Abstract
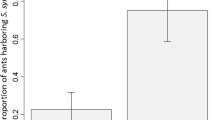
Some aphid species form close associations with ants: offering them honeydew and obtaining protection from ants in return. However, mutualistic interactions with ants can also have a negative influence on aphid physiological and morphological traits. Wolbachia are intracellular bacteria whose major genotypes are classified into 17 supergroups (A to S except G and R). Aphid species within the genus Tuberculatus feed on Fagaceae leaves and exhibit two contrasting ecological characteristics, ant-attendance and non-attendance. Previous work has found that ant-attended species exhibit lower dispersal and are likely to form aggregated colonies. Considering that host-parasitoid interactions may well be one of the most common horizontal transmission routes of Wolbachia, it is therefore expected that ant associations will be associated with higher Wolbachia infection rate in Tuberculatus aphid species. This study compared Wolbachia infection rates between 11 ant-attended and 12 non-attended Tuberculatus aphid species, which were collected throughout Japan and around Mt. Kariwangsan in South Korea. Mean infection rates of Wolbachia were 30.2% in ant-attended species and 3.1% in non-attended species. The Wolbachia haplotypes detected were classified into supergroups B, M, N, and O. A phylogenetic tree of Tuberculatus aphids constructed from a mitochondrial gene of cytochrome oxidase subunit I and nuclear gene of 18S rRNA was used to examine the correlation between Wolbachia infection rates and ant associations. The phylogenetic comparative analysis showed that Wolbachia infection rates were significantly higher in ant-attended species. Possible Wolbachia infection routes are discussed in terms of the differences in the ecological characteristics between ant-attended and non-attended aphid species. This study shows that the spread of microorganisms is affected by host species interactions, and contributes new insights into the evolution of mutualistic interactions.



Similar content being viewed by others
Data availability
Data are available by request to the author.
References
Ahmed MZ, Li SJ, Xue X, Yin XJ, Ren SX, Jiggins FM, Greeff JM, Qiu BL (2015) The intracellular bacterium Wolbachia uses parasitoid wasps as phoretic vectors for efficient horizontal transmission. PLoS Pathog 11:e1004672. https://doi.org/10.1371/journal.ppat.1004672
Augustinos AA, Santos-Garcia D, Dionyssopoulou E, Moreira M, Papapanagiotou A, Scarvelakis M, Doudoumis V, Ramos S, Aguiar AF, Borges PAV, Khadem M, Latorre A, Tsiamis G, Bourtzis K (2011) Detection and characterization of Wolbachia infections in natural populations of Aphids: is the hidden diversity fully unraveled? PLoS ONE 6:e28695. https://doi.org/10.1371/journal.pone.0028695
Baldo L, Hotopp JCD, Jolley KA, Bordenstein SR, Biber SA, Choudhury RR, Hayashi C, Maiden MCJ, Tettelin H, Werren JH (2006) Multilocus sequence typing system for the endosymbiont Wolbachia pipientis. Appl Environ Microbiol 72:7098–7110. https://doi.org/10.1128/AEM.00731-06
Barker SC, Whiting MF, Johnson KP, Murrell A (2003) Phylogeny of lice (Insecta: Phthiraptera) inferred from small subunit rRNA. Zool Scr 32:407–414. https://doi.org/10.1046/j.1463-6409.2003.00120.x
Bing XL, Xia WQ, Gui JD, Yan GH, Wang XW, Liu SS (2014) Diversity and evolution of the Wolbachia endosymbionts of Bemisia (Hemiptera: Aleyrodidae) whiteflies. Ecol Evol 4:2714–2737. https://doi.org/10.1002/ece3.1126
Brodeur J, Rosenheim JA (2000) Intraguild interactions in aphid parasitoids. Entomol Exp Appl 97:93–108. https://doi.org/10.1046/j.1570-7458.2000.00720.x
Casiraghi M, Anderson TJ, Bandi C, Bazzocchi C, Genchi C (2001) A phylogenetic analysis of filarial nematodes: comparison with the phylogeny of Wolbachia endosymbionts. Parasitology 122:93–103. https://doi.org/10.1017/s0031182000007149
