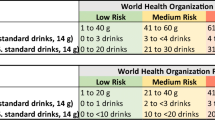
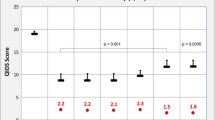
Abstract
Problematic substance use may attenuate the effect of treating depression in people living with HIV (PLWH). We examined the potential moderating effect of problematic substance use on depression and adherence outcomes in PLWH (N = 143) who participated in a randomized controlled trial of cognitive behavioral therapy for adherence and depression (CBT-AD) and were randomized to either CBT-AD or enhanced treatment as usual (ETAU). Problematic substance use was operationalized as either having substance use-related diagnosis or current substance use in the past 30 days with a lifetime history of problematic use. Acute (baseline to 4-month) and follow up (4, 8, and 12-month) general linear modeling with time, condition, problematic baseline substance use, and corresponding interactions demonstrated that substance use did not significantly moderate the effects of CBT-AD on adherence or depression improvements. Therefore, CBT-AD was beneficial for PLWH with depression, regardless of problematic substance use when starting depression treatment. Based on these results, clinicians should not withhold CBT treatment for depression in patients with HIV and problematic substance use to attain reductions in depression and gains in adherence.





Similar content being viewed by others
Data Availability
Contact corresponding author.
Code Availability
Contact corresponding author.
References
Gaynes BN, O’Donnell J, Nelson E, Heine A, Zinski A, Edwards M, et al. Psychiatric comorbidity in depressed HIV-infected individuals: common and clinically consequential. General Hospital Psychiatry. 2015 Jul 1;37(4):277–82.
Pence BW, Miller WC, Whetten K, Eron JJ, Gaynes BN. Prevalence of DSM-IV-defined mood, anxiety, and substance use disorders in an HIV clinic in the Southeastern United States. JAIDS J Acquir Immune Defic Syndr. 2006;42(3):298–306.
Bing EG, Burnam MA, Longshore D, Fleishman JA, Sherbourne CD, London AS, et al. Psychiatric Disorders and Drug Use Among Human Immunodeficiency Virus–Infected Adults in the United States. Archives of General Psychiatry. 2001 Aug 1;58(8):721–8.
McGowan CC, Weinstein DD, Samenow CP, Stinnette SE, Barkanic G, Rebeiro PF, et al. Drug use and receipt of highly active antiretroviral therapy among HIV-infected persons in two US clinic cohorts. PLoS ONE. 2011;6(4):e18462.
Rabkin JG, McElhiney MC, Ferrando SJ. Mood and substance use disorders in older adults with HIV/AIDS: methodological issues and preliminary evidence. Aids. 2004;18:43–8.
Skeer MR, Mimiaga MJ, Mayer KH, O’Cleirigh C, Covahey C, Safren SA. Patterns of substance use among a large urban cohort of HIV-infected men who have sex with men in primary care. AIDS Behav. 2012;16(3):676–89.
Gonzalez A, Barinas J, O’Cleirigh C. Substance Use: Impact on Adherence and HIV Medical Treatment. Curr HIV/AIDS Rep. 2011 Dec;8(4):223–34.
Uthman OA, Magidson JF, Safren SA, Nachega JB. Depression and adherence to antiretroviral therapy in low-, middle-and high-income countries: a systematic review and meta-analysis. Curr HIV/AIDS Rep. 2014;11(3):291–307.
Celentano DD, Vlahov D, Cohn S, Shadle VM, Obasanjo O, Moore RD. Self-reported Antiretroviral Therapy in Injection Drug Users. JAMA. 1998 Aug;12(6):544–6. 280(.
Lucas GM, Cheever LW, Chaisson RE, Moore RD. Detrimental effects of continued illicit drug use on the treatment of HIV-1 infection. J Acquir Immune Defic Syndr. 2001 Jul 1;27(3):251–9.
Amico KR, Harman JJ, Johnson BT. Efficacy of antiretroviral therapy adherence interventions: a research synthesis of trials, 1996 to 2004. J Acquir Immune Defic Syndr. 2006 Mar;41(3):285–97.
de Bruin M, Viechtbauer W, Schaalma HP, Kok G, Abraham C, Hospers HJ. Standard Care Impact on Effects of Highly Active Antiretroviral Therapy Adherence Interventions: A Meta-analysis of Randomized Controlled Trials. Archives of Internal Medicine. 2010 Feb 8;170(3):240–50.
