Abstract
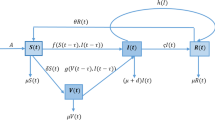
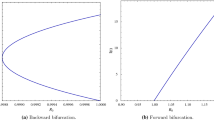
In recent times, several infectious diseases that breakthrough contagiousness in vaccinated people and reinfections in primarily infected counterparts have become more common. In most cases, reinfection occurs as a result of viral mutations. The mutated pathogens may possess alternate modes of transmission, and even the exposure period may vary in some cases. Hence, to gain a deeper understanding of disease transmission dynamics, it becomes eminent to incorporate a dedicated compartment for reinfection within a mathematical model. To account for these vital factors, this research presents an epidemic model that includes a specific compartment for individuals experiencing reinfection. The model also incorporates two separate time delay parameters that account for the incubation periods for the initial infection and the subsequent reinfection. The study reveals that the system exhibits forward bifurcation with respect to the parameter \(\beta _1\) in the absence of time delays. However, in the presence of time delay, Hopf bifurcation is identified when the delays exceed corresponding cut-off values. The research further highlights that an extension in the exposure period leads to a rise in the reinfected population, thereby contributing to the persistence of the disease over an extended timeframe. To demonstrate the practical applicability of the proposed model, we have fitted our model with the data of real-time vaccination cases for COVID-19 in India. Notably, the constructed model can be readily adapted for other infectious diseases by selecting appropriate parameter values based on pertinent real-time data. The theoretical results are validated via numerical simulation.








Similar content being viewed by others
Data availability statement
The data that support the findings of this study are openly available from the website https://github.com/owid/covid-19-data/blob/master/public/data/vaccinations/country_data/India.csv#L1.
References
T.Q. Tang, Z. Shah, R. Jan, E. Alzahrani, Modeling the dynamics of tumor-immune cells interactions via fractional calculus. Eur. Phys. J. Plus 137(3), 367 (2022)
R. Jan, Z. Shah, W. Deebani, E. Alzahrani, Analysis and dynamical behavior of a novel dengue model via fractional calculus. Int. J. Biomath. 15(6), 2250036 (2022)
A. Jan, R. Jan, H. Khan, M.S. Zobaer et al., Fractional-order dynamics of Rift Valley fever in ruminant host with vaccination. Commun. Math. Biol. Neurosci. 2020(79), 2052 (2020)
S. Gao, P. Binod, C.W. Chukwu, T. Kwofie et al., A mathematical model to assess the impact of testing and isolation compliance on the transmission of COVID-19. Infect. Dis. Model. 8(2), 427–444 (2023)
S. Gargouri, A. Souissi, N. Abid, A. Chtourou et al., Evidence of sars-cov-2 symptomatic reinfection in four healthcare professionals from the same hospital despite the presence of antibodies. Int. J. Infect. Dis. 117, 146–154 (2022)
T.P. Gujarati, G. Ambika, Virus antibody dynamics in primary and secondary dengue infections. J. Math. Biol. 69, 1773–1800 (2014)
G. Katriel, Epidemics with partial immunity to reinfection. Math. Biosci. 228(2), 153–159 (2010)
R. Puthiyedath, S. Kataria, U. Payyappallimana et al., Ayurvedic clinical profile of covid-19-a preliminary report. J. Ayurv. Integr. Med. 13(1), 100326 (2022)
