Abstract
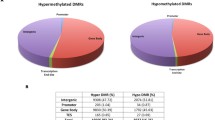
Epigenetic reprogramming of host genes via DNA methylation is increasingly recognized as critical for the outcome of diverse infectious diseases, but information for malaria is not yet available. Here, we investigate the effect of blood-stage malaria of Plasmodium chabaudi on the DNA methylation status of host gene promoters on a genome-wide scale using methylated DNA immunoprecipitation and Nimblegen microarrays containing 2,000 bp oligonucleotide features that were split into −1,500 to −500 bp Ups promoters and −500 to +500 bp Cor promoters, relative to the transcription site, for evaluation of differential DNA methylation. Gene expression was analyzed by Agilent and Affymetrix microarray technology. Challenging of female C57BL/6 mice with 106 P. chabaudi-infected erythrocytes resulted in a self-healing outcome of infections with peak parasitemia on day 8 p.i. These infections induced organ-specific modifications of DNA methylation of gene promoters. Among the 17,354 features on Nimblegen arrays, only seven gene promoters were identified to be hypermethylated in the spleen, whereas the liver exhibited 109 hyper- and 67 hypomethylated promoters at peak parasitemia in comparison with non-infected mice. Among the identified genes with differentially methylated Cor-promoters, only the 7 genes Pigr, Ncf1, Klkb1, Emr1, Ndufb11, and Tlr6 in the liver and Apol6 in the spleen were detected to have significantly changed their expression. Remarkably, the Cor promoter of the toll-like receptor Tlr6 became hypomethylated and Tlr6 expression increased by 3.4-fold during infection. Concomitantly, the Ups promoter of the Tlr1 was hypermethylated, but Tlr1 expression also increased by 11.3-fold. TLR6 and TLR1 are known as auxillary receptors to form heterodimers with TLR2 in plasma membranes of macrophages, which recognize different pathogen-associated molecular patterns (PAMPs), as, e.g., intact 3-acyl and sn-2-lyso-acyl glycosylphosphatidylinositols of P. falciparum, respectively. Our data suggest therefore that malaria-induced epigenetic fine-tuning of Tlr6 and Tlr1 through DNA methylation of their gene promoters in the liver is critically important for initial recognition of PAMPs and, thus, for the final self-healing outcome of blood-stage infections with P. chabaudi malaria.





Similar content being viewed by others
References
Akira S, Takeda K (2004) Toll-like receptors signalling. Nature Rev Imunol 4:499–511
Al-Quraishy S, Delic D, Sies H, Wunderlich F, Abdel-Baki AA, Dkhil MA (2011) Differential miRNA expression in the mouse jejunum during garlic treatment of Eimeria papillata infections. Parasitol Res 109:387–394
Al-Quraishy S, Dkhil M, Delic D, Abdel-Baki AA, Wunderlich F (2012) Organ-specific testosterone-insensitive response of miRNA expression of C57BL/6 mice to Plasmodium chabaudi malaria. Parasitol Res 111:1093–1101
Benten WPM, Ulrich P, Kühn-Velten WN, Vohr HW, Wunderlich F (1997) Testosterone-induced susceptibility to Plasmodium chabaudi malaria: persistence after withdrawal of testosterone. Parasite Immunol 13:357–367
Bierne H, Hamon M, Cossart P (2013) Epigenetics and bacterial infections. Cold Spring Harbor Perspect Med 2(12):a010272
Bilzer M, Roggel F, Gerbes AL (2006) Role of Kupffer cells in host defense and liver disease. Liver Int 26:1175–1186
Bird A (2002) DNA methylation patterns and epigenetic memory. Genes Dev 16:6–21
Bosman GJ, Willekens FL, Werre JM (2005) Erythrocyte aging: a more than superficial resemblance to apoptosis? Cell Physiol Biochem 16:1–8
Cedar H, Bergman Y (2012) Programming of DNA methylation patterns. Annu Rev Biochem 81:97–117
Chotivanich K, Udomsangpetch R, McGready R, Proux S, Newton P, Pukrittayakamee S, Looareesuwan S, White NJ (2002) Central role of the spleen in malaria parasite clearence. J Inf Dis 185:1538–1541
Cohen S, Lambert PH (1982) In: Cohen S, Warren KS (eds) Imunology of parasitic infections, 2nd edn. Blackwell, Oxford, pp 422–474
Crispe IN (2009) The liver as a lymphoid organ. Annu Rev Immunol 27:147–163
Delic D, Gailus N, Vohr HW, Dkhil M, Al-Quraishy S, Wunderlich F (2010a) Testosterone-induced permanent changes of hepatic gene expression in female mice sustained during Plasmodium chabaudi malaria infection. J Mol Endocrinol 45:379–390