Simoni JM, Kurth AE, Pearson CR, Pantalone DW, Merrill JO, Frick PA. Self-report measures of antiretroviral therapy adherence: a review with recommendations for HIV research and clinical management. AIDS Behav. 2006;10(3):227–45.
Simoni JM, Amico KR, Pearson CR, Malow R. Strategies for promoting adherence to antiretroviral therapy: a review of the literature. Curr Infect Dis Rep. 2008;10(6):515–21.
Simoni JM, Amico KR, Smith L, Nelson K. Antiretroviral adherence interventions: translating research findings to the real world clinic. Curr HIV/AIDS Rep. 2010;7(1):44–51.
Binford MC, Kahana SY, Altice FL. A Systematic Review of Antiretroviral Adherence Interventions for HIV-Infected People Who Use Drugs. Curr HIV/AIDS Rep. 2012 Dec 1;9(4):287–312.
Newcomb ME, Bedoya CA, Blashill AJ, Lerner JA, O’Cleirigh C, Pinkston MM, et al. Description and demonstration of cognitive behavioral therapy to enhance antiretroviral therapy adherence and treat depression in HIV-infected adults. Cogn Behav Pract. 2015;22(4):430–8.
Safren SA, O’Cleirigh C, Tan J, Raminani S, Reilly LC, Otto MW, et al. A Randomized Controlled Trial of Cognitive Behavioral Therapy for Adherence and Depression (CBT-AD) in HIV-infected Individuals. Health Psychol. 2009;28(1):1–10.
Safren SA, O’Cleirigh CM, Bullis JR, Otto MW, Stein MD, Pollack MH. Cognitive Behavioral Therapy for Adherence and Depression (CBT-AD) in HIV-infected Injection Drug Users: A Randomized Controlled Trial. J Consult Clin Psychol. 2012;80(3):404–15.
Safren SA, Bedoya CA, O’Cleirigh C, Biello KB, Pinkston MM, Stein MD, et al. Cognitive behavioural therapy for adherence and depression in patients with HIV: a three-arm randomised controlled trial. The Lancet HIV. 2016;3(11):e529–38.
Simoni JM, Wiebe JS, Sauceda JA, Huh D, Sanchez G, Longoria V, et al. A preliminary RCT of CBT-AD for adherence and depression among HIV-positive Latinos on the US-Mexico border: the Nuevo Dia study. AIDS Behav. 2013;17(8):2816–29.
Andersen LS, Magidson JF, O’Cleirigh C, Remmert JE, Kagee A, Leaver M, et al. A pilot study of a nurse-delivered cognitive behavioral therapy intervention (Ziphamandla) for adherence and depression in HIV in South Africa. J Health Psychol. 2018 May 1;23(6):776–87.
Morgenstern J, Morgan TJ, McCrady BS, Keller DS, Carroll KM. Manual-guided cognitive-behavioral therapy training: A promising method for disseminating empirically supported substance abuse treatments to the practice community. Psychol Addict Behav. 2001;15(2):83–8.
van Boekel LC, Brouwers EPM, van Weeghel J, Garretsen HFL. Stigma among health professionals towards patients with substance use disorders and its consequences for healthcare delivery: Systematic review. Drug and Alcohol Dependence. 2013 Jul 1;131(1):23–35.
Labbe AK, O’Cleirigh CM, Stein M, Safren SA. Depression CBT treatment gains among HIV-infected persons with a history of injection drug use varies as a function of baseline substance use. Psychol Health Med. 2015 Oct;3(7):870–7. 20(.
Dale SK, Traeger L, O’Cleirigh C, Bedoya CA, Pinkston M, Wilner JG, et al. Baseline Substance Use Interferes with Maintenance of HIV Medication Adherence Skills. AIDS Patient Care and STDs. 2016 May 1;30(5):215–20.
Safren SA, Otto MW, Worth JL, Salomon E, Johnson W, Mayer K, et al. Two strategies to increase adherence to HIV antiretroviral medication: life-steps and medication monitoring. Behav Res Ther. 2001;39(10):1151–62.
Safren SA, Otto MW, Worth JL. Life-steps: Applying cognitive behavioral therapy to HIV medication adherence. Cogn Behav Pract. 1999;6(4):332–41.
Safren SA, Gonzalez JS, Soroudi N. Coping with Chronic Illness: A Cognitive-Behavioral Therapy Approach for Adherence and Depression, Therapist Guide [Internet]. Coping with Chronic Illness. Oxford University Press; 2007 [cited 2020 Oct 27]. Available from: https://www.oxfordclinicalpsych.com/view/https://doi.org/10.1093/med:psych/9780195315165.001.0001/med-9780195315165.