De Clerck C, Tsuchida T, Massart S, Lepoivre P, Francis F, Jijakli MH (2014) Combination of genomic and proteomic approaches to characterize the symbiotic population of the banana aphid (Hemiptera: Aphididae). Environ Entomol 43:29–36. https://doi.org/10.1603/EN13107
De Clerck C, Fujiwara A, Joncour P, Léonard S, Félix ML, Francis F, Jijakli MH, Tsuchida T, Massart S (2015) A metagenomic approach from aphid’s hemolymph sheds light on the potential roles of co-existing endosymbionts. Microbiome 3:63. https://doi.org/10.1186/s40168-015-0130-5
Felsenstein J (1985) Phylogenies and the comparative method. Am Nat 125:1–15. https://doi.org/10.1086/284325
Fischer M, Shingleton A (2001) Host plant and ants influence the honeydew sugar composition of aphids. Funct Ecol 15:544–550. https://doi.org/10.1046/j.0269-8463.2001.00550.x
Fischer CY, Lognay GC, Detrain C, Heil M, Grigorescu A, Sabri A, Thonart P, Haubruge E, Verheggen FJ (2015) Bacteria may enhance species association in an ant–aphid mutualistic relationship. Chemoecology 25:223–232. https://doi.org/10.1007/s00049-015-0188-3
Flatt T, Weisser W (2000) The effects of mutualistic ants on aphid life history traits. Ecology 81:3522–3529. https://doi.org/10.1890/0012-9658(2000)081[3522:TEOMAO]2.0.CO;2
Folmer O, Black M, Hoeh W, Lutz R, Vrijenhoek R (1994) DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol Marine Biol Biotechnol 3:294–299
Frost CL, FernÁndez-MarÍn H, Smith JE, Hughes WOH (2010) Multiple gains and losses of Wolbachia symbionts across a tribe of fungus-growing ants. Mol Ecol 19:4077–4085. https://doi.org/10.1111/j.1365-294X.2010.04764.x
Garland T Jr, Harvey PH, Ives AR (1992) Procedures for the analysis of comparative data using phylogenetically independent contrasts. Syst Biol 41:18–32. https://doi.org/10.1093/sysbio/41.1.18
Gavotte L, Mercer DR, Stoeckle JJ, Dobson SL (2010) Costs and benefits of Wolbachia infection in immature Aedes albopictus depend upon sex and competition level. J Invertebr Pathol 105:341–346. https://doi.org/10.1016/j.jip.2010.08.005
Glowska E, Dragun-Damian A, Dabert M, Gerth M (2015) New Wolbachia supergroups detected in quill mites (Acari: Syringophilidae). Infect Genet Evol 30:140–146. https://doi.org/10.1016/j.meegid.2014.12.019
Gómez-Valero L, Soriano-Navarro M, Pérez-Brocal V, Heddi A, Moya A, García-Verdugo JM, Latorre A (2004) Coexistence of Wolbachia with Buchnera aphidicola and a secondary symbiont in the aphid Cinara cedri. J Bacteriol 186:6626–6633. https://doi.org/10.1128/JB.186.19.6626-6633.2004
Harvey PH, Pagel MD (1991) The comparative method in evolutionary biology. Oxford University Press, New York
Henry LM, Maiden MCJ, Ferrari J, Godfray HCJ (2015) Insect life history and the evolution of bacterial mutualism. Ecol Lett 18:516–525. https://doi.org/10.1111/ele.12425
Hertaeg C, Risse M, Vorburger C, De Moraes CM, Mescher MC (2021) Aphids harbouring different endosymbionts exhibit differences in cuticular hydrocarbon profiles that can be recognized by ant mutualists. Sci Rep 11:1–10. https://doi.org/10.1038/s41598-021-98098-2
Hilgenboecker K, Hammerstein P, Schlattmann P, Telschow A, Werren JH (2008) How many species are infected with Wolbachia? A statistical analysis of current data. FEMS Microbiol Lett 281:215–220. https://doi.org/10.1111/j.1574-6968.2008.01110.x
Hosokawa T, Koga R, Kikuchi Y, Meng XY, Fukatsu T (2010) Wolbachia as a bacteriocyte-associated nutritional mutualist. Proc Natl Acad Sci USA 107:769–774. https://doi.org/10.1073/pnas.0911476107
Kaneko S (2002) Aphid-attending ants increase the number of emerging adults of the aphid’s primary parasitoid and hyperparasitoids by repelling intraguild predators. Entomol Sci 5:131–146
