42] and thyroid function, which effects metabolism [
69]. The sex specificity of the observed results may be explained by potential interactions between sex hormones and taste signalling, other genes located on sex chromosomes, or social determinants of food choice that are gender specific. Further studies are needed to understand these relationships. Altogether, this study provides further evidence of a potential sex dimorphism in the relationship between
TAS2R4 and
TAS2R5 genotypes and BMI in elderly subjects.
It is important to note that the identified relationship between
TAS2R38 and BMI may also vary with age. This study found no significant association between
TAS2R4 or
TAS2R5 genotypes and BMI in elderly women, while other studies in children [
32,
41] and adults [
30,
31,
33,
34,
35] report potential links between
TAS2R38 genotype and BMI, and a previous study in elderly women found no significant association between
TAS2R38 genotype and BMI [
39]. It is well-documented that taste loss occurs during ageing [
70,
71]. Therefore, further studies are needed in children and adults to determine whether the relationship between
TAS2R4 and
TAS2R5 genotypes and BMI is age specific as well as sex specific.
The use of an elderly cohort means that these results may be specific to elderly and not necessarily applicable to younger populations. However, this cohort was useful in studying the long-term effects of
TAS2R genotypes on BMI. Another limitation of this study included estimations of energy intake and epicatechin intake. Dietary intake estimations are limited by low-accuracy and subject bias of food frequency questionnaires [
72]. Additionally, it is important to note that epicatechin intake is hard to quantify due to the high variability of food composition [
59] and the large variation in reported concentrations of epicatechin within foods [
58].
Overall, we propose that TAS2R genotypes, resulting in functional receptor changes, may alter metabolic hormone secretion in a sex-specific manner, with downstream effects on BMI. Additional studies in larger and more diverse age groups are needed to establish this potential association between TAS2R genotype(s) and BMI. Importantly, if these relationships are established, they may be used to predict obesity risk, and potentially combat conditions associated with a larger BMI in the form of personalised nutrition therapies. Ultimately, this study provides initial insight into the complex relationship between taste genetics and BMI and the potential roles for extra-oral T2Rs in obesity risk.