P. Tamilalagan, B. Krithika, P. Manivannan, S. karthiga, Is reinfection negligible effect in COVID-19? A mathematical study on the effects of reinfection in COVID-19. Math. Methods Appl. Sci. (2023). https://doi.org/10.1002/mma.9614
M. Cerón Gómez, H.M. Yang, A simple mathematical model to describe antibody-dependent enhancement in heterologous secondary infection in dengue. Math. Med. Biol. J. IMA. 36(4), 411–438 (2019)
F. Camargo, M. Adimy, L. Esteva et al., Modeling the relationship between antibody-dependent enhancement and disease severity in secondary dengue infection. Bull. Math. Biol. 83(8), 85 (2021)
A. Danchin, O. Pagani-Azizi, G. Turinici, G. Yahiaoui, Covid-19 adaptive humoral immunity models: weakly neutralizing versus antibody-disease enhancement scenarios. Acta. Biotheor. 70(4), 23 (2022)
H. Song, Z. Yuan, S. Liu, Z. Jin, G. Sun, Mathematical modeling the dynamics of sars-cov-2 infection with antibody-dependent enhancement. Nonlinear Dyn. 111(3), 2943–2958 (2023)
P. Tamilalagan, S. Karthiga, P. Manivannan, Dynamics of fractional order hiv infection model with antibody and cytotoxic t-lymphocyte immune responses. J. Comput. Appl. Math. 382, 113064 (2021)
P. Balasubramaniam, M. Prakash, P. Tamilalagan, Stability and hopf bifurcation analysis of immune response delayed hiv type 1 infection model with two target cells. Math. Methods Appl. Sci. 38(17), 3653–3669 (2015)
H.W. Hethcote, The mathematics of infectious diseases. SIAM Rev. 42, 599–653 (2000)
S. Pathak, A. Maiti, G.P. Samanta, Rich dynamics of an sir epidemic model. Nonlinear Anal. Model. Control. 15(1), 71–81 (2010)
S. Sharma, G.P. Samanta, Stability analysis and optimal control of an epidemic model with vaccination. Int. J. Biomath. 8(03), 1550030 (2015)
H. Sasidharakurup, G. Kumar, B. Nair, S. Diwakar, Mathematical modeling of severe acute respiratory syndrome coronavirus 2 infection network with cytokine storm, oxidative stress, thrombosis, insulin resistance, and nitric oxide pathways. OMICS 25(12), 770–781 (2021)
M.S. Narasima, D. John, S.P. Anbuudayasankar, G.R. Jammy, R. Pant, L. Choudhury, Shivir-an agent-based model to assess the transmission of covid-19 in India. medRxiv (2022). https://doi.org/10.1101/2022.05.26.22275624
S. Banerjee, Dynamics of the COVID-19 pandemic: nonlinear approaches on the modelling, prediction and control. Eur. Phys. J. Spec. Top. 231(18–20), 3275–3280 (2022)
F. Karim, S. Chauhan, J. Dhar, Analysing an epidemic-economic model in the presence of novel corona virus infection: capital stabilization, media effect, and the role of vaccine. Eur. Phys. J. Spec. Top. 231(8–20), 3545–3562 (2022)
M.B. Devi, A. Devi, P.K. Gupta et al., Response of vaccination on community transmission of COVID-19: a dynamical approach. Eur. Phys. J. Spec. Top. 231(18), 3749–3765 (2022)
O.J. Watson, G. Barnsley, J. Toor et al., Global impact of the first year of COVID-19 vaccination: a mathematical modelling study. Lancet. Infect. Dis 22(9), 1293–1302 (2022)
S. Alyobi, R. Jan, Qualitative and quantitative analysis of fractional dynamics of infectious diseases with control measures. Fractal Fract. 7(5), 400 (2023)
A. Jan, H.M. Srivastava, A. Khan, P.O. Mohammed et al., In vivo HIV dynamics, modeling the interaction of HIV and immune system via non-integer derivatives. Fractal Fract. 7(5), 361 (2023)
A. Alshehri, Z. Shah, R. Jan, Mathematical study of the dynamics of lymphatic filariasis infection via fractional-calculus. Eur. Phys. J. Plus. 138(3), 1–15 (2023)