Kaneko S (2003) Different impacts of two species of aphid-attending ants with different aggressiveness on the number of emerging adults of the aphid’s primary parasitoid and hyperparasitoids. Ecol Res 18:199–212. https://doi.org/10.1046/j.1440-1703.2003.00547.x
Katayama N, Suzuki N (2002) Cost and benefit of ant attendance for Aphis craccivora (Hemiptera: Aphididae) with reference to aphid colony size. Can Entomol 134:241–249. https://doi.org/10.4039/Ent134241-2
Kaur R, Shropshire JD, Cross KL, Leigh B, Mansueto AJ, Stewart V, Bordenstein SR, Bordenstein SR (2021) Living in the endosymbiotic world of Wolbachia: a centennial review. Cell Host Microbe 29:879–893. https://doi.org/10.1016/j.chom.2021.03.006
Keller L, Liautard C, Reuter M, Brown WD, Sundström L, Chapuisat M (2001) Sex ratio and Wolbachia infection in the ant Formica exsecta. Heredity 87:227–233. https://doi.org/10.1046/j.1365-2540.2001.00918.x
Lefoulon E, Clark T, Borveto F, Perriat-Sanguinet M, Moulia C, Slatko BE, Gavotte L (2020) Pseudoscorpion Wolbachia symbionts: diversity and evidence for a new supergroup S. BMC Microbiol 20:188. https://doi.org/10.1186/s12866-020-01863-y
Leroy PD, Wathelet B, Sabri A, Francis F, Verheggen FJ, Capella Q, Thonart P, Haubruge E (2011) Aphid-host plant interactions: does aphid honeydew exactly reflect the host plant amino acid composition? Arthropod-Plant Interact 5:193–199. https://doi.org/10.1007/s11829-011-9128-5
Li SJ, Ahmed MZ, Lv N, Shi PQ, Wang XM, Huang JL, Qiu BL (2017) Plant-mediated horizontal transmission of Wolbachia between whiteflies. ISME J 11:1019–1028. https://doi.org/10.1038/ismej.2016.164
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Manzano-Marín A (2020) No evidence for Wolbachia as a nutritional co-obligate endosymbiont in the aphid Pentalonia nigronervosa. Microbiome 8:72. https://doi.org/10.1186/s40168-020-00865-2
Moreira M, Aguiar AMF, Bourtzis K, Latorre A, Khadem M (2019) Wolbachia (Alphaproteobacteria: Rickettsiales) infections in isolated aphid populations from oceanic islands of the azores archipelago: revisiting the supergroups M and N. Environ Entomol 48:326–334. https://doi.org/10.1093/ee/nvy189
Mouratidis A, Vacas S, Herrero J, Navarro-Llopis V, Dicke M, Tena A (2021) Parasitic wasps avoid ant-protected hemipteran hosts via the detection of ant cuticular hydrocarbons. Proc R Soc Lond Ser B Biol Sci 288:20201684. https://doi.org/10.1098/rspb.2020.1684
Okayama K, Katsuki M, Sumida Y, Okada K (2016) Costs and benefits of symbiosis between a bean beetle and Wolbachia. Anim Behav 119:19–26. https://doi.org/10.1016/j.anbehav.2016.07.004
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O'Hara RB et al. (2012) vegan: Community Ecology Package 2012.
Oliver KM, Russell JA, Moran NA, Hunter MS (2003) Facultative bacterial symbionts in aphids confer resistance to parasitic wasps. Proc Natl Acad Sci USA 100:1803–1807
Oliver TH, Mashanova A, Leather SR, Cook JM, Jansen VAA (2007) Ant semiochemicals limit apterous aphid dispersal. Proc R Soc Lond Ser B Biol Sci 274:3127–3131. https://doi.org/10.1098/rspb.2007.1251
Paradis E, Schliep K (2019) ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35:526–528. https://doi.org/10.1093/bioinformatics/bty633
Posada D, Crandall KA (1998) Modeltest: testing the model of DNA substitution. Bioinformatics 14:817–818. https://doi.org/10.1093/bioinformatics/14.9.817
Purvis A, Rambaut A (1995) Comparative analysis by independent contrasts (CAIC): an Apple Macintosh application for analysing comparative data. Bioinformatics 11:247–251. https://doi.org/10.1093/bioinformatics/11.3.247
Quednau FW (1999) Atlas of the drepanosiphine aphids of the world. Part I: Panaphidini Oestlund, 1922-Myzocallidina Börner, 1942 (1930) (Hemiptera: Aphididae: Calaphidinae). Contributions of the American Entomological Institute, 31(1).
