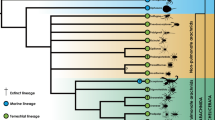
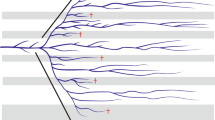
Abstract
A link between morphological diversity and species richness is often implied in several evolutionary concepts, but conflicting results hamper a more direct link between these variables. Using a morphologically and ecologically diverse clade of Neotropical cricetids, Akodontini, we (1) characterized the tribe’s patterns of morphological disparity and lineage diversification, contrasting the two major clades; and (2) tested whether morphological disparity and rates of morphological evolution are associated with their lineage diversification patterns. We found no correlation between diversification rates and morphological patterns; instead, our results reveal a pattern of ecological and morphological diversification that is independent of cladogenetic events. We found higher rates of morphological evolution in lineages with longer independent evolutionary histories, leading to fewer, but more disparate and specialized species occupying the peripheral areas of the ecomorphospace and increasing the overall morphological diversity of the group.






Similar content being viewed by others
References
Adams, D. C. (2014a). A generalized K statistic for estimating phylogenetic signal from shape and other high-dimensional multivariate data. Systematic Biology, 63(5), 685–697. https://doi.org/10.1080/10635150490889498
Adams, D. C. (2014b). Quantifying and comparing phylogenetic evolutionary rates for shape and other high-dimensional phenotypic data. Systematic Biology, 63(2), 166–177. https://doi.org/10.1093/sysbio/syt105
Adams, D. C., Berns, C. M., Kozak, K. H., & Wiens, J. J. (2009). Are rates of species diversification correlated with rates of morphological evolution? Proceedings of the Royal Society b: Biological Sciences, 276(1668), 2729–2738. https://doi.org/10.1098/rspb.2009.0543
Adams, D. C., Collyer, M., Kaliontzopoulou, A., & Sherratt, E. (2016). Geomorph: Software for geometric morphometric analyses. Retrieved June 15, 2019, from https://hdl.handle.net/1959.11/21330
Adams, D. C., Korneisel, D., Young, M., & Nistri, A. (2017). Natural history constrains the macroevolution of foot morphology in European plethodontid salamanders. The American Naturalist, 190(2), 292–297. https://doi.org/10.1086/692471
Agrawal, V. C. (1967). Skull adaptations in fossorial rodents. Mammalia, 31, 300–312. https://doi.org/10.1515/mamm.1967.31.2.300
Alhajeri, B. H., & Steppan, S. J. (2018). Disparity and evolutionary rate do not explain diversity patterns in muroid rodents (Rodentia: Muroidea). Evolutionary Biology, 45(3), 324–344. https://doi.org/10.1007/s11692-018-9453-z
Arbour, J. H., Curtis, A. A., & Santana, S. E. (2019). Signatures of echolocation and dietary ecology in the adaptive evolution of skull shape in bats. Nature Communications, 10(1), 1–13. https://doi.org/10.1038/s41467-019-09951-y
Arnqvist, G., & Martensson, T. (1998). Measurement error in geometric morphometrics: Empirical strategies to assess and reduce its impact on measures of shape. Acta Zoologica Academiae Scientiarum Hungaricae, 44(1–2), 73–96.
Astúa, D., Bandeira, I., & Geise, L. (2015). Cranial morphometric analyses of the cryptic rodent species Akodon cursor and Akodon montensis (Rodentia, Sigmodontinae). Oecologia Australis, 19(1), 143–157.
