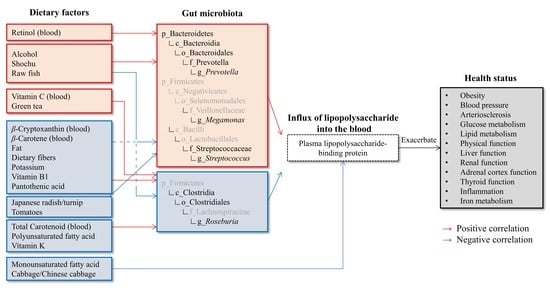
In this study, we conducted a cross-sectional study to investigate the association of dietary factors, gut microbiota, and health status with plasma LBP concentration as an index of blood LPS influx in general adult population. Results showed that some of clinical markers, not only for lifestyle-related diseases, but also for what had not been reported to be correlated with blood LPS in previous case-control studies, were also correlated with plasma LBP concentration. Additionally, we observed the relationships between food and nutrition intake in daily life and plasma LBP concentration, those had not been evaluated in previous intervention studies. Furthermore, plasma LBP concentration was correlated with four bacterial genera and their higher groups, and these intestinal bacteria were associated with some of the dietary factors above-mentioned. The individual relationships are discussed below.
4.2. Health Statuses Correlated with Plasma LBP Concentration
Previous research reported that blood LPS concentration or blood LBP concentration was correlated with obesity [
15,
17,
18], glucose metabolism [
15,
17], lipid metabolism [
15,
17,
18], vascular function [
15,
17,
32,
33], liver function [
34,
35], and inflammatory conditions [
15,
17]. The present study also found a significant relationship between plasma LBP concentration and these variables. This study also found that plasma LBP concentration was correlated with muscle mass, blood renin activity, blood cortisol concentration, blood free T4 concentration, and serum iron.
A decrease in muscle mass increases the risk of falls, need for nursing care, risk of chronic diseases, and risk of all-cause mortality in old age [
36], so maintaining muscle mass is important in the elderly. In addition, a gradual decline in muscle mass among Japanese people from their late 30s has been reported [
37], highlighting the importance of identifying the causes of muscle weakness and maintaining muscle strength not only in the elderly but also in young people. Evidence indicates that sepsis [
38] and administration of LPS in animal models [
39] induce muscle atrophy. However, epidemiological studies have only reported a correlation between blood LBP concentration and decreased skeletal muscle density in elderly men [
16]. The present study included both men and women across a wide age range, and a negative correlation was observed between plasma LBP concentration and muscle mass, independent of age or sex. This suggests that blood LPS influx may results in a decrease in muscle mass from a young age.
Previous research reported that a higher blood LBP concentration in patients with cirrhosis and ascites resulted in increased blood renin activity, and that the administration of the antibacterial drug norfloxacin decreased blood LBP concentration and renin activity [
40]. However, the relationship between LPS exposure and blood renin activity in the general adult population was unclear. In the present study, a significant positive correlation was observed between plasma LBP concentration and plasma renin activity, suggesting that LPS exposure is also associated with increased plasma renin activity in the general adult population. Renin is well known to constrict blood vessels and regulate blood pressure via the renin-angiotensin system [
41]. Previous research and the present study have shown that blood LBP concentration [
17] or blood LPS concentration [
32] were positively correlated with blood pressure, and the activation of the renin-angiotensin system resulting from exposure to LPS may partly explain this.
In the present study, plasma LBP concentration was significantly positively correlated with blood cortisol concentration. Previous research showed in an in vitro study that LPS directly acts on adrenal cells and promote cortisol secretion [
42]. Additionally, intravenous administration of LPS in humans increased blood cortisol concentration [
43,
44], suggesting that LPS promotes cortisol secretion. However, to date, the relationship between cortisol concentration and plasma LBP concentration in the general adult population remains unclear. Cortisol has been shown to contribute to increased blood pressure through the regulation of capillary function [
45], and like renin, cortisol may be involved in blood pressure elevation upon exposure to LPS. Cortisol is also a well-known stress response hormone and has been found to be associated with depression [
46]. In the present study, the score of Center for Epidemiological Studies Depression (CES-D) was used to evaluate depression, but no significant correlation was found between the total CES-D score and blood LBP concentration. Participants in the present study had a total CES-D score of 14 even in the third quartile. In CES-D, a score of 16 or higher is defined as a depressive state, so at least three quarters of the participants in this study were considered to be in a non-depressive state. Therefore, the present study was not suitable for evaluating the correlation between blood LBP concentration and depression. The correlation between increased plasma LBP concentration and increased blood cortisol level-mediated worsening of the depressive state needs to be re-examined in cross-sectional studies involving subjects with different depressive states.