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Reeves DD, Price SL, Ramalho MO, Moreau CS (2020) The diversity and distribution of Wolbachia, Rhizobiales, and Ophiocordyceps within the widespread Neotropical turtle ant, Cephalotes atratus (Hymenoptera: Formicidae). Neotrop Entomol 49:52–60. https://doi.org/10.1007/s13744-019-00735-z
Ren W, Wei H, Yang Y, Shao S, Wu H, Chen X, Yang Z (2020) Molecular detection and phylogenetic analyses of Wolbachia in natural populations of nine galling Aphid species. Sci Rep 10:12025. https://doi.org/10.1038/s41598-020-68925-z
Renyard A, Gries R, Lee J, Chalissery JM, Damin S, Britton R, Gries G (2021) All sugars ain’t sweet: selection of particular mono-, di- and trisaccharides by western carpenter ants and European fire ants. R Soc Open Sci 8:210804. https://doi.org/10.1098/rsos.210804
Ros VID, Fleming VM, Feil EJ, Breeuwer AJ (2009) How diverse is the genus Wolbachia? Multiple-gene sequencing reveals a putatively new Wolbachia supergroup recovered from spider mites (Acari: Tetranychidae). Appl Environ Microbiol 75:1036–1043. https://doi.org/10.1128/AEM.01109-08
Sadeghi-Namaghi H, Amiri-Jami A (2018) Success of aphid parasitoids and their hosts varies with ant attendance: a field study. Entomol Sci 21:406–411. https://doi.org/10.1111/ens.12319
Sanaei E, Charlat S, Engelstädter J (2021) Wolbachia host shifts: routes, mechanisms, constraints and evolutionary consequences. Biol Rev 96:433–453. https://doi.org/10.1111/brv.12663
Sanaei E, Lin YP, Cook LG, Engelstädter J (2022) Wolbachia in scale insects: a distinct pattern of infection frequencies and potential transfer routes via ant associates. Environ Microbiol 24:1326–1339. https://doi.org/10.1111/1462-2920.15833
Scarborough C, Ferrari J, Godfray H (2005) Aphid protected from pathogen by endosymbiont. Science 310:1781. https://doi.org/10.1126/science.1120180
Shoemaker DD, Ahrens M, Sheill L, Mescher M, Keller L, Ross KG (2003) Distribution and prevalence of Wolbachia infections in native populations of the fire ant Solenopsis invicta (Hymenoptera: Formicidae). Environ Entomol 32:1329–1336. https://doi.org/10.1603/0046-225X-32.6.1329
Simões PM, Mialdea G, Reiss D, Sagot MF, Charlat S (2011) Wolbachia detection: an assessment of standard PCR protocols. Mol Ecol Res 11:565–572. https://doi.org/10.1111/j.1755-0998.2010.02955.x
Stadler B, Dixon AFG (1998) Costs of ant attendance for aphids. J Anim Ecol 67:454–459. https://doi.org/10.1046/j.1365-2656.1998.00209.x
Stadler B, Dixon AFG (2005) Ecology and evolution of aphid-ant interactions. Annu Rev Ecol Evol Syst 36:345–372. https://doi.org/10.1146/annurev.ecolsys.36.091704.175531
Stadler B, Kindlmann P, Šmilauer P, Fiedler K (2003) A comparative analysis of morphological and ecological characters of European aphids and lycaenids in relation to ant attendance. Oecologia 135:422–430. https://doi.org/10.1007/s00442-003-1193-8
Swofford DL (2002) PAUP*. Phylogenetic analysis using parsimony (and other methods). Version 4. Sinauer Associates, Sunderland
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680. https://doi.org/10.1093/nar/22.22.4673
Tsutsui ND, Kauppinen SN, Oyafuso AF, Grosberg RK (2003) The distribution and evolutionary history of Wolbachia infection in native and introduced populations of the invasive argentine ant (Linepithema humile). Mol Ecol 12:3057–3068. https://doi.org/10.1046/j.1365-294X.2003.01979.x
Viljakainen L, Reuter M, Pamilo P (2008) Wolbachia transmission dynamics in Formica wood ants. BMC Evol Biol 8:55. https://doi.org/10.1186/1471-2148-8-55
Vorburger C, Gehrer P, Rodriguez P (2010) A strain of the bacterial symbiont Regiella insecticola protects aphids against parasitoids. Biol Lett 6:109–111. https://doi.org/10.1098/rsbl.2009.0642
