Abstract
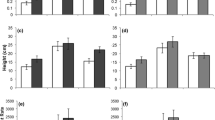
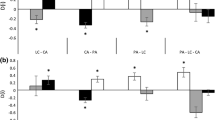
Many invasive species can respond opportunistically to favorable growing conditions. In a previous work, we found that invasive species in the family Commelinaceae were more opportunistic than their noninvasive congeners and could therefore outperform noninvasive relatives in an environment with abundant resources and no competition. Contrary to the expectation that superior performance under favorable conditions comes at the cost of reduced performance under stressful conditions, invasive species did not perform more poorly relative to noninvasive congeners under any conditions we examined. Here we expand our search for potential costs of opportunism in invasive species to additional environmental conditions in which invasive taxa have been shown or predicted to perform poorly. We grew four invasive and four noninvasive species in environments consisting of all possible combinations of high and low soil resources and presence and absence of clipping (removal of aboveground biomass). We also fed leaves of each species to a generalist herbivore to assess resistance to herbivory. We found that the advantage of invasive species is reduced but not eliminated by low soil resources and clipping. At low soil resources, invasive species produced softer leaves than noninvasive species and might therefore be less resistant to generalist herbivory than noninvasive species, although a direct comparison of resistance in a no-choice bioassay revealed no difference. The invasive species outperformed noninvasive species only under the most favorable conditions, and the noninvasive species did not outperform the invasive species in any environment.
Similar content being viewed by others
References
Agrawal AA, Kotanen PM (2003) Herbivores and the success of exotic plants: a phylogenetically controlled experiment. Ecol Lett 6:712–715
Arendt JD (1997) Adaptive intrinsic growth rate: an integration across taxa. Q Rev Biol 72:149–177
Barrett PM (2000) Evolutionary consequences of dating the Yixian Formation. Trends Ecol Evol 15:99–103
Baruch Z, Bilbao B (1999) Effects of fire and defoliation on the life history of native and invader C4 grasses in a Neotropical savanna. Oecologia 119:510–520
Baruch Z, Goldstein G (1999) Leaf construction cost, nutrient concentration, and net CO2 assimilation of native and invasive species in Hawaii. Oecologia 121:183–192
Bellingham PJ, Duncan RP, Lee WG, Buxton RP (2004) Seedling growth rate and survival do not predict invasiveness in naturalized woody plants in New Zealand. Oikos 106:308–316
Bergelson J, Purrington CB (1996) Surveying patterns in the cost of resistance in plants. Am Nat 148:536–558
Burke MJW, Grime JP (1996) An experimental study of plant community invasibility. Ecology 77:776–790
Burns JH (2004) A comparison of invasive and noninvasive dayflowers (Commelinaceae) across experimental nutrient and water gradients. Divers Distrib 10:387–397
Burns JH, Winn AA (2006) Are invasive species more plastic? A comparison of plastic responses to competition by invasive and noninvasive congeners in the Commelinaceae. Biol Invasions 00:1–11
Caldwell MM, Richards JH, Johnson DA, Nowak RS, Dzurec RS (1981) Coping with herbivory: photosynthetic capacity and resource allocation in two semiarid Agropyron bunchgrasses. Oecologia 50:14–24
Capinera JL (1999) Featured creatures: fall armyworm (Spodoptera frugiperda). University of Florida <http://www.creatures.ifas.ufl.edu/field/fall_armyworm.htm>. Accessed June 29, 2005
Carpenter D, Cappuccino N (2005) Herbivory, time since introduction and the invasiveness of exotic plants. J␣Ecol 93:315–321
Chapin FSI (1980) The mineral nutrition of wild plants. Ann Rev Ecol Syst 11:233–260
Chapin FSI, Autman K, Pugnaire F (1993) Evolution of suites of traits in response to environmental stress. Am Nat 142:S78–S92
Colautti RI, Ricciardi A, Grigorovich IA, MacIsaac HJ (2004) Is invasion success explained by the enemy release hypothesis? Ecol Lett 7:721–733
Coley PD (1983) Herbivory and defensive characteristics of tree species in a lowland tropical forest. Ecol Monogr 53:209–233
Connor EF (1988) Plant water deficits and insect responses: The preference of Corythucha arcuata (Heteroptera: Tingidae) for the foliage of white oak, Quarcus alba. Ecol Entomol 13:375–381
Daehler CC (2003) Performance comparisons of co-occuring native and alien invasive plants: implications for conservation and restoration. Ann Rev Ecol Syst 34: 183–211
Daehler CC, Carino DA (2000) Predicting invasive plants:prospects for a general screening system based on current regional models. Biol Invasions 2:92–102
del-Val E, Crawley MJ (2005) Are grazing increaser species better tolerators than decreasers? An experimental assessment of defoliation tolerance in eight British grassland species. J Ecol 93:1005–1016
Durand LZ, Goldstein G (2001) Photosynthesis, photoinhibition, and nitrogen use efficiency in native and invasive tree ferns in Hawaii. Oecologia 126:345–354