It has been previously reported that sepsis induces apoptosis of thyroid epithelial cells, decreased thyroid function, and decreased free T4, which is a thyroid hormone [
47,
48]. However, the effect of metabolic endotoxemia on thyroid function has not been elucidated. In the present study, we showed for the first time that there was a positive correlation between plasma LBP concentration and free T4 concentration. Free T4 secretion from the thyroid gland is controlled by the hypothalamic–pituitary–thyroid axis (HPT axis), which is affected by cortisol [
49]. However, in the present study, no significant correlation was found between thyrotropin, which constitutes the HPT axis, and plasma LBP concentration. Therefore, it is possible that LPS exposure is involved in the regulation of free T4 secretion through a pathway that does not involve the HPT axis.
We observed negative correlations between plasma LBP concentration and serum iron. Iron in the body is distributed in the liver/spleen, red blood cells, and blood; ferritin, hemoglobin, and serum iron serve as their respective indices. Among these, plasma LBP concentration was significantly correlated only with serum iron. Iron carried in the blood includes iron bound to transferrin that is produced by the liver and iron that is not, and the serum iron measured in the present study refers the former. Previous research has reported that LPS stimulation suppresses transferrin secretion from liver cells [
50], suggesting that the negative correlation between plasma LBP concentration and serum iron that was observed in the present study may be related to the suppression of transferrin production. Iron bound to transferrin plays an important role in neuronal metabolism in the brain [
51]. Low serum iron has been reported in patients with Alzheimer’s disease compared to healthy subjects [
52], and low serum iron that is correlated with plasma LBP concentration may lead to decreased brain function. In the present study, the score of Mini-Mental state examination (MMSE) was used to evaluate cognitive function, although no significant correlations were found between the plasma LBP concentration and MMSE score. The total MMSE score of participants in the present study was 29 even in the first quartile. A MMSE score of 26 or less is interpreted as mild dementia, indicating that at least three-fourths of the participants in the present study did not experience any decline in cognitive function. Therefore, the present study was not suitable for evaluating the relationship between plasma LBP concentration and cognitive function. The relationship between elevated LBP concentration and cognitive decline needs to be re-examined in cross-sectional studies involving study subjects with different cognitive functions.
4.3. Dietary Factors Correlated to Plasma LBP Concentration
Here, we discuss the present study’s consistency with previous research and the mechanism of action of the dietary factors that were found to be associated with plasma LBP concentration. The mechanism mediated by intestinal bacteria is described in the section “Relationship between dietary factors and intestinal bacteria” below.
The following dietary factors were found to be correlated with reduced exposure to LPS: dietary fiber [
1], MUFA [
53], PUFA [
53], total carotenoids [
54], and
β-carotene [
54]. Our findings are consistent with these results. Additionally, the present study found that plasma LBP concentration was negatively correlated with cabbage/Chinese cabbage intake, Japanese radish/turnip intake, tomato intake, potassium intake, vitamin B1 intake, pantothenic acid intake, vitamin K intake, lipid intake, and serum
β-cryptoxanthin concentration.
Cabbage/Chinese cabbage and Japanese radish/turnips are cruciferous vegetables. In a previous study, we reported that extracts of broccoli sprouts, a type of cruciferous vegetable, improved gut microbiota in mice that were fed a high-fat diet, thereby reducing blood LPS concentration [
55]. Furthermore, in mice, isothiocyanates that derived from cruciferous vegetables were shown to normalize the intestinal barrier function [
56]. Ingestion of cruciferous vegetables may have suppressed the influx of LPS into the blood through this mechanism.
Lycopene, which is one of the carotenoids characteristically contained in tomatoes, has been reported to suppress intestinal inflammation and lower blood LPS concentration in mice [
57]. Therefore, the correlation between tomato intake and plasma LBP concentration observed in the present study may reflect the efficacy of lycopene. However, in the present study, there was no significant correlation between serum lycopene concentration and plasma LBP concentration. Evidence indicates that lycopene is considerably attenuated by digestive juices [
58], so there may have been a discrepancy between the action of lycopene in the intestinal tract and the serum lycopene concentration. Additionally, a comprehensive analysis of the components contained in 25 varieties of tomatoes using liquid chromatography tandem-mass spectrometry resulted in the detection of 7118 peaks, of which 1577 were annotated [
59]. Many of these are components with anti-inflammatory effects [
60]. Therefore, it is thought that components other than lycopene contained in tomatoes may also contribute to the maintenance of the intestinal barrier function and the suppression of metabolic endotoxemia.
