Abstract
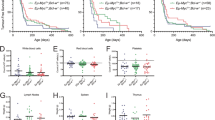
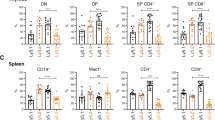
Signals that control the fine balance between cell death and cell survival are altered during tumorigenesis. Understanding the mechanisms by which this balance is perturbed, leading to excessive cell survival, is important for designing effective therapies. Proteins belonging to the B-cell lymphoma (BCL) family are known to regulate death responses to apoptotic signals, especially those originating within cells. A subset of BCL family members capable of inhibiting cell death is known to contribute to tumorigenesis; however, it is not known whether all six antiapoptotic BCL family members play a causal role in tumor development. Using a mouse model of MYC-driven leukemia, we showed that, in addition to the well characterized BCL2 and BCLxl (BCL2L1), the other four family members—BCLw (BCL2L2), BCLb (BCL2L10), BFL1 (BCL2A1) and MCL1—also cooperate with MYC to accelerate leukemogenesis. In addition, high levels of each family member are found in either solid human tumors or cell lines derived from human leukemias or lymphomas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chen L, Willis SN, Wei A, Smith BJ, Fletcher JI, Hinds MG et al. (2005). Differential targeting of prosurvival Bcl-2 proteins by their BH3-only ligands allows complementary apoptotic function. Mol Cell 17: 393–403.
Chen S, Dai Y, Harada H, Dent P, Grant S . (2007). Mcl-1 down-regulation potentiates ABT-737 lethality by cooperatively inducing Bak activation and Bax translocation. Cancer Res 67: 782–791.
Cory S, Huang DC, Adams JM . (2003). The Bcl-2 family: roles in cell survival and oncogenesis. Oncogene 22: 8590–8607.
Cosulich SC, Worrall V, Hedge PJ, Green S, Clarke PR . (1997). Regulation of apoptosis by BH3 domains in a cell-free system. Curr Biol 7: 913–920.
Hemann MT, Bric A, Teruya-Feldstein J, Herbst A, Nilsson JA, Cordon-Cardo C et al. (2005). Evasion of the p53 tumour surveillance network by tumour-derived MYC mutants. Nature 436: 807–811.
Holinger EP, Chittenden T, Lutz RJ . (1999). Bak BH3 peptides antagonize Bcl-xL function and induce apoptosis through cytochrome c-independent activation of caspases. J Biol Chem 274: 13298–13304.
Horvitz HR . (1999). Genetic control of programmed cell death in the nematode Caenorhabditis elegans. Cancer Res 59: 1701s–1706s.
Kawasaki T, Yokoi S, Tsuda H, Izumi H, Kozaki K, Aida S et al. (2007). BCL2L2 is a probable target for novel 14q11.2 amplification detected in a non-small cell lung cancer cell line. Cancer Sci 98: 1070–1077.
Kohl TM, Hellinger C, Ahmed F, Buske C, Hiddemann W, Bohlander SK et al. (2007). BH3 mimetic ABT-737 neutralizes resistance to FLT3 inhibitor treatment mediated by FLT3-independent expression of BCL2 in primary AML blasts. Leukemia 21: 1763–1772.
Konopleva M, Contractor R, Tsao T, Samudio I, Ruvolo PP, Kitada S et al. (2006). Mechanisms of apoptosis sensitivity and resistance to the BH3 mimetic ABT-737 in acute myeloid leukemia. Cancer Cell 10: 375–388.
Lanave C, Santamaria M, Saccone C . (2004). Comparative genomics: the evolutionary history of the Bcl-2 family. Gene 333: 71–79.
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H et al. (2007). Clustal W and Clustal X version 2.0. Bioinformatics 23: 2947–2948.
Letai A, Sorcinelli MD, Beard C, Korsmeyer SJ . (2004). Antiapoptotic BCL-2 is required for maintenance of a model leukemia. Cancer Cell 6: 241–249.
