Abstract
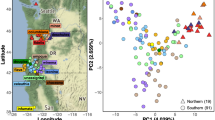
We evaluated the genetic diversity of a snake species with color polymorphism to understand the evolutionary processes that drive genetic structure across a large geographic region. Specifically, we analyzed genetic structure of the highly polymorphic ground snake, Sonora semiannulata, (1) among populations, (2) among color morphs (3) at regional and local spatial scales, using an amplified fragment length polymorphism dataset and multiple population genetic analyses, including FST-based and clustering analytical techniques. Based upon these methods, we found that there was moderate to low genetic structure among populations. However, this diversity was not associated with geographic locality at either spatial scale. Similarly, we found no evidence for genetic divergence among color morphs at either spatial scale. These results suggest that despite dramatic color polymorphism, this phenotypic diversity is not a major driver of genetic diversity within or among populations of ground snakes. We suggest that there are two mechanisms that could explain existing genetic diversity in ground snakes: recent range expansion from a genetically diverse founder population and current or recent gene flow among populations. Our findings have further implications for the types of color polymorphism that may generate genetic diversity in snakes.




Similar content being viewed by others
References
Antao T, Beaumont MA (2011) Mcheza: a workbench to detect selection using dominant markers. Bioinformatics 27:1717–1718
Avise JC, Alisauskas RT, Nelson WS, Ankney CD (1992) Matriarchal population genetic structure in an avian species with female natal philopatry. Evolution 46:1084–1096
Balkenhol N, Waits LP, Dezzani RJ (2009) Statistical approaches in landscape genetics: an evaluation of methods for linking landscape and genetic data. Ecography 32:818–830
Beaumont MA, Balding DJ (2004) Identifying adaptive genetic divergence among populations from genome scans. Mol Ecol 13:969–980
Bohonak AJ (1999) Dispersal, gene flow, and population structure. Q Rev Biol 74:21–45
Bonnet E, van de Peer Y (2002) zt: a software tool for simple and partial Mantel tests. J Stat Softw 7:1–12
Bouzat JL, Johnson K (2004) Genetic structure among closely spaced leks in a peripheral population of lesser prairie chickens. Mol Ecol 13:499–505
Brodie ED III, Brodie ED Jr (2004) Venomous snake mimicry. In: Campbell JA, Lamar WW (eds) The venomous reptiles of the Western hemisphere, vol II. Comstock Publishing Associates, Ithaca p vii + 870 pp
Brodie ED Jr, Ridenhour BJ, Brodie ED III (2002) The evolutionary response of predators to dangerous prey: hotspots and coldspots in the geographic mosaic of coevolution between garter snakes and newts. Evolution 56:2067–2082
Brown JH, Nicoletto PF (1991) Spatial scaling of species composition: body masses of North American land mammals. Am Nat 138(6):1478–1512
Castoe TA, Spencer CL, Parkinson CL (2007) Phylogeographic structure and historical demography of the western diamondback rattlesnake (Crotalus atrox): a perspective on North American desert biogeography. Mol Phylogenet Evol 42:193–212
Cavender-Bares J, Keen A, Miles B (2006) Phylogenetic structure of Floridian plant communities depends on taxonomic and spatial scale. Ecology 87:S109–S122
Chamberlain NL, Hill RI, Kapan DD, Gilbert LE, Kronforst MR (2009) Polymorphic butterfly reveals the missing link in ecological speciation. Science 326:847–850
Corl A, Davis AR, Kuchta SR, Sinervo B (2010) Selective loss of polymorphic mating types is associated with rapid phenotypic evolution during morphic speciation. Proc Natl Acad Sci USA 107:4254–4259
Cox CL, Davis Rabosky ARD (2013) Spatial and temporal drivers of phenotypic diversity in polymorphic snakes. Am Nat 182:E40–E57
Cox CL, Boback SM, Guyer C (2011) Spatial dynamics of body size frequency distributions for North American squamates. Evol Biol 38:453–464
Cox CL, Rabosky ARD, Reyes-Velasco J, Ponce-Campos P, Smith EN, Flores-Villela O, Campbell JA (2012a) Molecular systematics of the genus Sonora (Squamata: Colubridae) in central and western Mexico. Syst Biodivers 10:93–108