Bardua, C., Wilkinson, M., Gower, D. J., Sherratt, E., & Goswami, A. (2019). Morphological evolution and modularity of the caecilian skull. BMC Evolutionary Biology, 19(1), 1–23. https://doi.org/10.1186/s12862-018-1342-7
Bastide, P., Ané, C., Robin, S., & Mariadassou, M. (2018). Inference of adaptive shifts for multivariate correlated traits. Systematic Biology, 67(4), 662–680. https://doi.org/10.1093/sysbio/syy005
Bastos, A. D., Nair, D., Taylor, P. J., Brettschneider, H., Kirsten, F., Mostert, E., von Maltitz, E., Lamb, J. M., van Hooft, P., Belmain, S. R., Contrafatto, G., Downs, S., & Chimimba, C. T. (2011). Genetic monitoring detects an overlooked cryptic species and reveals the diversity and distribution of three invasive Rattus congeners in South Africa. BMC Genetics, 12(1), 1–18. https://doi.org/10.1186/1471-2156-12-26
Bezerra, A. M., & Pardiñas, U. F. J. (2016). Kunsia tomentosus (Rodentia: Cricetidae). Mammalian Species, 48(930), 1–9. https://doi.org/10.1093/mspecies/sev013
Blomberg, S. P., Garland, T., Jr., & Ives, A. R. (2003). Testing for phylogenetic signal in comparative data: Behavioral traits are more labile. Evolution, 57(4), 717–745. https://doi.org/10.1111/j.0014-3820.2003.tb00285.x
Brito, J., Tinoco, N., Pinto, C. M., García, R., Koch, C., Fernandez, V., Burneo, S., & Pardiñas, U. F. J. (2022). Unlocking Andean sigmodontine diversity: five new species of Chilomys (Rodentia: Cricetidae) from the montane forests of Ecuador. PeerJ, 10, e13211. https://doi.org/10.7717/peerj.13211
Bromham, L., Woolfit, M., Lee, M. S., & Rambaut, A. (2002). Testing the relationship between morphological and molecular rates of change along phylogenies. Evolution, 56(10), 1921–1930. https://doi.org/10.1111/j.0014-3820.2002.tb00118.x
Burbrink, F. T., Chen, X., Myers, E. A., Brandley, M. C., & Pyron, R. A. (2012). Evidence for determinism in species diversification and contingency in phenotypic evolution during adaptive radiation. Proceedings of the Royal Society B: Biological Sciences, 279(1748), 4817–4826. https://doi.org/10.1098/rspb.2012.1669
Camargo, N. F., Machado, L. F., Mendonça, A. F., & Vieira, E. M. (2019). Cranial shape predicts arboreal activity of Sigmodontinae rodents. Journal of Zoology, 308(2), 128–138. https://doi.org/10.1111/jzo.12659
Cardini, A. (2016). Lost in the other half: Improving accuracy in geometric morphometric analyses of one side of bilaterally symmetric structures. Systematic Biology, 65(6), 1096–1106. https://doi.org/10.1093/sysbio/syw043
Castiglione, S., Tesone, G., Piccolo, M., Melchionna, M., Mondanaro, A., Serio, C., Di Febbraro, M., & Raia, P. (2018). A new method for testing evolutionary rate variation and shifts in phenotypic evolution. Methods in Ecology and Evolution, 9(4), 974–983. https://doi.org/10.1111/2041-210X.12954
Cerca, J., Meyer, C., Stateczny, D., Siemon, D., Wegbrod, J., Purschke, G., Dimitrov, D., & Struck, T. H. (2020). Deceleration of morphological evolution in a cryptic species complex and its link to paleontological stasis. Evolution, 74(1), 116–131. https://doi.org/10.1111/evo.13884
Chira, A. M., & Thomas, G. H. (2016). The impact of rate heterogeneity on inference of phylogenetic models of trait evolution. Journal of Evolutionary Biology, 29(12), 2502–2518. https://doi.org/10.1111/jeb.12979
Ciampaglio, C. N., Kemp, M., & McShea, D. W. (2001). Detecting changes in morphospace occupation patterns in the fossil record: Characterization and analysis of measures of disparity. Paleobiology, 27(4), 695–715. https://doi.org/10.1666/0094-8373(2001)0272.0.CO;2
Claude, J., Pritchard, P. C., Tong, H., Paradis, E., & Auffray, J. C. (2004). Ecological correlates and evolutionary divergence in the skull of turtles: A geometric morphometric assessment. Systematic Biology, 53(6), 933–948. https://doi.org/10.1080/10635150490889498
Collar, D. C., Near, T. J., & Wainwright, P. C. (2005). Comparative analysis of morphological diversity: Does disparity accumulate at the same rate in two lineages of centrarchid fishes? Evolution, 59(8), 1783–1794. https://doi.org/10.1111/j.0014-3820.2005.tb01826.x
Cooney, C. R., & Thomas, G. H. (2021). Heterogeneous relationships between rates of speciation and body size evolution across vertebrate clades. Nature Ecology & Evolution, 5(1), 101–110. https://doi.org/10.1038/s41559-020-01321-y
D’Elía, G., & Pardiñas, U. F. J. (2015a). Subfamily Sigmodontinae Wagner, 1843. In J. L. Patton, U. F. J. Pardiñas, & G. D’Elía (Eds.), Mammals of South America (1st ed., pp. 63–688). University of Chicago Press.