Luo H, Li Q, O’Neal J, Kreisel F, Le Beau MM, Tomasson MH . (2005). c-Myc rapidly induces acute myeloid leukemia in mice without evidence of lymphoma-associated antiapoptotic mutations. Blood 106: 2452–2461.
Mandal M, Borowski C, Palomero T, Ferrando AA, Oberdoerffer P, Meng F et al. (2005). The BCL2A1 gene as a pre-T cell receptor-induced regulator of thymocyte survival. J Exp Med 201: 603–614.
Reed JC . (2006). Drug insight: cancer therapy strategies based on restoration of endogenous cell death mechanisms. Nat clin pract Oncol 3: 388–398.
Riedl SJ, Salvesen GS . (2007). The apoptosome: signalling platform of cell death. Nat Rev Mol Cell Biol 8: 405–413.
Schultz J, Milpetz F, Bork P, Ponting CP . (1998). SMART, a simple modular architecture research tool: identification of signaling domains. Proc Natl Acad Sci USA 95: 5857–5864.
Shoemaker AR, Mitten MJ, Adickes J, Ackler S, Refici M, Ferguson D et al. (2008). Activity of the Bcl-2 family inhibitor ABT-263 in a panel of small cell lung cancer xenograft models. Clin Cancer Res 14: 3268–3277.
Swanson PJ, Kuslak SL, Fang W, Tze L, Gaffney P, Selby S et al. (2004). Fatal acute lymphoblastic leukemia in mice transgenic for B cell-restricted bcl-xL and c-myc. J Immunol 172: 6684–6691.
Valdez BC, Murray D, Ramdas L, de Lima M, Jones R, Kornblau S et al. (2008). Altered gene expression in busulfan-resistant human myeloid leukemia. Leuk Res 32: 1684–1697.
Vaux DL, Cory S, Adams JM . (1988). Bcl-2 gene promotes haemopoietic cell survival and cooperates with c-myc to immortalize pre-B cells. Nature 335: 440–442.
Wesarg E, Hoffarth S, Wiewrodt R, Kröll M, Biesterfeld S, Huber C et al. (2007). Targeting BCL-2 family proteins to overcome drug resistance in non-small cell lung cancer. Int J Cancer 121: 2387–2394.
Yasui K, Mihara S, Zhao C, Okamoto H, Saito-Ohara F, Tomida A et al. (2004). Alteration in copy numbers of genes as a mechanism for acquired drug resistance. Cancer Res 64: 1403–1410.
Youle RJ, Strasser A . (2008). The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol 9: 47–59.
Zhai D, Jin C, Satterthwait AC, Reed JC . (2006). Comparison of chemical inhibitors of antiapoptotic Bcl-2-family proteins. Cell Death Differ 13: 1419–1421.
Acknowledgements
We thank members of the Varmus laboratory, especially G Sanchez, A Giannakou and MA Melnick, for expert handling of the mouse colony, including genotyping, and for performing immunohistochemistry on embedded tissue sections. Funding for this project was provided, in part, by NIH P01 2PO1CA094060-06A1 to HEV.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Beverly, L., Varmus, H. MYC-induced myeloid leukemogenesis is accelerated by all six members of the antiapoptotic BCL family. Oncogene 28, 1274–1279 (2009). https://doi.org/10.1038/onc.2008.466
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.466
Keywords
This article is cited by
BCL-W makes only minor contributions to MYC-driven lymphoma development
Oncogene (2023)
MYC: a multipurpose oncogene with prognostic and therapeutic implications in blood malignancies
Journal of Hematology & Oncology (2021)
Increased level of long non coding RNA H19 is correlated with the downregulation of miR-326 and BCL-2 genes in pediatric acute lymphoblastic leukemia, a possible hallmark for leukemogenesis
Molecular Biology Reports (2021)
BCL-w: apoptotic and non-apoptotic role in health and disease
Cell Death & Disease (2020)
MYC selects against reduced BCL2A1/A1 protein expression during B cell lymphomagenesis
Oncogene (2017)