Cox CL, Streicher JW, Sheehy CM, Campbell JA, Chippindale PT (2012b) Patterns of genetic differentiation among populations of Smilisca fodiens. Herpetologica 68:226–235
Cox CL, Rabosky ARD, Chippindale PT (2013) Sequence variation in the Mc1R gene for a group of polymorphic snakes. Gene 513:282–286
Degenhardt WG, Painter CW, Price AH (1996) Amphibians and reptiles of New Mexico, vol xiv + 431. University of New Mexico Press, New Mexico
Ehrich D (2006) AFLPdat: a collection of R functions for convenient handling of AFLP data. Mol Ecol Notes 6:603–604
Elmer KR, Lehtonen TK, Meyer A (2009) Color assortative mating contributes to sympatric divergence of neotropical cichlid fish. Evolution 63:2750–2757
Epps CW, Pallsbøll PJ, Wehausen JD, Roderick GK, Ramey RR, McCullough DR (2005) Highways block gene flow and cause a rapid decline in genetic diversity of bighorn sheep. Ecol Lett 8:1029–1038
Ernst CH, Ernst EM (2003) Snakes of the United States and Canada. Smithsonian Books, Washington
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction sites. Genetics 131:479–491
Excoffier L, Foll M, Petit R (2009) Genetic consequences of range expansions. Annu Rev Ecol Evol Syst 40:481–501
Falush D, Stephens M, Pritchard JK (2003) Inference of population structure using multilocus genotype data: linked loci and correlated gene frequencies. Genetics 164:1567–1587
Foll M, Gaggiotti O (2008) A genome-scan method to identify selected loci appropriate for both dominant and codominant markers: a Bayesian perspective. Genetics 180:977–993
Fontenot BE, Makowsky R, Chippindale PT (2011) Nuclear-mitochondrial discordance and gene flow in a recent radiation of toads. Mol Phylogenet Evol 59:66–80
Forsman A (1995) Opposing fitness consequences of colour pattern in male and female snakes. J Evol Biol 8:53–70
Frankham R (1997) Do island populations have less genetic variation than mainland populations? Heredity 78:311–327
Goosens B, Chikhi L, Ancrenaz M, Lackman-Ancrenaz I, Andau P, Bruford MW (2006) Genetic signature of anthropogenic population collapse in orang-utans. PLoS Biol 4:e25
Gray SM, McKinnon JS (2006) Linking color polymorphism maintenance and speciation. Trends Ecol Evol 22(2):71–79
Hamilton CA, Formanowicz DR, Bond JE (2011) Species delimitation and phylogeography of Aphonopelma hentzi (Araneae, Mygalomorphae, Theraphosidae): cryptic diversity in North American tarantulas. PLoS One 6:e26207
Hartl DL, Clark AG (2007) Principles of population genetics, 4th edn. Sinauer Associates, Sunderland
Huff DR, Peakall R, Smouse PE (1993) RAPD variation within and among natural populations of outcrossing buffalograss Buchloe dactyloides (Nutt) Engelm. Theor Appl Genet 86:927–934
Hugall AF, Stuart-Fox D (2012) Accelerated speciation in colour polymorphic birds. Nature 485:631–634
Hughes AR, Inouye BD, Johnson MTJ, Underwood N, Vellend M (2008) Ecological consequences of genetic diversity. Ecol Lett 11:609–623
Ibrahim KI, Nichols RA, Hewitt GM (1996) Spatial patterns of genetic variation generated by different forms of dispersal during range expansion. Heredity 77:282–291
Jiggins CD, Naisbit RE, Coe RL, Mallet J (2001) Reproductive isolation caused by colour pattern mimicry. Nature 411:302–305
Johnson JA (2008) Recent range expansion and divergence among North American prairie grouse. J Hered 99:165–173
Johnson JA, Toepfer JE, Dunn PO (2003) Contrasting patterns of mitochondrial and microsatellite population structure in fragmented populations of greater prairie chickens. Mol Ecol 12:3335–3347
Jombart T (2008) adagenet: a R package for the multivariate analysis of genetic markers. Bioinformatics 24:1403–1405
Jombart T, Devillard S, Balloux F (2010) Discriminant analysis of principal components: a new method for the analysis of genetically structured populations. BMC Genet 11:94
Kark S, Warburg I, Werner YL (1997) Polymorphism in the snake Psammophis schokari on both sides of the desert edge. J Arid Environ 37:513–527
King RB (1987) Color pattern polymorphism in the Lake Erie water snake, Nerodia sipedon insularum. Evolution 41:241–255
King RB (1988) Polymorphic populations of the garter snake Thamnophis sirtalis near Lake Erie. Herpetologica 44:451–458