Völkl W (1992) Aphids or their parasitoids: who actually benefits from ant-attendance? J Anim Ecol 61:273–281. https://doi.org/10.2307/5320
Wang Z, Shen ZR, Song Y, Liu HY, Li ZX (2009) Distribution and diversity of Wolbachia in different populations of the wheat aphid Sitobion miscanthi (Hemiptera: Aphididae) in China. Eur J Entomol 106:49–55. https://doi.org/10.14411/eje.2009.007
Way MJ (1963) Mutualism between ants and honeydew-producing Homoptera. Annu Rev Entomol 8:307–344. https://doi.org/10.1146/annurev.en.08.010163.001515
Weinert L, Araujo-Jnr EV, Ahmed MZ, Welch JJ (2015) The incidence of bacterial endosymbionts in terrestrial arthropods. Proc R Soc Lond Ser B Biol Sci 282:20150249. https://doi.org/10.1098/rspb.2015.0249
Werren JH, Windsor DM (2000) Wolbachia infection frequencies in insects: evidence of a global equilibrium? Proc R Soc Lond Ser B Biol Sci 267:1277–1285. https://doi.org/10.1098/rspb.2000.1139
Werren JH, Baldo L, Clark ME (2008) Wolbachia: master manipulators of invertebrate biology. Nat Rev Microbiol 6:741–751. https://doi.org/10.1038/nrmicro1969
White JA, Kelly SE, Cockburn SN, Perlman SJ, Hunter MS (2011) Endosymbiont costs and benefits in a parasitoid infected with both Wolbachia and Cardinium. Heredity 106:585–591. https://doi.org/10.1038/hdy.2010.89
Yao I (2010) Contrasting patterns of genetic structure and dispersal ability in ant-attended and non-attended Tuberculatus aphids. Biol Lett 6:282–286. https://doi.org/10.1098/rsbl.2009.0781
Yao I (2011) Phylogenetic comparative methods reveal higher wing loading in ant-attended Tuberculatus aphids (Hemiptera: Aphididae). Can Entomol 143:35–43. https://doi.org/10.4039/n10-050
Yao I (2014) Costs and constraints in aphid-ant mutualism. Ecol Res 29:383–391. https://doi.org/10.1007/s11284-014-1151-4
Yao I (2019) Seasonal changes in the density of the symbionts Buchnera and Wolbachia of the aphid Tuberculatus macrotuberculatus on Quercus dentata. Entomol Exp Appl 167:261–268. https://doi.org/10.1111/eea.12743
Yao I, Akimoto SI (2001) Ant attendance changes the sugar composition of the honeydew of the drepanosiphid aphid Tuberculatus quercicola. Oecologia 128:36–43. https://doi.org/10.1007/s004420100633
Yao I, Akimoto SI (2002) Flexibility in the composition and concentration of amino acids in honeydew of the drepanosiphid aphid Tuberculatus quercicola. Ecol Entomol 27:745–752. https://doi.org/10.1046/j.1365-2311.2002.00455.x
Yao I, Shibao H, Akimoto SI (2000) Costs and benefits of ant attendance to the drepanosiphid aphid Tuberculatus quercicola. Oikos 89:3–10. https://doi.org/10.1034/j.1600-0706.2000.890101.x
Zhou W, Rousset F, O’Neill S (1998) Phylogeny and PCR-based classification of Wolbachia strains using wsp gene sequences. Proc R Soc Lond Ser B Biol Sci 265:509–515. https://doi.org/10.1098/rspb.1998.0324
Acknowledgements
I thank S. Sugimoto of Plant Protection Station and the members of SEHU for collecting aphids. I thank K. Yoshizawa, T. Kanbe, S. Suzuki, and S. Akimoto of Hokkaido University for providing help during this study. This work was supported by JSPS Kakenhi Grant Nos. 24570016 and 15K07210 for IY.
Funding
Funding was provided by Japan Society for the Promotion of Science (Grant Nos. 24570016, 15K07210)
Author information
Authors and Affiliations
Contributions
IY conceived the study, collected samples, carried out molecular work, analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The author declares that they have no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yao, I. Mutualism enhances Wolbachia infection rates in ant-attended Tuberculatus aphid species (Hemiptera: Aphididae). Evol Ecol 37, 627–643 (2023). https://doi.org/10.1007/s10682-023-10237-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-023-10237-5