Fat promotes LPS influx, although its effect on blood LPS concentration differs depending on its origin and composition [
53]. MUFA have the potential to reduce blood LPS concentration. Results of participant diet analysis showed that the composition ratio of MUFA was the highest among all the fatty acids ingested (
Table S3). Therefore, the effect of this may have been a negative correlation between fat intake and plasma LBP concentration.
β-cryptoxanthin is a carotenoid that is contained in mandarin oranges, paprika, and persimmons. Blood concentration of
β-cryptoxanthin have been reported to show negative correlations with severity of NAFLD [
61], HOMA-IR [
62], and BMI [
63] in humans.
β-cryptoxanthin concentration has also been reported to be negatively correlated with the lactulose:mannitol (L/M) ratio, which is an index of intestinal barrier function [
64]. Provitamin A carotenoids, including
β-cryptoxanthin, are cleaved in the intestine by
β-carotene oxygenase 1, which results in the formation of vitamin A [
65]. Vitamin A has been suggested to contribute to the maintenance of the intestinal barrier function [
66]. Therefore,
β-cryptoxanthin may have affected plasma LBP concentration through its provitamin A properties.
Potassium intake or pantothenic acid intake has been suggested to be contribute the reduction of intestinal inflammation [
67] or the maintenance of intestinal barrier function [
68], respectively. There is limited evidence on vitamin B1 and the intestinal barrier function. However, vitamin B1 deficiency was found to cause sepsis-like conditions [
69], suggesting that vitamin B1 may contribute to maintaining the intestinal barrier function. The association of vitamin K and intestinal permeability, barrier function, and inflammation has not been previously reported and further research is necessary to investigate this relationship.
Alcohol is associated with increased LPS exposure [
70], and in the present study a significantly positive correlation was found between the intake of alcohol and shochu, which is a type of Japanese distilled liquor, and plasma LBP concentration. Furthermore, our study found that plasma LBP concentration was positively correlated with green tea intake, raw fish intake, plasma vitamin C concentration, and serum retinol concentration.
Catechin, which is a flavonoid contained in green tea, may be beneficial in human health through its antioxidant and anti-inflammatory effects [
71]. Green tea extract suppresses metabolic endotoxemia in mice [
72,
73]. This suggests that it is unlikely that green tea itself increases blood LPS concentration. Meanwhile, epidemiological studies have reported that people with a dietary habit of consuming both green tea and alcohol have an increased risk of esophageal cancer [
74] and colorectal cancer [
75]. In other words, the ingestion of green tea and alcohol may damage the epithelium of the gastrointestinal tract, which may reduce the intestinal barrier function and promote LPS influx.
BDHQ, which was used in the dietary survey of the present study, investigated three methods of cooking seafood (raw, grilled, boiled), and only raw fish was found to have a significant correlation with plasma LBP concentration. Therefore, it is postulated that eating raw fish, not the fish itself, increases plasma LBP concentration. Raw fish consumption is the cause of food poisoning caused by
Vibrio parahaemolyticus. PCR testing detected
Vibrio parahaemolyticus at a frequency of 13–97% in commercially available seafood [
76]. Therefore, it is presumed that consumption of raw fish leads to ingestion of
Vibrio parahaemolyticus toxin in trace amounts, even if it does not lead to food poisoning. The
Vibrio parahaemolyticus toxin has been reported to damage intestinal epithelial cells [
77], so its ingestion may contribute to increased intestinal permeability and blood LPS influx. It is also known that preserving raw fish causes histamine production by the bacteria present on the fish, causing health hazards to humans if the fish is eaten (i.e., histamine fish poisoning). It has been confirmed that histamine content increases during storage in fish eaten in Japan [
78]. Oral histamine has been suggested to increase intestinal permeability [
79]. Therefore, it is possible that histamine fish poisoning is one of the reasons for the positive correlation between raw fish intake and plasma LBP concentration. Another possibility is that Japanese people with a diet pattern that consumes large quantities of alcohol often consume fish dishes [
80,
81], so it should be noted that the association between raw fish intake and plasma LBP concentration may be a pseudo-correlation that reflects the amount of alcohol consumed.
