Abstract
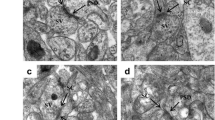
Glyphosate is the active ingredient of several widely used herbicide formulations. Studies based on Glyphosate exposure in different experimental models have suggested that the nervous system represented a key target for its toxicity. Previously, we demonstrated that exposure to glyphosate during gestation induces deficits on behavioral and cognitive function in rats. The aim of the present work was to examine whether cognitive dysfunction induced by Glyphosate was connected to changes on synapse formation and maturation. To understand how glyphosate affects synaptic assembly, we performed in vitro assays on cultured hippocampal neurons that were exposed to the herbicide (0.5 or 1 mg/mL) for 5 or 10 days. Biochemical and immunocytochemical approaches revealed that Glyphosate treated neurons showed a decrease on dendritic complexity and synaptic spine formation and maturation. Moreover, results indicated that Glyphosate decreased synapse formation in hippocampal neurons. To evaluate these effects in vivo, pup rats were treated with 35 or 70 mg/kg of Glyphosate from PND 7 to PND 27, every 48 h. Results indicated that Glyphosate postnatal exposure induced cognitive impairments, since recognition and spatial memory were altered. To go further, we evaluated synaptic protein expression and synaptic organization in hippocampus. Images revealed that Glyphosate treatment downregulates synapsin-1, PSD-95, and CaMKII expression, and also decreased PSD-95 clustering in hippocampus. Taken together, these findings demonstrate for the first time that Glyphosate exposure affects synaptic assembly and reduced synaptic protein expression in hippocampus and that likely triggers the impairment of cognitive function and neuronal connectivity.





Similar content being viewed by others
References
Ait-Bali Y, Ba-M’hamed S, Gambarotta G, et al (2020) Pre- and postnatal exposure to glyphosate-based herbicide causes behavioral and cognitive impairments in adult mice: evidence of cortical ad hippocampal dysfunction. Arch Toxicol 94:1703–1723. https://doi.org/10.1007/s00204-020-02677-7
Anadón A, Martínez-Larrañaga MR, Martínez MA et al (2009) Toxicokinetics of glyphosate and its metabolite aminomethyl phosphonic acid in rats. Toxicol Lett 190:91–95. https://doi.org/10.1016/j.toxlet.2009.07.008
Asrican B, Lisman J, Otmakhov N (2007) Synaptic strength of individual spines correlates with bound Ca 2+-calmodulin-dependent kinase II. J Neurosci 27:14007–14011. https://doi.org/10.1523/JNEUROSCI.3587-07.2007
Aziz W, Kraev I, Mizuno K et al (2019) Multi-input synapses, but not LTP-strengthened synapses, correlate with hippocampal memory storage in aged mice. Curr Biol 29:3600-3610.e4. https://doi.org/10.1016/j.cub.2019.08.064
Baier CJ, Gallegos CE, Raisman-Vozari R, Minetti A (2017) Behavioral impairments following repeated intranasal glyphosate-based herbicide administration in mice. Neurotoxicol Teratol 64:63–72. https://doi.org/10.1016/j.ntt.2017.10.004
Bali YA, Kaikai N, Ba-M’hamed S, Bennis M (2019) Learning and memory impairments associated to acetylcholinesterase inhibition and oxidative stress following glyphosate based-herbicide exposure in mice. Toxicology 415:18–25. https://doi.org/10.1016/j.tox.2019.01.010
Barbosa ER, Leiros da Costa MD, Bacheschi LA et al (2001) Parkinsonism after glycine-derivate exposure. Mov Disord 16:565–568. https://doi.org/10.1002/mds.1105
Barnhart CD, Yang D, Lein PJ (2015) Using the Morris water maze to assess spatial learning and memory in weanling mice. PLoS ONE 10:e0124521. https://doi.org/10.1371/journal.pone.0124521