Lande R (1980) Genetic variation and phenotypic evolution during allopatric speciation. Am Nat 116:463–479
Legendre P, Fortin M-J (2010) Comparison of the Mantel test and alternative approaches for detecting complex multivariate relationships in the spatial analysis of genetic data. Mol Ecol Res 10:831–844
Lenormand T (2002) Gene flow and the limits to natural selection. Trends Ecol Evol 17:183–189
Lewontin RC (1972) The apportionment of human diversity. Evol Biol 6:381–398
Lyrholm T, Leimar O, Johanneson B, Gyllensten U (1999) Sex-biased dispersal in sperm whales: contrasting mitochondrial and nuclear genetic structure of global populations. Proc R Soc B 266:347–354
Maguire TL, Peakall R, Saenger P (2002) Comparative analysis of genetic diversity in the mangrove species Avicennia marina (Forsk.) Vierh. (Avicenniaceae) detected by AFLPs and SSRs. Theor Appl Genet 104:388–398
Manel S, Schwartz MK, Luikart G, Taberlet P (2003) Landscape genetics: combining landscape ecology and population genetics. Trends Ecol Evol 18:189–197
McMillan WO, Weigt LA, Palumbi SR (1999) Color pattern evolution, assortative mating, and genetic differentiation in brightly colored butterflyfishes (Chaetodontidae). Evolution 53:247–260
Miller AL, Makowsky RA, Formanowicz DR, Prendini L, Cox CL (2014) Cryptic genetic diversity and complex phylogeography of the boral North American scorpion, Paruroctonus boreus (Vaejovidae). Mol Phylogenet Evol 71:298–307
Nei M (1972) Genetic distance between populations. Am Nat 106:283–392
Nei M (1978) Estimation of average heterozygosity and genetic distance form a small number of individuals. Genetics 89:583–590
Neiswenter SA, Riddle BR (2010) Diversification of the Perognathus plavus species in emerging arid grasslands of western North America. J Mammal 9:348–362
Nosil P (2008) Speciation with gene flow could be common. Mol Ecol 17:2103–2106
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295
Peakall R, Smouse PE, Huff DR (1995) Evolutionary implication of allozyme and RAPD variation in diploid populations of dioecious buffalograss Buchloe dactyloides. Mol Ecol 4:135–147
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155(2):945–959
Pryke S, Griffith SC (2006) Red dominates black: agonistic signalling among head morphs in the colour polymorphic Gouldian finches. Proc R Soc B 273:949–957
Puebla O, Bermingham E, Guichard F, Whiteman E (2007) Colour pattern as a single trait driving speciation in Hypoplectrus coral reef fishes? Proc R Soc B 274:1265–1271
Pyron RA, Burbrink FT (2009) Lineage diversification in a widespread species: roles for niche divergence and conservatism in the common kingsnake. Mol Ecol 18:3443–3457
R Development Core Team (2008) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Reed DH, Frankham R (2003) Correlation between fitness and genetic diversity. Conserv Biol 17:230–237
Reynolds RG, Fitzpatrick BM (2007) Assortative mating in poison-dart frogs based on an ecologically important trait. Evolution 61:2253–2259
Runemark A, Hansson B, Pafilis P, Valakos ED, Svensson EI (2010) Island biology and morphological divergence of the Skyros wall lizard Podarcis gaigeae: a combined role for local selection and genetic drift on color morph frequency divergence? BMC Evol Biol 10:269
Savage JM, Slowinski JB (1992) The colouration of venomous coral snakes (family Elapidae) and their mimics (families Aniliidae and Colubridae). Biol J Linn Soc 45:235–254
Seehausen O, van Alphen JJM, Lande R (1999) Color polymorphism and sex ratio distortion in a cichlid fish as an incipient stage in sympatric speciation by sexual selection. Ecol Lett 2:367–378
Seehausen O, Terai Y, Magalhaes IS, Carleton KL, Mrosso HDJ, Miyagi R, van der Sluijs I, Schneider MV, Maan ME, Tachida H, Imai H, Okada N (2008) Speciation through sensory drive in cichlid fish. Nature 455:620–626
Shannon CE (1948) A mathematic theory of communication. Bell Syst Tech J 27:379–423
Shine R, Madsen T (1994) Sexual dichromatism in snakes of the genus Vipera: a review and a new evolutionary hypothesis. J Herpetol 28:114–117
Shine R, Ambariyanto, Harlow PS, Mumpumi (1998) Ecological divergence among sympatric colour morphs in blood pythons. Oecologia 116:113–119