Vitamin C [
82] and vitamin A [
83] maintain the intestinal barrier function, and we consider that it is unlikely that these directly contributed to the increase in blood LPS concentration. It has been reported that vitamin C intake consist of 10–11% from green tea, and vitamin A intake consist of 30–41% from fish/shellfish in Japanese [
84]. Thus, it is possible that the blood concentrations of these two components may indirectly correlate with the plasma LBP concentration due to the association with green tea or raw fish intake.
4.4. Correlation between Plasma LBP Concentration and Intestinal Bacteria
In the present study, plasma LBP concentration was positively correlated with genus Prevotella and its higher groups (phylum Bacteroidetes, class Bacteroidia, order Bacteroidales, family Prevotellaceae), genus Megamonas, genus Streptococcus and its higher group (family Streptococcaceae); and negatively correlated with genus Roseburia and its higher groups (class Clostridia, order Clostridiales).
The correlation between the composition ratio of
Prevotella and blood LPS concentration was reported in patients with type 2 diabetes [
85,
86].
Prevotella produces succinate as a result of glucose metabolism [
87]. Succinate from intestinal bacteria supports the growth of the pathogenic bacteria
Salmonella serovar Typhimurium [
88] and
Clostridium difficile [
89]. Furthermore, succinate induces colitis and promotes colonic fibrosis through succinate receptors [
90].
Prevotella has also been reported to utilize intestinal mucin [
91,
92] and increases in mucin-utilizing intestinal bacteria cause disruption of the mucin layer and intestinal inflammation [
93]. Therefore, in terms of its nutrient requirements,
Prevotella may also disrupt the intestinal barrier.
Megamonas was first detected in human feces in 2008 [
94], and little is known about its behavior in the human intestinal tract. However, a study comparing the gut microbiota of urban and rural elderly found that gut microbiota composition in the urban elderly had a higher proportion of
Megamonas and higher fecal LPS concentration [
95]. Furthermore, the administration of Kampo medicine (Gegen Qinlian decoction) to patients with colorectal cancer caused a decrease of
Megamonas among gut microbiota and an improvement in intestinal barrier function [
96]. These findings suggest that
Megamonas is involved in the influx of LPS in the intestinal tract, although the underlying mechanism has not been elucidated.
Streptococcus is a Gram-positive coccus to which many pathogenic bacteria belong. Patients with alcoholic liver disease have a higher composition ratio of
Streptococcus in the fecal flora, intestinal permeability, and blood LBP concentration compared to healthy subjects [
97].
Streptococcus is a bacterium that is abundant in the oral cavity; since the composition ratio in the intestinal tract increases in patients treated with proton pump inhibitors [
98], it is thought that
Streptococcus in the oral cavity reaches the intestinal tract due to the decreased digestive capacity of the stomach. This influx of
Streptococcus has also been suggested to induce an inflammatory response in the colon [
99].
The gut microbiota composition in patients with type 1 diabetes with high intestinal permeability and high blood LPS concentration is characterized by a decreased proportion of
Roseburia [
100].
Roseburia has been shown to strengthen the intestinal barrier function in mice through the production of butyrate and to suppress LPS influx [
101]. Regarding the balance of intestinal bacteria at the order level, in the present study, a negative correlation was found between the composition ratio of order Clostridiales and plasma LBP concentration, and a positive correlation was found between the composition ratio of order Bacteroidales and plasma LBP concentration. A previous study comparing gut microbiota of rural and urban children in Thailand found that rural children had the following characteristics [
102]: (1) a high composition ratio of order Clostridiales, a low composition ratio of order Bacteroidales; (2) presence of genes involved in the metabolism of plant-derived components and butyric acid production; and (3) butyric acid concentration in feces was high. As mentioned above, butyric acid enhances the barrier function of the intestinal tract. Therefore, order Clostridiales is considered to be a group of bacteria that contribute to the suppression of LPS influx into the blood via butyrate production. We postulated that the balance of intestinal bacteria at the order level observed in the present study has important significance in controlling the influx of LPS into the blood.
4.5. Relationship between Dietary Factors and Intestinal Bacteria
In the present study, we showed a positive correlation between serum retinol concentration and order Bacteroidales. In an interventional study in which autistic children were given supplements that contained vitamin A, vitamin A intake increased the composition ratio of Bacteroidales [
103]. However, there is little knowledge about the mechanism by which vitamin A increases Bacteroidales, and further investigation is needed.