Benachour N, Moslemi S, Sipahutar H, Seralini GE (2007) Cytotoxic effects and aromatase inhibition by xenobiotic endocrine disrupters alone and in combination. Toxicol Appl Pharmacol 222:129–140. https://doi.org/10.1016/j.taap.2007.03.033
Bevins RA, Besheer J (2006) Object recognition in rats and mice: a one-trial non-matching-to-sample learning task to study “recognition memory.” Nat Protoc 1:1306–1311. https://doi.org/10.1038/nprot.2006.205
Bourne JN, Harris KM (2008) Balancing structure and function at hippocampal dendritic spines. Annu Rev Neurosci 31:47–67. https://doi.org/10.1146/annurev.neuro.31.060407.125646
Cattani D, Cesconetto PA, Tavares MK et al (2017) Developmental exposure to glyphosate-based herbicide and depressive-like behavior in adult offspring: implication of glutamate excitotoxicity and oxidative stress. Toxicology 387:67–80. https://doi.org/10.1016/j.tox.2017.06.001
Coullery RP, Ferrari ME, Rosso SB (2016) Neuronal development and axon growth are altered by glyphosate through a WNT non-canonical signaling pathway. Neurotoxicology 52:150–161. https://doi.org/10.1016/j.neuro.2015.12.004
Coullery R, Pacchioni AM, Rosso SB (2020) Exposure to glyphosate during pregnancy induces neurobehavioral alterations and downregulation of Wnt5a-CaMKII pathway. Reprod Toxicol 96:390–398. https://doi.org/10.1016/j.reprotox.2020.08.006
Davoren MJ, Schiestl RH (2018) Glyphosate-based herbicides and cancer risk: a post-IARC decision review of potential mechanisms, policy and avenues of research. Carcinogenesis 39:1207–1215. https://doi.org/10.1093/carcin/bgy105
Duke SO (2018) The history and current status of glyphosate. Pest Manag Sci 74:1027–1034. https://doi.org/10.1002/ps.4652
Eriguchi M, Iida K, Ikeda S et al (2019) Parkinsonism relating to intoxication with glyphosate. Intern Med 58:1935–1938. https://doi.org/10.2169/internalmedicine.2028-18
Ferreira TA, Blackman AV, Oyrer J et al (2014) Neuronal morphometry directly from bitmap images. Nat Methods 11:982–984. https://doi.org/10.1038/nmeth.3125
Fiala JC, Spacek J, Harris KM (2002) Dendritic spine pathology: cause or consequence of neurological disorders? Brain Res Rev 39:29–54. https://doi.org/10.1016/S0165-0173(02)00158-3
Gallegos CE, Bartos M, Bras C et al (2016) Exposure to a glyphosate-based herbicide during pregnancy and lactation induces neurobehavioral alterations in rat offspring. Neurotoxicology 53:20–28. https://doi.org/10.1016/j.neuro.2015.11.015
Gallegos CE, Baier CJ, Bartos M et al (2018) Perinatal glyphosate-based herbicide exposure in rats alters brain antioxidant status, glutamate and acetylcholine metabolism and affects recognition memory. Neurotox Res 34:363–374. https://doi.org/10.1007/s12640-018-9894-2
Gasnier C, Dumont C, Benachour N et al (2009) Glyphosate-based herbicides are toxic and endocrine disruptors in human cell lines. Toxicology 262:184–191. https://doi.org/10.1016/j.tox.2009.06.006
Guyton KZ, Loomis D, Grosse Y et al (2015) Carcinogenicity of tetrachlorvinphos, parathion, malathion, diazinon, and glyphosate. Lancet Oncol 16:490–491. https://doi.org/10.1016/S1470-2045(15)70134-8
Halbach VBU, O, Zacher C, Gass P, Unsicker K, (2006) Age-related alterations in hippocampal spines and deficiencies in spatial memory in mice. J Neurosci Res 83:525–531. https://doi.org/10.1002/jnr.20759
Harris KM, Kater SB (1994) Dendritic Spines: Cellular Specializations Imparting Both Stability and Flexibility to Synaptic Function. Annu Rev Neurosci 17:341–371. https://doi.org/10.1146/annurev.ne.17.030194.002013
Hedberg D, Wallin M (2010) Effects of Roundup and glyphosate formulations on intracellular transport, microtubules and actin filaments in Xenopus laevis melanophores. Toxicol Vitr 24:795–802. https://doi.org/10.1016/J.TIV.2009.12.020