D’Elía, G., & Pardiñas, U. F. J. (2015b). Tribe Akodontini Vorontsov 1959. In J. L. Patton, U. F. J. Pardiñas, & G. D’Elia (Eds.), Mammals of South America (1st ed., pp. 140–279). University of Chicago Press.
Eastman, J. M., Alfaro, M. E., Joyce, P., Hipp, A. L., & Harmon, L. J. (2011). A novel comparative method for identifying shifts in the rate of character evolution on trees. Evolution, 65(12), 3578–3589. https://doi.org/10.1111/j.1558-5646.2011.01401.x
Eldredge, N., & Gould, D. J. (1972). Punctuated equilibria: An alternative to phyletic gradualism. In T. Schopf (Ed.), Models in Paleobiology (pp. 82–115). Freeman Cooper.
Erwin, D. H. (2007). Disparity: Morphological pattern and developmental context. Palaeontology, 50(1), 57–73. https://doi.org/10.1111/j.1475-4983.2006.00614.x
Fabre, P. H., Hautier, L., Dimitrov, D., & Douzery, E. J. P. (2012). A glimpse on the pattern of rodent diversification: A phylogenetic approach. BMC Evolutionary Biology, 12(1), 1–19. https://doi.org/10.1186/1471-2148-12-88
Felice, R. N., Pol, D., & Goswami, A. (2021). Complex macroevolutionary dynamics underly the evolution of the crocodyliform skull. Proceedings of the Royal Society B, 288(1954), 20210919. https://doi.org/10.1098/rspb.2021.0919
Felsenstein, J. (1985). Phylogenies and the comparative method. The American Naturalist, 125(1), 1–15. https://doi.org/10.1086/284325
Foote, M. (1993a). Discordance and concordance between morphological and taxonomic diversity. Paleobiology, 19(2), 185–204. https://doi.org/10.1017/S0094837300015864
Foote, M. (1993b). Contributions of individual taxa to overall morphological disparity. Paleobiology, 19(4), 403–419. https://doi.org/10.1017/S0094837300014056
Foote, M. (1997). The evolution of morphological diversity. Annual Review of Ecology and Systematics, 28(1), 129–152. http://www.jstor.org/stable/2952489
Fruciano, C. (2016). Measurement error in geometric morphometrics. Development Genes and Evolution, 226(3), 139–158. https://doi.org/10.1007/s00427-016-0537-4
Geise, L., Bergallo, H. G., Esberard, C. E., Rocha, C. F., & Van Sluys, M. (2008). The karyotype of Blarinomys breviceps (Mammalia: Rodentia: Cricetidae) with comments on its morphology and some ecological notes. Zootaxa, 1907(1), 47–60. https://doi.org/10.11646/ZOOTAXA.1907.1.3
Geise, L., Smith, M. F., & Patton, J. L. (2001). Diversification in the genus Akodon (Rodentia: Sigmodontinae) in southeastern South America: Mitochondrial DNA sequence analysis. Journal of Mammalogy, 82(1), 92–101. https://doi.org/10.1644/1545-1542(2001)082%3c0092:DITGAR%3e2.0.CO;2
Gillespie, R. G., Bennett, G. M., De Meester, L., Feder, J. L., Fleischer, R. C., Harmon, L. J., Hendry, A. P., Knope, M. L., Mallet, J., Martin, C., Parent, C. E., Patton, A. H., Pfennig, K. S., Rubinoff, D., Schluter, D., Seehausen, O., Shaw, K. L., Stacy, E., Stervander, M., … Wogan, G. O. U. (2020). Comparing adaptive radiations across space, time, and taxa. Journal of Heredity, 111(1), 1–20. https://doi.org/10.1093/jhered/esz064
Gingerich, P. D. (2001). Rates of evolution on the time scale of the evolutionary process. In A. P. Hendry & M. T. Kinnison (Eds.), Microevolution rate, pattern, process (pp. 127–144). Springer.