R. Jan, S. Qureshi, S. Boulaaras et al., Optimization of the fractional-order parameter with the error analysis for human immunodeficiency virus under Caputo operator. Discrete Contin. Dyn. Syst.-S. 16(8), 2118–2140 (2023)
S. Boulaaras, Z.U. Rehman, F.A. Abdullah et al., Coronavirus dynamics, infections and preventive interventions using fractional-calculus analysis. AIMS Math. 8(4), 8680–8701 (2023)
M.G.M. Gomes, L.J. White, G.F. Medley, Infection, reinfection, and vaccination under suboptimal immune protection: epidemiological perspectives. J. Theor. Biol. 228(4), 539–549 (2004)
N. Stollenwerk, J. Martins, A. Pinto, The phase transition lines in pair approximation for the basic reinfection model siri. Phys. Lett. A 371, 379–388 (2007)
P. Rodrigues, A. Margheri, C. Rebelo, M.G.M. Gomes, Heterogeneity in susceptibility to infection can explain high reinfection rates. J. Theor. Biol. 259(2), 280–290 (2009)
J. Gómez-Gardeñes, A.S. Barros, S.T. Pinho, R.F. Andrade, Abrupt transitions from reinfections in social contagions. Europhys. Lett. 110, 58006 (2015)
R. Pagliara, B. Dey, N.E. Leonard, Bistability and resurgent epidemics in reinfection models. IEEE Control Syst. Lett. 2(2), 290–295 (2018)
S. Zhai, M. Du, Y. Wang et al., Effects of heterogeneous susceptibility on epidemiological models of reinfection. Nonlinear Dyn. 111, 1890–1902 (2023)
R. Jan, S. Boulaaras, S. Alyobi et al., Transmission dynamics of Hand-Foot-Mouth Disease with partial immunity through non-integer derivative. Int. J. Biomath. 16(06), 2250115 (2023)
D.M. Morens, J.K. Taubenberger, A.S. Fauci, Rethinking next-generation vaccines for coronaviruses, influenzaviruses, and other respiratory viruses. Cell Host Microbe. 31(1), 146–157 (2023)
P. Driessche, J. Watmough, Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math. Biosci. 180(1–2), 29–48 (2002)
O. Diekmann, J.A.P. Heesterbeek, J.A.J. Metz, On the definition and the computation of the basic reproduction ratio r 0 in models for infectious diseases in heterogeneous populations. J. Math. Biol. 28, 365–282 (1990)
Z. Zhang, S. Kundu, J..P. Tripathi, S. Bugalia, Stability and hopf bifurcation analysis of an sveir epidemic model with vaccination and multiple time delays. Chaos Solitons Fractals 131, 109483 (2020)
Average life expectancy of India. https://data.worldbank.org/indicator/SP.DYN.LE00.IN?locations=IN. Accessed 5 Feb 2020
Coronavirus (COVID19) Statistics India. https://www.worldometers.info/coronavirus/country/india/. Accessed 12 Dec 2022
COVID-19 Vaccinations - India. https://github.com/owid/covid-19-data/blob/master/public/data/vaccinations/country_data/India.csv. Accessed 7 Mar 2023
K.S. Mathur, P. Narayan, Dynamics of an sveirs epidemic model with vaccination and saturated incidence rate. Int. J. Appl. Comput. Math. 4(5), 118 (2018)
B.D. Hassard, B.D. Hassard, N.D. Kazarinoff, Y.H. Wan et al., Theory and Applications of hopf Bifurcation (CUP Archive, 1981)
Acknowledgements
The research work of P. Tamilalagan and B. Krithika is supported by National Board for Higher Mathematics, Department of Atomic Energy, Mumbai, India under the Grant no. 02011/8/2020/NBHM/R &D-II/8071.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Appendices
Appendix 1
Appendix 2
Proof of Theorem 3(c)
For \(\tau _1=0\), the Eq. (11) becomes
where
If \(\lambda =im\) is a root of the Eq. (19), we obtain
Where,