Heyer DB, Meredith RM (2017) Environmental toxicology: sensitive periods of development and neurodevelopmental disorders. Neurotoxicology 58:23–41. https://doi.org/10.1016/j.neuro.2016.10.017
Jourdain P, Fukunaga K, Muller D (2003) Calcium/Calmodulin-Dependent Protein Kinase II Contributes to Activity-Dependent Filopodia Growth and Spine Formation. J Neurosci 23:10645–10649. https://doi.org/10.1523/jneurosci.23-33-10645.2003
Kim K, Hayashi Y (2014) CaMKII: the Swiss army knife of synaptic plasticity. J Physiol 592:4807–4808. https://doi.org/10.1113/jphysiol.2014.284414
Lajmanovich RC, Sandoval MT, Peltzer PM (2003) Induction of mortality and malformation in Scinax nasicus tadpoles exposed to glyphosate formulations. Bull Environ Contam Toxicol 70:612–618. https://doi.org/10.1007/s00128-003-0029-x
Laroche S, Davis S, Jay TM (2000) Plasticity at hippocampal to prefrontal cortex synapses: Dual roles in working memory and consolidation. Hippocampus 10:438–446. https://doi.org/10.1002/1098-1063(2000)10:4%3c438::AID-HIPO10%3e3.0.CO;2-3
Le Couteur DG, McLean AJ, Taylor MC et al (1999) Pesticides and Parkinson’s disease. Biomed Pharmacother 53:122–130. https://doi.org/10.1016/S0753-3322(99)80077-8
Mahmmoud RR, Sase S, Aher YD et al (2015) Spatial and working memory is linked to spine density and mushroom spines. PLoS ONE 10:e0139739. https://doi.org/10.1371/journal.pone.0139739
Marc J, Mulner-Lorillon O, Bellé R (2004) Glyphosate-based pesticides affect cell cycle regulation. Biol Cell 96:245–249. https://doi.org/10.1016/j.biolcel.2003.11.010
Martínez M-A, Ares I, Rodríguez J-L et al (2018) Neurotransmitter changes in rat brain regions following glyphosate exposure. Environ Res 161:212–219. https://doi.org/10.1016/j.envres.2017.10.051
Matsuzaki M, Honkura N, Ellis-Davies GCR, Kasai H (2004) Structural basis of long-term potentiation in single dendritic spines. Nature 429:761–766. https://doi.org/10.1038/nature02617
Meijering E, Jacob M, Sarria JCF et al (2004) Design and validation of a tool for neurite tracing and analysis in fluorescence microscopy images. Cytom Part A 58:167–176. https://doi.org/10.1002/cyto.a.20022
Menkes DB, Temple WA, Edwards IR (1991) Intentional self-poisoning with glyphosate-containing herbicides. Hum Exp Toxicol 10:103–107. https://doi.org/10.1177/096032719101000202
Neves G, Cooke SF, Bliss TVP (2008) Synaptic plasticity, memory and the hippocampus: a neural network approach to causality. Nat Rev Neurosci 9:65–75. https://doi.org/10.1038/nrn2303
Nishiyori Y, Nishida M, Shioda K et al (2014) Unilateral hippocampal infarction associated with an attempted suicide: a case report. J Med Case Rep 8:219. https://doi.org/10.1186/1752-1947-8-219
Okamoto KI, Narayanan R, Lee SH et al (2007) The role of CaMKII as an F-actin-bundling protein crucial for maintenance of dendritic spine structure. Proc Natl Acad Sci USA 104:6418–6423. https://doi.org/10.1073/pnas.0701656104
Paganelli A, Gnazzo V, Acosta H et al (2010) Glyphosate-based herbicides produce teratogenic effects on vertebrates by impairing retinoic acid signaling. Chem Res Toxicol 23:1586–1595. https://doi.org/10.1021/tx1001749
Patel S, Singh V, Kumar A et al (2006) Status of antioxidant defense system and expression of toxicant responsive genes in striatum of maneb- and paraquat-induced Parkinson’s disease phenotype in mouse: mechanism of neurodegeneration. Brain Res 1081:9–18. https://doi.org/10.1016/j.brainres.2006.01.060
Penzes P, Cahill ME, Jones KA et al (2011) Dendritic spine pathology in neuropsychiatric disorders : nature neuroscience : nature publishing group. Nat Neurosci 14:285–293. https://doi.org/10.1038/nn.2741.Dendritic
Planche V, Vergnet S, Auzou N et al (2019) Acute toxic limbic encephalopathy following glyphosate intoxication. Neurology 92:534–536. https://doi.org/10.1212/WNL.0000000000007115