In the present study, shochu and alcohol intake were associated with an increase in genus
Prevotella and genus
Megamonas, and a decrease in order Clostridiales. Previous research has also reported that alcohol intake was correlated with an increase in the order Bacteroidales, which is a higher classification of genus
Prevotella [
104]; increase in the genus
Megamonas [
105]; and decrease in order Clostridiales [
104]. A possible mechanism is that there are differences in alcohol tolerance among these bacteria. Oral bacteria such as
Prevotella possess alcohol dehydrogenase and have been reported to acquire alcohol tolerance by metabolizing alcohol to produce acetaldehyde [
106]. Meanwhile,
Clostridium belonging to Clostridiales has been reported to have low alcohol tolerance due to weak alcohol dehydrogenase activity [
107]. Based on these reports, we postulate that alcohol intake exerts a selective pressure on intestinal bacteria with low alcohol tolerance, which may affect the composition ratio of the gut microbiota.
The intake of raw fish dishes was correlated with an increase in the order Bacteroidales and a decrease in the genus
Roseburia. Since few countries have a custom of eating raw fish, there are no reports identifying the relationships between raw fish dishes and gut microbiota. One study found that humans with a diet consisting mainly of fish had a high composition ratio of family Bacteroidetes in the gut microbiota and low composition ratio of
Roseburia [
108]. However, people whose diet mainly includes fish also consume large quantities of protein foods, refined carbohydrates, vegetables, fruit, juice and sweetened beverages, kid’s meals and snacks and sweets, so the relationship between changes in gut microbiota and fish intake is unclear. In the present study, the pattern of intestinal bacteria associated with the intake of raw fish was similar to that associated with the intake of shochu and alcohol. As mentioned above, Japanese people with a diet pattern that consumes large quantities of alcohol often consume fish dishes. Therefore, there is a need to pay attention to the possibility of a pseudo-correlation that reflects alcohol intake as a background factor for the correlation between raw fish intake and order Bacteroidales as well as genus
Roseburia.
Serum
β-carotene and serum
β-cryptoxanthin concentrations were both negatively correlated with genus
Streptococcus and positively correlated with order Clostridiales. There was also a negative correlation between
β-carotene and genus
Megamonas. However, no previous reports were found regarding the mechanism by which
β-cryptoxanthin and
β-carotene regulate these bacteria. These carotenoids are abundantly contained in vegetables and fruits. As will be described later, dietary fiber intake that is contained in vegetable and fruit intake are reported to be associated with these intestinal bacteria. Therefore, it is postulated that the correlation between
β-cryptoxanthin and
β-carotene and intestinal bacteria may be a pseudo-correlation that reflects dietary fiber intake. However, there is an interesting report regarding order Clostridiales. In a previous report comparing the intestinal microbiota of normal subjects and atherosclerosis patients, it was shown that a group of bacteria including Clostridia living in the intestines of normal subjects has phytoene dehydrogenase and may produce
β-carotene from phytoene in the intestines [
109]. In this paper, it is also shown that the blood
β-carotene concentration of healthy individuals is actually higher than that of atherosclerotic patients, suggesting that carotenoids produced by intestinal bacteria may be absorbed from the intestinal tract and help maintain health. In the present study,
β-carotene intake that was calculated from BDHQ was not correlated with plasma LBP concentration, raising questions about the origin of serum
β-carotene. One possible explanation is that bacteria belonging to the order Clostridiales may produce
β-carotene.
Potassium and vitamin K were both positively correlated with order Clostridiales. However, the mechanism by which potassium regulates Clostridiales has not been previously reported. Potassium is abundant in vegetables, so it is possible that a pseudo-correlation that was mediated by dietary fiber was observed, similar to
β-cryptoxanthin and
β-carotene. Meanwhile, vitamin K has been reported to support the growth of
Faecalibacterium belonging to the order Clostridiales [
110]. Additionally, in this study, a negative correlation was found between potassium intake and genus
Streptococcus. High concentrations of potassium have been reported to suppress the expression of the pyruvate transporter required for the survival of
Streptococcus mutans [
111], suggesting that potassium intake inhibited the survival of
Streptococcus.