Delic D, Warskulat U, Borsch E, Al-Qahtani A-GS, Häussinger D, Wunderlich F (2010b) Loss of ability to self-heal malaria upon taurine transporter deletion. Infect Immun 78:1642–1649
Delic D, Dkhil M, Al-Quraishy S, Wunderlich F (2011a) Hepatic miRNA expression reprogrammed by Plasmodium chabaudi malaria. Parsitol Res 108:1111–1121
Delic D, Ellinger-Ziegelbauer H, Vohr HW, Dkhil M, Al-Quraishy S, Wunderlich F (2011b) Testosterone response of hepatic gene expression in female mice having acquired testosterone-unresponsive immunity to Plasmodium chabaudi malaria. Steroids 76:1204–1212
Dkhil MA, Al-Quraishy S, Delic D, Abdel-Baki AA, Wunderlich F (2013) Testosterone-induced persistent susceptibility to Plasmodium chabaudi malaria: long-term changes of lincRNA and mRNA expression in the spleen. Steroids 78:220–227
Doolan DL, Dobafio C, Baird JK (2009) Acquired immunity to malaria. Clin Microbiol Rev 22:13–36
Garcia LS (2010) Malaria. Clin Lab Med 30(1):93–129
Gardiner-Garden M, Frommer M (1987) CpG islands in vertebrate genomes. J Mol Biol 20:261–282
Häussinger D, Kubitz R, Reinehr R, Bode JG, Schliess F (2004) Molecular aspects of medicine: from experimental to clinical hepatology. Mol Aspects Med 25:221–360
Jarra W, Brown KN (1989) Protective immunity to malaria: studies with cloned lines of rodent malaria in CBA/Ca mice. IV. The specificity of mechanisms resulting in crisis and resolution of the primary acute phase parasitaemia of Plasmodium chabaudi chabaudi and P. yoelii yoelii. Parasite Immunol 11:1–13
Jensen M, Mehlhorn H (2009) Seventy-five years of resorchin in the fight against malaria. Parasitol Res 105:609–627
Jones PA (2012) Functions of DNA methylation: islands, start sites, gene bodies and beyond. Nat Rev Genet 29:484–492
Junpee A, Tencomnao T, Sanprasert V, Nuchprayoon S (2010) Association between toll-like receptor 2 (TLR2) polymorphisms and asymptomatic bancroftian filariasis. Parasitol Res 107:807–816
Krishnegowa G, Hajjar AM, Zhu J, Douglass EJ, Uematsu S, Akira S, Woods AS, Gowda DC (2005) Induction of proinflammatory responses in macrophages by the glycosylphsophatidylinositols of Plasmodium falciparum. J Biol Chem 9:8606–8616
Kruecken J, Dkhil MA, Braun JV, Schroetel RM, El-Khadragy M, Carmeliet P, Mossmann H, Wunderlich F (2005a) Testosterone suppresses protective responses of the liver to blood-stage malaria. Infect Immun 73:436–443
Kruecken J, Mehnert LI, Dkhil M, El-Khadragy M, Benten WPM, Mossman H, Wunderlich F (2005b) Massive destruction of malaria-parasitized red blood cells despite spleen closure. Infect Immun 73:6390–6398
Kruecken J, Delic D, Pauen H, Wojtalla A, El-Khadragy M, Dkhil MA, Mossmann H, Wunderlich F (2009) Augmented partcle trapping and attenuated inflammation in the liver by protective vaccination against Plasmodium chabaudi malaria. Malar J 8:54–64
Langhorne J, Ndungu FM, Sponaas AM, Marsk K (2008) Immunity to malaria: more questions than answers. Nat Immunol 7:725–732
Lempiäinen H, Müller A, Brasa S, Teo SS, Roloff TC, Morawiec L, Zamurovic N, Vicart A, Funhoff E, Couttet P, Schübeler D, Grenet O, Marlowe J, Moggs J, Terranova R (2011) Phenobarbital mediates an epigenetic switch at the constitutive androstan receptor (CAR) target gene Cyp2b10 in the liver of B6C3F1 mice. Plos One 6(3):e18216
Liang P, Song F, Ghosh S, Morien E, Qin M, Mahmood S, Fujiwara K, Igarashi J, Nagase H, Held WA (2011) Genome-wide survey reveals dynamic widespread tissue-specific changes in DNA methylation during development. BMC Genomics 12:231–247
Liu Q, Tuo W, Gao H, Zhu XQ (2010) MicroRNAs of parasites: current status and future perspectives. Parasitol Res 107:501–507
MacKinnon MJ, Read AF (2003) Virulence in malaria: an evolutionary viewpoint. Phil Trans R Soc Lond B Biol Sci 359:965–986
Mannoor MK, Halder RC, Morshed SR, Ariyasinghe A, Bakir HY, Kawamura H, Watanabe H, Sekikawa H, Abo T (2002) Essential role of extrathymic T cells in protection against malaria. J Immunol 169:301–306
McCall CE, Yoza B, Liu T, El-gazzar M (2010) Gene-specific epigenetic regulation in serious infections with systemic inflammation. J Innate Immun 2:395–405
Mehlhorn H (ed) (2008) Enzyclopedic reference of parasitology, vol 1, 3rd edn. Berlin, Springer
Mertens I, ClinckspoorI JT, Nachman R, Schoofs L (2006) FMRFamide related peptide ligands activate the Caenorhabditis elegans orphan GPCR Y59H11AL.1. Peptides 27:1291–1296