Faden RB (1982) Commelinaceae. In: Flora of North America Editorial (ed) Flora of North America, Oxford University Press, New York, pp 170–197
Feeny P (1970) Seasonal changes in oak leaf tannins and nutrients as a cause of spring feeding by winter moth caterpillars. Ecology 51: 565–581
Feller IC (1995) Effects of nutrient enrichment on growth and herbivory of dwarf red mangrove (Rhizophora mangle). Ecol Monogr 65:477–505
Felsenstein J (1985) Phylogenies and the comparative method. The Am Nat 125:1–15
Fernández RJ, Reynolds JF (2000) Potential growth and drought tolerance of eight desert grasses: lack of a trade-off? Oecologia 123:90–98
Grime JP, Hunt R (1975) Relative growth-rate: its range and adaptive significace in a local flora. J Ecol 63: 393–422
Grotkopp E, Rejmánek M, Rost TL (2002) Toward a causal explanation of plant invasiveness: seedling growth and life-history strategies of 29 pine (Pinus) species. Am Nat 159: 396–419
Harvey PH, Pagel MD (1991) The comparative method in evolutionary biology. Oxford University Press Inc., New York, NY
Hobbs RJ, Huenneke LF (1992) Disturbance, diversity, and invasion: implications for conservation. Conserv Biol 6:324–337
Holm LG, Plucknett DL, Pancho JV, Herberger JP (1977) Commelina. In: Holm LG, Plucknett DL, Pancho JV, Herberger JP (eds) The world’s worst weeds: distribution and biology. Krieger Publishing Company, Honolulu, Hawai’i, pp 225–235
Keane RM, Crawley MJ (2002) Exotic plant invasions and the enemy release hypothesis. Trends Ecol Evol 17:164–170
Korner C (1991) Some often overlooked plant characteristics as determinants of plant growth: a reconsideration. Funct Ecol 5:162–173
Lambers H, Poorter H (1992) Inherent variation in growth rate between higher plants: a search for physiological causes and ecological consequences. Adv Ecol Res 23:187–261
Maron JL, Vilá M (2001) When do herbivores affect plant invasion? Evidence for the natural enemies and biotic resistance hypotheses. Oikos 95:361–373
Pammenter NW, Drennan PM, Smith VR (1986) Physiological and anatomical aspects of photosynthesis of two Agrostis species at a sub-Antarctic island. New Phytol 102:143–160
Perrings C, Williamson M, Dalmazzone S (2000) Introduction. In: Perrings C, Williamson M, Dalmazzone S (eds) The economics of biological invasions. Edward Elgar, Cheltenham, UK, pp 1–13
Rejmánek M, Richardson D (1996) What attributes make some plant species more invasive? Ecology 77:1655–1661
Richardson DM, Pyšek P, Rejmánek M, Barbour MG, Panetta FD, West CJ (2000) Naturalization and invasion of alien plants: concept and definitions. Divers Distrib 6:93–107
Rogers WE, Siemann E (2002) Effects of simulated herbivory and resource availability on native and invasive exotic tree seedlings. Basic Appl Ecol 3:297–307
SAS Institute I (1999) SAS/STAT Users Guide
Schierenbeck KA, Mack RN, Sharitz RR (1994) Effects of herbivory on growth and biomass allocation in native and introduced species of Lonicera. Ecology 75:1661–1672
Siemann E, Rogers WE (2003) Reduced resistance of invasive varieties of the alien tree Sapium sebiferum to a generalist herbivore. Oecologia 135:451–457
Sokal RR, Rohlf FJ (1981) Biometry: the principles and practice of statistics in biological research. W. H. Freeman, San Francisco, CA
Sparks AN (1979) A review of the biology of the fall armyworm. Fla Entomol 62:82–87
Standish RJ, Robertson AW, Williams PA (2001) The impact of an invasive weed Tradescantia flumenensis on native forest regeneration. J Appl Ecol 38:1253–1263
Strauss SY, Rudgers JA, Lau JA, Irwin RE (2002) Direct and ecological costs of resistance to herbivory. Trends Ecol Evol 17:278–285
USDA [United States Department of Agriculture] (2002) Animal Plant Health Inspection Service. Accessed online <http://www.invasivespecies.gov>
Vitousek PM, D’Antonio CM, Loope LL, Westbrooks R (1996) Biological invasions as global environmental change. Am Sci 84:468–478
Wilson AK (1981) Commelinaceae: a review of the distribution, biology and control of the important weeds belonging to this family. Trop Pest Manage 27:405–418
Acknowledgements
We thank Roy Weidner for constructing our leaf penetrometer; Sarah Gray, Robin Hopkins, Ken Moriuchi, and three NSF Young Scholars Program students for assistance in harvesting 600 plants from wet sand under outrageously hot and humid conditions; Rob Meager for providing Spodoptera frugiperda eggs; and Nora Underwood for larval rearing advice and supplies. This manuscript was improved by comments from Katherine M. Howe, Thomas E. Miller, Anne B. Thistle, Nora Underwood, and two anonymous reviewers. Funds were provided by the Young Scholars Program, administered by the Office of Science Teaching Activities at Florida State University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Burns, J.H., Halpern, S.L. & Winn, A.A. A test for a cost of opportunism in invasive species in the Commelinaceae. Biol Invasions 9, 213–225 (2007). https://doi.org/10.1007/s10530-006-9027-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-006-9027-3