Gittenberger, E. (1991). What about non-adaptive radiation? Biological Journal of the Linnean Society, 43(4), 263–272. https://doi.org/10.1111/j.1095-8312.1991.tb00598.x
Gonçalves, P. R., Myers, P., Vilela, J. F., & de Oliveira, J. A. (2007). Systematics of species of the genus Akodon (Rodentia: Sigmodontinae) in southeastern Brazil and implications for the biogeography of the campos de altitude. Miscellaneous Publications, Museum of Zoology, University of Michigan, 197, 1–24.
Hansen, T. F., Bolstad, G. H., & Tsuboi, M. (2022). Analyzing disparity and rates of morphological evolution with model-based phylogenetic comparative methods. Systematic Biology, 71(5), 1054–1072. https://doi.org/10.1093/sysbio/syab079
Hershkovitz, P. (1966). South American swamp and fossorial rats of the scapteromyine group (Cricetinae, Muridae) with comments on the glans penis in murid taxonomy. Zeitschrift Für Säugetierkunde, 31, 81–149.
Höhna, S., Landis, M. J., Heath, T. A., Boussau, B., Lartillot, N., Moore, B. R., Huelsenbeck, J. P., & Ronquist, F. (2016). RevBayes: Bayesian phylogenetic inference using graphical models and an interactive model-specification language. Systematic Biology, 65(4), 726–736. https://doi.org/10.1093/sysbio/syw021
Hopkins, M. J. (2013). Decoupling of taxonomic diversity and morphological disparity during decline of the Cambrian trilobite family Pterocephaliidae. Journal of Evolutionary Biology, 26(8), 1665–1676. https://doi.org/10.1111/jeb.12164
Hopkins, M. J. (2016). Magnitude versus direction of change and the contribution of macroevolutionary trends to morphological disparity. Biological Journal of the Linnean Society, 118(1), 116–130. https://doi.org/10.1111/bij.12759
Hopkins, M. J., & Gerber, S. (2018). Morphological disparity. In L. N. de la Rosa & G. B. Müller (Eds.), Evolutionary Developmental Biology: A reference guide (1st edn., pp. 965–976). Springer.
Hopkins, M. J., & Lidgard, S. (2012). Evolutionary mode routinely varies among morphological traits within fossil species lineages. Proceedings of the National Academy of Sciences USA, 109(50), 20520–20525. https://doi.org/10.1073/pnas.1209901109
Jones, K. E., Smaers, J. B., & Goswami, A. (2015). Impact of the terrestrial-aquatic transition on disparity and rates of evolution in the carnivoran skull. BMC Evolutionary Biology, 15(1), 1–19. https://doi.org/10.1186/s12862-015-0285-5
Kass, R. E., & Raftery, A. E. (1995). Bayes factors. Journal of the American Statistical Association, 90(430), 773–795. https://doi.org/10.1080/01621459.1995.10476572
Lee, M. S., Sanders, K. L., King, B., & Palci, A. (2016). Diversification rates and phenotypic evolution in venomous snakes (Elapidae). Royal Society Open Science, 3(1), 150277. https://doi.org/10.1098/rsos.150277
Louca, S., & Pennell, M. W. (2020). Extant timetrees are consistent with a myriad of diversification histories. Nature, 580(7804), 502–505. https://doi.org/10.1038/s41586-020-2176-1
Louca, S., & Pennell, M. W. (2021). Why extinction estimates from extant phylogenies are so often zero. Current Biology, 31(14), 3168–3173. https://doi.org/10.1016/j.cub.2021.04.066
Maestri, R., Luza, A. L., de Barros, L. D., Hartz, S. M., Ferrari, A., de Freitas, T. R. O., & Duarte, L. D. (2016b). Geographical variation of body size in sigmodontine rodents depends on both environment and phylogenetic composition of communities. Journal of Biogeography, 43(6), 1192–1202. https://doi.org/10.1111/jbi.12718
Maestri, R., Luza, A. L., Hartz, S. M., de Freitas, T. R. O., & Patterson, B. D. (2022). Bridging macroecology and macroevolution in the radiation of sigmodontine rodents. Evolution, 76(8), 1790–1805. https://doi.org/10.1111/evo.14561