Mohn F, Weber M, Schübeler D, Roloff TC (2009) Methylated DNA immunoprecipitation (MeDIP). Meth Mol Biol 507:55–64
Moore LD, Le T, Fan G (2012) DNA methylation and its basic function. Neuropsychopharmacol 38:23–38
Muehlfeld S, Schmitt-Wrede HP, Harder A, Wunderlich F (2009) FMRFamide-like neuropeptides as putative ligands of the latrophilin-like HC110-R from Haemonchus contortus. Mol Biochem Parasitol 164(2):162–164
Murthi P, Kalionis B, Ghabrial H, Dunlop NE, Smallwood RA, Sewell RB (2006) Kupffer cell function during the erythrocytic stages of malaria. J Gastroenterol Hepatol 21:313–318
Ndlovu MN, Denis H, Fuks F (2011) Exposing the DNA methylome iceberg. Trends Biochem Sci 36:381–387
Oliveira-Nascimento L, Massari P, Wetzler LM (2012) The role of TLR2 in infection and immunity. Front Immunol 3:1–17
Paschos K, Allday MJ (2010) Epigenetic reprogrammimg of host genes in viral and microbial pathogenesis. Trends Microbiol 18:439–447
Raghavendra K, Barik TK, Reddy BP, Sharma P, Dash AP (2011) Malaria vector control: from past to future. Parasitol Res 108:757–779
Santourlidis S, Trompeter HI, Weinhold S, Eisermann B, Meyer KL, Wernet P, Uhrberg M (2002) Crucial role of DNA methylation in determination of clonally distributed killer cell Ig-like receptor expression patterns in NK cells. J Immunol 15:4253–4261
Santourlidis S, Wernet P, Ghanjati F, Graffmann N, Springer J, Kriegs C, Zhao X, Brands J, Arauzo-Bravo MJ, Neves R, Koegler G, Uhrberg M (2011) Unrestricted somatic stemm cells (USSC) from human umbilical cord blood display uncommitted epigenetic signatures of the major stem cell pluripotency genes. Stem Cell Res 6:60–69
Shames DS, Minna JD, Gazdar AF (2007) DNA methylation in health, disease, and cancer. Curr Mol Med 7:85–102
Smith ZD, Meissner A (2013) DNA methylation: roles in mammalian development. Nat Rev Genet 14:204–220
Stein RA (2012) Epigenetics and environmental exposures. J Epidemiol Community Health 66:8–134
Stephens R, Culleton RL, Lamb TJ (2011) The contribution of Plasmodium chabaudi to our understanding of malaria. Trends Parasitol 28:73–82
Suarez-Alvarez B, Rodriguez RM, Fraga MF, Lopez-larrea C (2012) DNA methylation: a promising landscape for immune system-related diseases. Trens Genet 28:506–514
WHO (2010) World malaria report. World Health Organization, Geneva
Wunderlich CM, Delic D, Behnke K, Ströhle P, Chaurasia B, Al-Quraishy S, Wunderlich F, Brüning JC, Wunderlich FT (2012) Cutting edge: inhibition of IL-6 trans-signalling protects from malaria-induced lethality in mice. J Immunol 188:4141–4144
Wunderlich F, Dkhil M, Mehnert LI, Braun JV, El-Khadragy M, Borsch E, Hermsen D, Benten WPM, Pfeffer K, Mossmann H, Kruecken J (2005) Testosterone responsiveness of spleen and liver in female lymphotoxin b receptor-deficient mice resitant to blood-stage malaria. Microbes Infect 7:399–409
Wunderlich F, Mossmann H, Helwig M, Schillinger G (1988) Resistance to Plasmodium chabaudi in B10 mice. Infect Immun 56:2400–2406
Wunderlich F, Stübig H, Königk E (1982) Dvelopment of Plasmodium chabaudi in mice red blood cells: Structural properties of the host and parasite membranes. J Protozool 29:60–66
Yazdani SS, Mukherjee P, Chauhan VS, Chitnis CE (2006) Immune responses to asexual blood-stages of malaria parasites. Curr Mol Med 6:187–203
Zhang X, Ho SM (2011) Epigenetics meets endocrinology. J Mol Endocrinol 46:R11–R32
Zhang Z, Tang H, Wang Z, Zhang B, Liu W, Lu H, Xiao L, Liu X, Wnag R, Li X, Wu M, Li G (2011) MiR-185 targets the DNA methyltransferases 1 and regulates global DNA methylation in human glioma. Mol Cancer 10:124–140
Acknowledgments
We thank Dr. MJ Arauzo-Bravo for expert advice in bioinformatic evaluation of differential DNA methylation. This research was supported by King Saud University, National Program for Science and Technology, through grant no. 10-BIO1212-02.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al-Quraishy, S., Dkhil, M.A., Abdel-Baki, A.A.S. et al. Genome-wide screening identifies Plasmodium chabaudi-induced modifications of DNA methylation status of Tlr1 and Tlr6 gene promoters in liver, but not spleen, of female C57BL/6 mice. Parasitol Res 112, 3757–3770 (2013). https://doi.org/10.1007/s00436-013-3565-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-013-3565-2