Let \(m^2=t\). Then Eq. (20) becomes
Consider that Hypothesis 2: \(t_{20}\) is a positive root of Eq. (21). Then Eq. (20) has a positive root \(m_{20}=\sqrt{t_{20}}\) and further we obtain the following
with
Also, we obtain, Re\([d\lambda /d\tau _2]^{-1}_{\lambda =im_{20}}\ne 0\) if \(\left. \varUpsilon _2^{'}(t)=\frac{d\varUpsilon _2(t)}{dt}\right| _{t=t_{20}}\ne 0\) holds, where \(\varUpsilon _2(t)=t^5+P_{61}t^4+P_{62}t^3+P_{63}t^2+P_{64}t+P_{65}.\) Thus according to the Hopf bifurcation theorem in [45], the system undergoes Hopf bifurcation whenever the above transversality condition holds. \(\hfill\square\)
Proof of Theorem 3(d)
When \(\tau _1=\tau _2=\tau >0\), the Eq. (11) becomes
where
Multipliying \(e^{\lambda \tau }\) on both sides of Eq. (22), we get the following
Let \(\lambda =im\) is a root of the Eq. (23), then we get the following equations
Where,
From the above set of equations, we obtain,
with,
Where we obtain the following equation,
We consider the Hypothesis 3: \(m_{0}\) is a positive root of Eq. (25). If the Hypothesis 3 holds, then, we arrive at the following
Following in similar manner as in the above cases, we see that Re\([d\lambda /d\tau ]^{-1}_{\lambda =im_{30}}\ne 0\) if \(\left. \varUpsilon _3^{'}(t)=\frac{d\varUpsilon _3(t)}{dt}\right| _{t=t_{0}}\ne 0\) holds, where \(\varUpsilon _3(t)=t^5+P_{71}t^4+P_{72}t^3+P_{73}t^2+P_{74}t+P_{75}.\) with \(P_{71}=-{A_{{31}}}^{2}+2\,{\alpha _{{11}}}^{2}-4\,\alpha _{{12}}\), \(P_{72}=2M_{{33}} \left( \alpha _{{15}}-f_{{14}} \right) \left( \alpha _{{14} }-f_{{13}} \right) A_{{35}}+2\, \left( \alpha _{{15}}+f_{{14}} \right) A_{{35}}M_{{33}} \left( -\alpha _{{14}}-f_{{13}} \right)\), \(P_{73}=- \left( \alpha _{{14}}-f_{{13}} \right) A_{{33}}+ \left( -\alpha _{{12} }+f_{{11}} \right) A_{{35}}\), \(P_{74}=- \left( \alpha _{{15}}+f_{{14}} \right) A_{{31}}+ \left( -\alpha _{{13} }-f_{{12}} \right) A_{{33}}-\alpha _{{11}}A_{{35}}\) and \(P_{75}=2\,\alpha _{{11}} \left( -\alpha _{{13}}+f_{{12}} \right)\). Thus according to the Hopf bifurcation theorem in [45], we determine the existence of Hopf bifurcation whenever the Hypothesis 3 and the above transversality condition holds. \(\hfill\square\)
Proof of Theorem 3(e)
When \(\tau _1\ge 0\) and \(\tau _2\in (0,\tau _{20})\). Letting \(\lambda =im\) in Eq. (11) and simplifying further we obtain
Where,
By simple calculation, we obtain the following characteristic equation
where,
We consider the Hypothesis 4: \(m_{100}\) is a positive root of Eq. (30). If the Hypothesis 4 holds, then, we arrive at the following
Following in similar manner as in the above cases, we see that Re\([d\lambda /d\tau ]^{-1}_{\lambda =im_{100}}\ne 0\) if \(\left. \varUpsilon _4^{'}(t)=\frac{d\varUpsilon _4(t)}{dt}\right| _{t=t_{0}}\ne 0\) holds, where \(\varUpsilon _4(t)=t^5+P_{81}t^4+P_{82}t^3+P_{83}t^2+P_{84}t+P_{85}\). Thus according to the Hopf bifurcation theorem in [45], we determine the existence of Hopf bifurcation whenever the Hypothesis 4 and the above transversality condition holds. \(\hfill\square\)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Krithika, B., Tamilalagan, P. Unveiling the spread of epidemics involving partial immunity and reinfection: insights from a time-delayed mathematical model. Eur. Phys. J. Spec. Top. 232, 2657–2673 (2023). https://doi.org/10.1140/epjs/s11734-023-00995-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1140/epjs/s11734-023-00995-2