Maestri, R., Monteiro, L. R., Fornel, R., Upham, N. S., Patterson, B. D., & de Freitas, T. R. O. (2017). The ecology of a continental evolutionary radiation: Is the radiation of sigmodontine rodents adaptive? Evolution, 71(3), 610–632. https://doi.org/10.1111/jbi.12718
Maestri, R., & Patterson, B. D. (2016). Patterns of species richness and turnover for the South American rodent fauna. PLoS ONE, 11(3), e0151895. https://doi.org/10.1371/journal.pone.0151895
Maestri, R., Patterson, B. D., Fornel, R., Monteiro, L. R., & De Freitas, T. R. O. (2016a). Diet, bite force and skull morphology in the generalist rodent morphotype. Journal of Evolutionary Biology, 29(11), 2191–2204. https://doi.org/10.1111/jeb.12937
Maestri, R., Upham, N. S., & Patterson, B. D. (2019). Tracing the diversification history of a Neogene rodent invasion into South America. Ecography, 42(4), 683–695. https://doi.org/10.1111/ecog.04102
Mammal Diversity Database. (2022). Mammal Diversity Database (Version 1.9.1) Rodentia. Zenodo. https://doi.org/10.5281/zenodo.4139818
Michaud, M., Toussaint, S. L. D., & Gilissen, E. (2022). The impact of environmental factors on the evolution of brain size in carnivorans. Communications Biology, 5, 998. https://doi.org/10.1038/s42003-022-03748-4|
Michaud, M., Veron, G., Peignè, S., Blin, A., & Fabre, A. C. (2018). Are phenotypic disparity and rate of morphological evolution correlated with ecological diversity in Carnivora? Biological Journal of the Linnean Society, 124(3), 294–307. https://doi.org/10.1093/biolinnean/bly047
Missagia, R. V., & Perini, F. A. (2018). Skull morphology of the Brazilian shrew mouse Blarinomysbreviceps (Akodontini; Sigmodontinae), with comparative notes on Akodontini rodents. Zoologischer Anzeiger, 277, 148–161. https://doi.org/10.1016/j.jcz.2018.09.005
Missagia, R. V., Patterson, B. D., Krentzel, D., & Perini, F. A. (2021). Insectivory leads to functional convergence in a group of Neotropical rodents. Journal of Evolutionary Biology, 34(2), 391–402. https://doi.org/10.1111/jeb.13748
Missagia, R. V., Patterson, B. D., & Perini, F. A. (2019). Stable isotope signatures and the trophic diversification of akodontine rodents. Evolutionary Ecology, 33(6), 855–872. https://doi.org/10.1007/s10682-019-10009-0
Morlon, H., Robin, S., & Hartig, F. (2022). Studying speciation and extinction dynamics from phylogenies: Addressing identifiability issues. Trends in Ecology & Evolution, 37, 497–506. https://doi.org/10.1016/j.tree.2022.02.004
Mundry, R. (2014). Statistical issues and assumptions of phylogenetic generalized least squares. In L. Z. Garamszegi (Ed.), Modern phylogenetic comparative methods and their application in evolutionary biology (1st edn., pp. 131–153). Springer.
Novacek, M. J. (1993). Patterns of diversity in the mammalian skull. In J. Hanken, & B. K. Hall (Eds.), The Skull 2: Patterns of Structural and Systematic Diversity (1st edn., pp. 438–545). The University of Chicago Press.
Ojeda, A. A., Teta, P., Pablo Jayat, J., Lanzone, C., Cornejo, P., Novillo, A., & Ojeda, R. A. (2021). Phylogenetic relationships among cryptic species of the Phyllotis xanthopygus complex (Rodentia, Cricetidae). Zoologica Scripta, 50(3), 269–281. https://doi.org/10.1111/zsc.12472
Parada, A., D’Elía, G., & Palma, R. E. (2015). The influence of ecological and geographical context in the radiation of Neotropical sigmodontine rodents. BMC Evolutionary Biology, 15(1), 1–17. https://doi.org/10.1186/s12862-015-0440-z
Pardiñas, U. F. J., D’Elía, G., & Ortiz, P. E. (2002). Sigmodontinos fosiles (Rodentia: Muroidea, Sigmodontinae) de America del Sur: Estado actual de su conocimiento y prospectiva. Mastozoologia Neotropical, 9, 209–252.
