Abstract
Differently from computed tomography (CT), well-defined terminology for chest radiography (CXR) findings and standardized reporting in the setting of known or suspected COVID-19 are still lacking. We propose a revision of CXR major imaging findings in SARS-CoV-2 pneumonia derived from the comparison of CXR and CT, suggesting a precise and standardized terminology for CXR reporting. This description will consider asymptomatic patients, symptomatic patients, and patients with SARS-CoV-2-related pulmonary complications. We suggest using terms such as ground-glass opacities, consolidation, and reticular pattern for the most common findings, and characteristic chest radiographic pattern in presence of one or more of the above-mentioned findings with peripheral and mid-to-lower lung zone distribution.
Similar content being viewed by others
Introduction
Chest radiography (CXR) quickly became the initial imaging modality for the diagnosis and clinical management of COVID-19 pneumonia in many countries, despite lower sensitivity than computed tomography (CT) [1]. CXR has several advantages, including lower risk of contamination, lower costs and radiation exposure, and higher availability. Most EU and US imaging guidelines recommend the use of CXR as the initial imaging modality for the diagnosis of SARS-CoV-2 infection, especially in high prevalence settings [2, 3].
Thus, for patients with suspected or confirmed SARS-CoV-2 infection, CXR is routinely performed, while chest CT is generally reserved for cases with a discrepancy between clinical, laboratory, and radiologic findings.
Patients with COVID-19 pneumonia show typical CT imaging findings which are well described in the literature [3,4,5]. The expert consensus statement on reporting chest CT, published by the Radiological Society of North America, provides standardized terminology based on typical, indeterminate, and atypical features [3]. In contrast, a well-defined terminology and a standardized reporting for CXR imaging in COVID-19 pneumonia are still lacking, with multiple terms that have been used, such as alveolar opacities, reticular opacities, reticulo-nodular opacities, mild opacities, ground–glass opacities, and consolidation.
Our aim is to provide a series of cases illustrating CXR major findings in COVID-related pneumonia and to suggest precise and standardized terminology for CXR reporting, thanks to the comparison with CT. In order to do that, we decided to use definitions from “Glossary of terms for thoracic imaging” published by the multidisciplinary Fleischner Society [6].
This description will consider three common clinical scenarios in patients with confirmed SARS-CoV-2 infection: asymptomatic patients, symptomatic patients, and patients with pulmonary complications related to COVID-19 pneumonia.
Typical COVID-19 pneumonia CXR findings in clinical scenarios
Asymptomatic patients
Statements from world-wide scientific societies agree that imaging is not recommended for the diagnosis and management of asymptomatic individuals with suspected or confirmed SARS-CoV-2 infection [2, 7, 8].
Despite this, there was a significant increase in rates of chest CT utilization in ED following the start of the COVID pandemic, compared to pre-pandemic baseline [9]. It is desirable a stricter observation of the guidelines addressing new waves of this pandemic and/or future pandemics with the objective of avoiding inappropriate CT exams and of reducing infection risks [9, 10].
In most asymptomatic patients with confirmed SARS-CoV-2 infection, CXR shows no lung abnormalities [11]. On the contrary, according to a meta-analysis conducted by Vafea and colleagues [12], chest CT showed positive results in 63% of asymptomatic patients with confirmed SARS-CoV-2 infection. The most common CT finding was ground–glass opacity (GGO) in 71%, which may be not detected on CXR [12].
Teaching points:
Imaging is not indicated in asymptomatic patients with suspected or confirmed COVID-19 without risk factors for disease progression.
In asymptomatic COVID-19 patients the most common CT finding is bilateral GGO, while chest radiography has limited value.
Symptomatic patients
Not all symptomatic patients with COVID-19 have an abnormal CXR, with varying percentages reported depending on the population studied. In one large series of non-hospitalized patients presenting to an urgent care, 58.3% had a normal CXR and an additional 30.7% had only mild radiographic abnormalities [13]. In contrast, 69% of hospitalized patients had an abnormal CXR at the time of hospital admission on one study [1].
The most typical CXR lung abnormalities of COVID-19 pneumonia are discussed below.