Vitamin B1 was negatively correlated with the composition ratios of genus
Prevotella and genus
Megamonas, and positively correlated with the composition ratios of order Clostridiales. To the best of our knowledge, there are no reports demonstrating the relationship between vitamin B1 and
Prevotella and
Megamonas. On the other hand, it has been reported that vitamin B1 plays an important role in the growth of Clostridiales bacteria [
112].
Pantothenic acid was positively correlated with class Clostridia and order Clostridiales; and negatively correlated with family Prevotellaceae. Pantothenic acid is a component of coenzyme A (CoA), which is essential for energy acquisition by the glycolytic system, not only in order Clostridiales. CoA is required for the production of butyric acid by bacteria belonging to Clostridiales inhabiting the human intestinal tract [
113]. It has been suggested that bacteria belonging to Clostridiales colonize and survive in intestinal mucin [
114]. Butyric acid is known to promote mucin production from intestinal epithelial cells and contributes to the adhesion of bacteria to the mucin layer [
115]. It is inferred from the above that one of the factors that may have contributed to the positive correlation of pantothenic acid with order Clostridiales was its contribution to the settlement of bacteria belonging to Clostridiales in the intestinal tract through the promotion of butyric acid and mucin production. Regarding the relationship between pantothenic acid and Prevotellaceae, one report indicates that pantothenic acid intake increases the composition ratio of
Prevotella, which belongs to Prevotellaceae, among intestinal bacteria [
116]. However, the underlying mechanisms is unclear.
Dietary fiber was negatively correlated with the composition ratios of order Bacteroidales and genus
Streptococcus; and positively correlated with the composition ratio of order Clostridiales. Bacteria belonging to the Order Bacteroidales, including
Prevotella, are characterized by their ability to utilize with both dietary fiber [
117] and mucin [
91,
92]. It has been reported that such bacteria express mucin-degrading genes when host dietary fiber intake is reduced, then switch nutrient sources, and increase the composition ratio [
93]. Therefore, it is postulated that high dietary fiber intake be related to the decrease in the composition ratio of order Bacteroidales.
Streptococcus is thought to flow into the intestinal tract when the digestive capacity of the stomach declines, as mentioned above. Dietary fiber intake increases the gastric residence time of food [
118].
Streptococcus has low gastric acid resistance, and its viability may significantly decrease with prolonged residence time in the stomach [
119]. These reports suggest that dietary fiber intake inhibits the survival of
Streptococcus by retaining it in the stomach, contributing to the decrease in its composition ratio in the intestinal tract. Bacteria belonging to the order Clostridiales have been reported to proliferate by utilizing dietary fiber [
120,
121,
122], so it is thought that a positive correlation was observed between dietary fiber intake and order Clostridiales.
Fat intake was negatively correlated with family Prevotellaceae and genus
Megamonas, and positively correlated with order Clostridiales. An epidemiological study conducted on Leyte Island showed that the lipid ratio in energy intake was negatively correlated with the composition ratio of family Prevotellaceae and positively correlated with the order Clostridiales [
123]. Additionally, animal fat intake has been reported to decrease the composition ratio of
Megamonas [
124]. However, the mechanism by which lipids regulate the composition ratio of these bacteria is unknown.
PUFA intake was positively correlated with order Clostridiales. PUFAs may have antibacterial activity against specific bacteria and may inhibit biofilm formation [
125], although Clostridiales may metabolize PUFAs [
126], avoiding their antibacterial effects [
125]. Therefore, the order Clostridiales may be more likely to survive in the intestines of humans with high PUFA intake, and composition ratios may be higher in the gut microbiota.
Japanese radish/turnip intake was negatively correlated with the genus
Streptococcus. It has been reported that the 5-methylsulfinyl-1-(4-methylsulfinyl-but-3-enyl)-pent-4-enylidene]-sulfamic acid in radishes has antibacterial activity against
Streptococcus [
127]. Isothiocyanate, which is abundant in cruciferous vegetables such as Japanese radishes and turnips, also has an antibacterial effect on
Streptococcus [
128].
Tomato intake was negatively correlated with genus Streptococcus. However, no previous reports have been found on the association of tomatoes and their characteristic component, lycopene, with Streptococcus, and further verification is needed to understand the mechanism.
We postulate that a mechanism that is mediated by intestinal bacteria underpins the correlation between dietary factors and plasma LBP concentration observed in the present study may be. However, many correlations have been observed that cannot be fully explained by previous reports. Clarifying the relationship between dietary factors and gut microbiota requires verification experiments using experimental models that reflect the intestinal ecosystem.