Sillman AJ, Carver JK, Loew ER (1999) The photoreceptors and visual pigments in the retina of a boid snake, the ball python (Python regius). J Exp Biol 202:1931–1938
Sinervo B, Svensson E (2002) Correlational selection and the evolution of genomic architecture. Heredity 89:329–338
Sinervo B, Heulin B, Surget-Groba Y, Clobert J, Miles DB, Corl A, Chaine A, Davis A (2007) Models of density-dependent genic selection and a new rock-paper-scissors social system. Am Nat 170:663–680
Slatkin M (1985) Gene flow in natural populations. Annu Rev Ecol Syst 16:393–430
Slatkin M (1987) Gene flow and the geographic structure of natural populations. Science 236:787–792
Smith JM (1962) Disruptive selection, polymorphism and sympatric speciation. Nature 195:50–62
Streicher JW, Cox CL, Campbell JA, Smith EN, de Sa RO (2012) Rapid range expansion in the Great Plains narrow-mouthed toad (Gastrophryne olivacea) and a revised taxonomy for North American microhylids. Mol Phylogenet Evol 64:645–653
Tanaka K (2007) Thermal biology of a colour dimorphic snake, Elaphe quadrivirgata, in a montane forest: do melanistic snakes enjoy thermal advantages? Biol J Linn Soc 92:309–322
Taulman JF, Robbins LW (1996) Recent range expansion and distributional limits of the nine-banded armadillo (Dasypus novemcinctus) in the United States. J Biogeogr 23:635–648
Tenant A (1984) The snakes of Texas. Texas Monthly Press, Texas
Vos P, Hogers R, Bleeker M, Reijans M, Lee TVD, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M (1995) AFLP: a new technique for DNA fingerprinting. Nucleic Acids Res 23(21):4407–4414
Walls GL (1942) The vertebrate eye and its adaptive radiation. Cranbrook Press, Bloomfield Hills
Wang IJ, Summers K (2010) Genetic structure is correlated with phenotypic divergence rather than geographic isolation in the highly polymorphic strawberry poison-dart frog. Mol Ecol 19:447–458
West-Eberhard MJ (1986) Alternative adaptations, speciation, and phylogeny. Proc Natl Acad Sci USA 83:1388–1392
Whitlock R, Hipperson H, Mannerelli H, Butlin RK, Burke T (2008) An objective, rapid and reproducible method for scoring AFLP peak-height data that minimizes genotyping error. Mol Ecol Res 8:725–735
Acknowledgments
We extend our sincere gratitude to A. R. Davis Rabosky for her contributions to fieldwork, study design and the writing of this manuscript. We thank the University of Texas at Arlington Herpetology graduate students, the Texas Herpetological Society (THS), and the Kansas Herpetological Society (KHS) for tissue donation, specimen collection, and logistical assistance. In particular, we are grateful to C. E. Roelke and J. F. Stringer for their logistical assistance in the field and the laboratory. We especially thank the staff and curators at the following museum collections for access to tissues: Museum of Vertebrate Zoology at the University of California, Texas Natural History Collection at the University of Texas, University of Texas—El Paso, the Amphibian and Reptile Diversity Research Center at the University of Texas—Arlington, Sam Noble Museum at the University of Oklahoma, and the Sternberg Museum of Natural History at Fort Hayes State University. We also thank B. E. Fontenot, C. E. Roelke, M. Sandel, J. W. Streicher, C. M. Watson, and the R.M. Cox and E. D. Brodie III labs at the University of Virginia for helpful criticism during this project and preparation of the manuscript. We thank the wildlife departments from the states of Kansas, Oklahoma, Texas, Colorado, and New Mexico for necessary scientific collecting permits. This project was funded by the Theodore Roosevelt Memorial Fund from the American Museum of Natural History to C.L. Cox, as well as a Systematics Research Fund Award from The Linnean Society and an NSF Postdoctoral Fellowship in Biology (DBI-0906046) to A.R. Davis (Rabosky).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cox, C.L., Chippindale, P.T. Patterns of genetic diversity in the polymorphic ground snake (Sonora semiannulata). Genetica 142, 361–370 (2014). https://doi.org/10.1007/s10709-014-9780-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10709-014-9780-7