Reger ML, Hovda DA, Giza CC (2009) Ontogeny of rat recognition memory measured by the novel object recognition task. Dev Psychobiol 51:672–678. https://doi.org/10.1002/dev.20402
Richardson JR, Roy A, Shalat SL et al (2014) Elevated serum pesticide levels and risk for Alzheimer disease. JAMA Neurol 71:284–290. https://doi.org/10.1001/jamaneurol.2013.6030
Robinson TE, Kolb B (2004) Structural plasticity associated with exposure to drugs of abuse. Neuropharmacology 47:33–46. https://doi.org/10.1016/j.neuropharm.2004.06.025
Romero AM, Renau-Piqueras J, Pilar Marin M et al (2013) Chronic alcohol alters dendritic spine development in neurons in primary culture. Neurotox Res 24:532–548. https://doi.org/10.1007/s12640-013-9409-0
Rosso S, Bollati F, Bisbal M et al (2004) LIMK1 regulates Golgi dynamics, traffic of Golgi-derived vesicles, and process extension in primary cultured neurons. Mol Biol Cell 15:3433–3449. https://doi.org/10.1091/mbc.E03-05-0328
Santos HR, Cintra WM, Aracava Y et al (2004) Spine density and dendritic branching pattern of hippocampal CA1 pyramidal neurons in neonatal rats chronically exposed to the organophosphate paraoxon. Neurotoxicology 25:481–494. https://doi.org/10.1016/j.neuro.2004.01.007
Sato C, Kamijo Y, Yoshimura K, Ide T (2011) Aseptic meningitis in association with glyphosate-surfactant herbicide poisoning. Clin Toxicol 49:118–120. https://doi.org/10.3109/15563650.2011.552065
Semple BD, Blomgren K, Gimlin K et al (2013) Brain development in rodents and humans: Identifying benchmarks of maturation and vulnerability to injury across species. Prog Neurobiol 106–107:1–16. https://doi.org/10.1016/j.pneurobio.2013.04.001
Stamatakou E, Marzo A, Gibb A, Salinas PC (2013) Activity-Dependent Spine Morphogenesis: A Role for the Actin-Capping Protein Eps8. J Neurosci 33:2661–2670. https://doi.org/10.1523/JNEUROSCI.0998-12.2013
Vorhees CV, Williams MT (2006) Morris water maze: Procedures for assessing spatial and related forms of learning and memory. Nat Protoc 1:848–858. https://doi.org/10.1038/nprot.2006.116
Wang XF, Li S, Chou AP, Bronstein JM (2006) Inhibitory effects of pesticides on proteasome activity: Implication in Parkinson’s disease. Neurobiol Dis 23:198–205. https://doi.org/10.1016/j.nbd.2006.02.012
Wang G, Fan XN, Tan YY et al (2011) Parkinsonism after chronic occupational exposure to glyphosate. Park Relat Disord 17:486–487
Williams GM, Kroes R, Munro IC (2000) Safety evaluation and risk assessment of the herbicide Roundup and its active ingredient, glyphosate, for humans. Regul Toxicol Pharmacol 31:117–165. https://doi.org/10.1006/rtph.1999.1371
Williams AL, Watson RE, DeSesso JM (2012) Developmental and reproductive outcomes in humans and animals after glyphosate exposure: a critical analysis. J Toxicol Environ Heal Part B 15:39–96. https://doi.org/10.1080/10937404.2012.632361
Zhao ZH, Zheng G, Wang T et al (2018) Low-level gestational lead exposure alters dendritic spine plasticity in the hippocampus and reduces learning and memory in rats. Sci Rep. https://doi.org/10.1038/s41598-018-21521-8
Acknowledgements
This work was supported by grants from the Agencia de Promoción Científica y Tecnológica (PICT 2014-1326, to SBR), Consejo Nacional de Investigaciones Científicas y Tecnológicas (PIP 0947, to SBR), and Universidad Nacional de Rosario (UNR BIO 382, to SBR), Argentina.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Luna, S., Neila, L.P., Vena, R. et al. Glyphosate exposure induces synaptic impairment in hippocampal neurons and cognitive deficits in developing rats. Arch Toxicol 95, 2137–2150 (2021). https://doi.org/10.1007/s00204-021-03046-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-021-03046-8