Pardiñas, U. F. J., Myers, P., León-Paniagua, L., Ordóñez-Garza, N., Cook, J., Kryštufek, B., Haslauer, R., Bradley, R., Shenbrot, G., & Patton, J. (2017). Family cricetidae. In D. E. Wilson, T. E. Lacher, & R. A. Mittermeier (Eds.), Handbook of the mammals of the world, volume 7: Rodents II (pp. 204–279). Lynx Editions.
Pardiñas, U. F. J., Teta, P., Alvarado-Serrano, D., Geise, L., Jayat, J. P., Ortiz, P. E., Gonçalves, P. R., & D’Elía, G. (2015). Genus Akodon Meyen, 1833. In J. L. Patton, U. F. J. Pardiñas, & G. D’Elía (Eds.), Mammals of South America (1st edn., pp. 144–204). University of Chicago Press.
Patterson, B. D. (2020). On drivers of Neotropical mammal diversification. Mastozoología Neotropical, 27, 15–26. https://doi.org/10.31687/saremMN_SI.20.27.1.03
Patton, J. L., Pardiñas, U. F. J., & D’Elía, G. (Eds.). (2015). Mammals of South America, volume 2: Rodents. University of Chicago Press.
Pennell, M. W., Eastman, J. M., Slater, G. J., Brown, J. W., Uyeda, J. C., FitzJohn, R. G., Alfaro, M. E., & Harmon, L. J. (2014b). geiger v2. 0: An expanded suite of methods for fitting macroevolutionary models to phylogenetic trees. Bioinformatics, 30(15), 2216–2218. https://doi.org/10.1093/bioinformatics/btu181
Pennell, M. W., Harmon, L. J., & Uyeda, J. C. (2014a). Is there room for punctuated equilibrium in macroevolution? Trends in Ecology & Evolution, 29(1), 23–32. https://doi.org/10.1016/j.tree.2013.07.004
Plummer, M., Best, N., Cowles, K., & Vines, K. (2006). CODA: Convergence diagnosis and output analysis for MCMC. R News, 6(1), 7–11.
Price, M. V. (1983). Ecological consequences of body size: A model for patch choice in desert rodents. Oecologia, 59(2), 384–392. https://doi.org/10.1007/BF00378866
R Core Team. (2022). R: A language and environment for statistical computing. R Core Team.
Rabosky, D. L. (2010). Extinction rates should not be estimated from molecular phylogenies. Evolution, 64(6), 1816–1824. https://doi.org/10.1111/j.1558-5646.2009.00926.x
Rabosky, D. L. (2014). Automatic detection of key innovations, rate shifts, and diversity-dependence on phylogenetic trees. PLoS ONE, 9(2), e89543. https://doi.org/10.1371/journal.pone.0089543
Rabosky, D. L., & Adams, D. C. (2012). Rates of morphological evolution are correlated with species richness in salamanders. Evolution, 66(6), 1807–1818. https://doi.org/10.1111/j.1558-5646.2011.01557.x
Rabosky, D. L., Grundler, M., Anderson, C., Title, P., Shi, J. J., Brown, J. W., Huang, H., & Larson, J. G. (2014). BAMM tools: An R package for the analysis of evolutionary dynamics on phylogenetic trees. Methods in Ecology and Evolution, 5(7), 701–707. https://doi.org/10.1111/2041-210X.12199
Rabosky, D. L., & Huang, H. (2016). A robust semi-parametric test for detecting trait-dependent diversification. Systematic Biology, 65(2), 181–193. https://doi.org/10.1093/sysbio/syv066
Rabosky, D. L., Santini, F., Eastman, J., Smith, S. A., Sidlauskas, B., Chang, J., & Alfaro, M. E. (2013). Rates of speciation and morphological evolution are correlated across the largest vertebrate radiation. Nature Communications, 4, 1958. https://doi.org/10.1038/ncomms2958
Rambaut, A., Drummond, A. J., Xie, D., Baele, G., & Suchard, M. A. (2018). Posterior summarization in Bayesian phylogenetics using Tracer 1.7. Systematic Biology, 67(5), 901–904. https://doi.org/10.1093/sysbio/syy032
Reig, O. A. (1987). An assessment of the systematics and evolution of the Akodontini, with the description of new fossil species of Akodon (Cricetidae: Sigmodontinae). In B. D. Patterson, & R. M. Timm (Eds.), Studies in Neotropical Mammalogy. Essays in honor of Philip Hershkovitz (pp. 347–399). Field Museum of Natural History, Fieldiana Zoology, 39.