Safren SA, Gonzalez JS, Soroudi N. Coping with chronic illness: Cognitive behavioral therapy for adherence and depression, client workbook. NY: Oxford University Press; 2007.
Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E, et al. The Mini-International Neuropsychiatric Interview (MINI): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;59(20):22–33.
Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J psychiatry. 1979;134(4):382–9.
National Institute of Mental Health. CGI: Clinical Global Impression Scale - NIMH. Psychopharmacol Bull. 1985;21:839–44.
Radloff LS. The CES-D scale: A self-report depression scale for research in the general population. Appl Psychol Meas. 1977;1(3):385–401.
Cacciola JS, Alterman AI, McLellan AT, Lin YT, Lynch KG. Initial evidence for the reliability and validity of a “Lite” version of the Addiction Severity Index. Drug and Alcohol Dependence. 2007 Mar 16;87(2):297–302.
McLellan AT, Kushner H, Metzger D, Peters R, Smith I, Grissom G, et al. The fifth edition of the addiction severity index. Journal of Substance Abuse Treatment. 1992 Jun 1;9(3):199–213.
McLellan AT, Lubrosky L, O?brien CP. An improved evaluation instrument for substance abuse patients: the Addiction Severity Index. In: Problems of Drug Dependence, 1979: Proceedings of the 41st Annual Scientific Meeting, the Committee on Problems of Drug Dependence, Inc; Editor, Louis S Harris. Department of Health, Education, and Welfare, Public Health Service, Alcohol &; 1980. p. 142.
Acknowledgements
Research reported in this publication was supported by a National Institute of Mental Health grant R01XXXXX. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Mental Health or the National Institutes of Health. Some of the author time, and resources for statistical consultation, were also supported by grants National Institute of Allergy and Infectious Diseases (NIAID) XXXXX and P30XXXXX. This study was also funded by a National Institute of Mental Health grant P30XXXXX and National Institute of Drug Abuse grant K24XXXX.
Funding
This study was funded by a National Institute of Mental Health grant R01MH084757 to Dr. Safren. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Mental Health or the National Institutes of Health. Some of the author time, and resources for statistical consultation, were also supported by grants National Institute of Allergy and Infectious Diseases (NIAID) P30AI042853 and the National Institute for Minority Health and Health Disparities (NIMHD) K23MD015690. This study was also funded by a National Institute of Mental Health grant P30MH11687 and National Institute of Drug Abuse grant K24DA040489.
Author information
Authors and Affiliations
Contributions
SAS conceptualized the parent study and provided the data. SAS and JNS conceptualized the current study, and JNS completed this project as part of her master’s thesis. TRG directed and oversaw the statistical analyses. DM assisted with writing the introduction section. SKD and AH provided suggestions and edits throughout many phases of this project.
Corresponding author
Ethics declarations
Conflicts of Interest/competing Interests
Dr. Safren receives royalties from Oxford University Press, Guilford Publications, and Springer/Humana Press on books related to the delivery of cognitive behavioral treatments, such as the one described in this article.
Ethics Approval
All study procedures were approved and reviewed by the respective institutional review boards (IRBs) at the Miriam Hospital, Fenway Health, and Massachusetts General Hospital.
Consent to Participate
Participants were recruited from three HIV clinics and community self-referral (e.g., advertisements on internet sites and newspapers). In addition, two of the clinics (Fenway Health and Massachusetts General Hospital) utilized the patient health questionnaire-2 (PHQ-2) to screen their patients for depression during clinic visits. Patients who scored two or greater, which indicated possible depression, were invited to participate. Interested patients completed a pre-consent screening over the phone (or in-person), and then potential participants were scheduled for a full baseline visit to determine their study eligibility. To participate, eligible participants also completed an informed consent process at the baseline visit; those unwilling or unable to provide informed consent were excluded.
Consent for Publication
The senior/corresponding author has provided consent to publish this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Appendix
Appendix
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sanders, J.N., Glynn, T.R., Mayo, D. et al. Does Problematic Substance Use Moderate Efficacy of Cognitive Behavioral Therapy for Adherence and Depression in HIV?. AIDS Behav 27, 1123–1132 (2023). https://doi.org/10.1007/s10461-022-03849-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10461-022-03849-5