Reis, J., Bidau, C. J., Maestri, R., & Martinez, P. A. (2018). Diversification of the climatic niche drove the recent processes of speciation in Sigmodontinae (Rodentia, Cricetidae). Mammal Review, 48(4), 328–332. https://doi.org/10.1111/mam.12128
Renaud, S., Chevret, P., & Michaux, J. (2007). Morphological vs. molecular evolution: Ecology and phylogeny both shape the mandible of rodents. Zoologica Scripta, 36(5), 525–535. https://doi.org/10.1111/j.1463-6409.2007.00297.x
Revell, L. J. (2012). phytools: An R package for phylogenetic comparative biology (and other things). Methods in Ecology and Evolution, 2, 217–223. https://doi.org/10.1111/j.2041-210X.2011.00169.x
Ricklefs, R. E. (2005). Small clades at the periphery of passerine morphological space. The American Naturalist, 165(6), 651–659. https://doi.org/10.1086/429676
Ricklefs, R. E. (2006). Time, species, and the generation of trait variance in clades. Systematic Biology, 55(1), 151–159. https://doi.org/10.1080/10635150500431205
Rohlf, F. J. (2015). The Tps series of software. Hystrix, 26(1), 9–12. https://doi.org/10.4404/hystrix-26.1-11264
Roycroft, E., MacDonald, A. J., Moritz, C., Moussalli, A., Miguez, R. P., & Rowe, K. C. (2021). Museum genomics reveals the rapid decline and extinction of Australian rodents since European settlement. Proceedings of the National Academy of Sciences, 118(27), e2021390118. https://doi.org/10.1073/pnas.202139011
Rowe, K. C., Aplin, K. P., Baverstock, P. R., & Moritz, C. (2011). Recent and rapid speciation with limited morphological disparity in the genus Rattus. Systematic Biology, 60(2), 188–203. https://doi.org/10.1093/sysbio/syq092
Rundell, R. J., & Price, T. D. (2009). Adaptive radiation, nonadaptive radiation, ecological speciation and nonecological speciation. Trends in Ecology & Evolution, 24(7), 394–399. https://doi.org/10.1016/j.tree.2009.02.007
Sakamoto, M., & Venditti, C. (2018). Phylogenetic non-independence in rates of trait evolution. Biology Letters, 14(10), 20180502. https://doi.org/10.1098/rsbl.2018.0502
Samuels, J. X. (2009). Cranial morphology and dietary habits of rodents. Zoological Journal of the Linnean Society, 156(4), 864–888. https://doi.org/10.1111/j.1096-3642.2009.00502.x
Samuels, J. X., & van Valkenburgh, B. (2008). Skeletal indicators of locomotor adaptations in living and extinct rodents. Journal of Morphology, 269(11), 1387–1411. https://doi.org/10.1002/jmor.10662
Schluter, D. (2000). The ecology of adaptive radiation. Oxford University Press.
Sidlauskas, B. (2008). Continuous and arrested morphological diversification in sister clades of characiform fishes: A phylomorphospace approach. Evolution, 62(12), 3135–3156. https://doi.org/10.1111/j.1558-5646.2008.00519.x
Slater, G. J., Price, S. A., Santini, F., & Alfaro, M. E. (2010). Diversity versus disparity and the radiation of modern cetaceans. Proceedings of the Royal Society b: Biological Sciences, 277(1697), 3097–3104. https://doi.org/10.1098/rspb.2010.0408
Stayton, C. T. (2005). Morphological evolution of the lizard skull: A geometric morphometrics survey. Journal of Morphology, 263(1), 47–59. https://doi.org/10.1002/jmor.10288
Stein, B. R. (2000). Morphology of subterranean rodents. In E. A. Lacey, J. L. Patton, & J. N. Cameron (Eds.), Life underground: The biology of subterranean rodents (pp. 19–61). University of Chicago Press.
Suárez-Villota, E. Y., Carmignotto, A. P., Brandão, M. V., Percequillo, A. R., & Silva, M. J. D. J. (2018). Systematics of the genus Oecomys (Sigmodontinae: Oryzomyini): Molecular phylogenetic, cytogenetic and morphological approaches reveal cryptic species. Zoological Journal of the Linnean Society, 184(1), 182–210. https://doi.org/10.1093/zoolinnean/zlx095
Tavares, W. C., Coutinho, L. C., & de Oliveira, J. A. (2021). Locomotor habits and phenotypic evolution of the appendicular skeleton in the oryzomyalian radiation in the Neotropics (Sigmodontinae, Cricetidae, Rodentia). Journal of Zoological Systematics and Evolutionary Research, 59(8), 2457–2480. https://doi.org/10.1111/jzs.12551
Upham, N. S., Esselstyn, J. A., & Jetz, W. (2019). Inferring the mammal tree: Species-level sets of phylogenies for questions in ecology, evolution, and conservation. PLoS Biology, 17(12), e3000494. https://doi.org/10.1371/journal.pbio.3000494
Venditti, C., Meade, A., & Pagel, M. (2011). Multiple routes to mammalian diversity. Nature, 479(7373), 393–396. https://doi.org/10.1038/nature10516
Zelditch, M. L., Li, J., & Swiderski, D. L. (2020). Stasis of functionally versatile specialists. Evolution, 74(7), 1356–1377. https://doi.org/10.1111/evo.13956
Zelditch, M. L., Li, J., Tran, L. A., & Swiderski, D. L. (2015). Relationships of diversity, disparity, and their evolutionary rates in squirrels (Sciuridae). Evolution, 69(5), 1284–1300. https://doi.org/10.1111/evo.12642
Zelditch, M. L., Swiderski, D. L., Sheets, H. D., & Fink, W. L. (2012). Geometric morphometrics for biologists: A primer (2nd ed.). Elsevier/Academic Press.
Acknowledgements
We thank the curators of the mammal collections who allowed access to specimens under their care (AMNH, Robert Voss; USNM, Darrin Lunde; MCNM, Claudia Costa; MN, João Oliveira). We kindly thank Fábio Machado (Oklahoma State University) for insightful discussions on geometric morphometrics and phylogenetic comparative methods during the development of this study; and Caroline Oswald (Universidade Federal de Santa Catarina) for reading a preliminary version of the manuscript. The insightful comments of the editor and two anonymous reviewers have greatly contributed to improving the final version of the manuscript.
Funding
This work was made possible with financial support of Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES—Finance Code 0001) which granted RVM with regular and sandwich fellowships (88881.133833/2016-1). We also thank Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG, APQ-02066-21) and the Grant #2022/00044-7, São Paulo Research Foundation (FAPESP), for the financial support to RVM and DMC, respectively.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analyses were performed by RVM and DMC. The first draft of the manuscript was written by RVM and DMC, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
11692_2022_9596_MOESM5_ESM.csv
Supplementary file Summary of raw values for disparity, size, morphological evolutionary rates, speciation and extinction rates for all taxa sampled in the present study. Variable names as in Figure 4 legend. (CSV 10 kb)
11692_2022_9596_MOESM9_ESM.xlsx
Supplementary file Posterior probabilities (PP) for the estimated number of rate shifts in BAMM diversification rates analyses and the 95% Highest Posterior Density (HPD) interval for rate shift configurations and their associated PPs. (XLSX 11 kb)
11692_2022_9596_MOESM11_ESM.xlsx
Supplementary file Speciation and extinction rates AOV tables, for comparison of clades A and B, and comparisons of Akodon and all other akodonties. (XLSX 11 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Missagia, R.V., Casali, D.M., Patterson, B.D. et al. Decoupled Patterns of Diversity and Disparity Characterize an Ecologically Specialized Lineage of Neotropical Cricetids. Evol Biol 50, 181–196 (2023). https://doi.org/10.1007/s11692-022-09596-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11692-022-09596-8