Ground–glass opacity
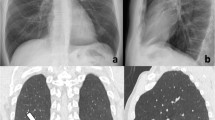
Ground–glass opacity (GGO) is defined on both, CXR and CT scans, as hazy increased lung opacity within which margins of pulmonary vessels are still visible, and less dense than consolidation which obscures vessel walls (Fig. 1) [6].
49-year-old male patient presented at the Emergency Department with one-week history of fever and recent onset of dyspnea. He was tested positive for SARS-CoV-2 and hospitalized. A Antero-posterior CXR at the admission to the ED showing peripheral patchy ground glass opacities in both lungs (close view in B, corresponding to the white box in A, better displays the hazy aspects of GGO). C, D Coronal reformatted and axial CT scan confirm the presence of bilateral peripheral and basal predominant GGO; this pattern is typically seen in the early phase of diffuse alveolar damage related to COVID-19
GGO depicted on CT is often not radiographically visible. In addition, the lower anatomic resolution of CXR can lead to an overlap of opacities, making harder to distinguish pure GGO from crazy paving or consolidation, that instead are well identified on CT (Figs. 2, 3, and 4).
57-year-old male admitted to the hospital with COVID-19 pneumonia, from 7 days under ventilation support on continuous positive airway pressure (CPAP). A Antero-posterior CXR showed bilateral interstitial-alveolar infiltrates. In the left middle lung periphery, an opaquer area of consolidation was seen (close view in B, corresponding to the white box in A, better displays the consolidation). C, D Coronal reformatted and axial CT scan revealed a greater consolidation in the right lung. The consolidative area detected on the CXR in the left middle lung was caused by the overlap in the axial plane of ground glass opacities, reticulation, and consolidation
62-year-old male admitted to the Emergency Department with persistent cough and dyspnea for 4 days. Patient was tested positive for SARS-CoV-2 infection. A Postero-anterior CXR shows bilateral patchy ground glass opacities (GGO). The opacity in the right upper lobe seems to be opaquer with superimposed subtle reticulation (close view in B, corresponding to the white box in A). C, D Coronal reformatted and axial CT scan confirm the presence of patchy bilateral GGO, being denser in the dorsal segment of the right upper lobe for the compresence of distortive reticulation
69-year-old male patient with one week history of respiratory symptoms, presented at the ED for the worsening of dyspnea. He was tested positive for SARS-CoV-2 infection and hospitalized. A Antero-posterior CXR at the admission to the ED shows bilateral opacities with a greater involvement of the right lung (in the close view in B, corresponding to the white box in A, it is difficult to differentiate the radiological patterns). C, D Coronal reformatted and axial CT scan clarifies the radiologic patterns: ground glass is predominant in the middle lung lobe, while crazy paving is detected in dorsal regions of the right lower lobe
GGO represents a typical finding of COVID-19 pneumonia during the acute exudative phase of lung injury, often referred to as acute diffuse alveolar damage (DAD), which typically occurs within 7 days of infection onset (Fig. 1) [14, 15].
As a result, suboptimal CXR diagnostic performance is expected in patients who have experienced symptoms for few days (Fig. 5) [16].
28-year-old male with essential thrombocythemia, tested positive for SARS-CoV-2. Patient had few symptoms and underwent CXR and chest CT scan 5 days after the molecular test. A Postero-anterior CXR showed no lung abnormalities. B, C Coronal reformatted and axial CT scan showed small pseudo nodular isolated ground-glass opacities (arrowheads), consistent with initial lung involvement
In the early phase of COVID-19 pneumonia, GGO typically presents as patchy areas, with a peripheral distribution in the lower lungs. As the disease progresses, GGO tend to become more confluent and the extent of abnormalities increases [17]. Progression is well depicted both on CXR and CT [18].
Another common CT finding of COVID-19 pneumonia, usually seen with clinical progression towards the peak stage of disease, is crazy-paving. This CT pattern is similar to ground glass, with superimposed thickened interlobular and intralobular septa [17, 19]. Based on our experience, CXR is unable to distinguish this pattern from GGO alone, or from a combination of GGO with early consolidation (Figs. 6 and 7).
62-year-old female with one-week history of cough and fever, admitted to the ED for the recent onset of dyspnea. She was tested positive for SARS-CoV-2 infection and hospitalized. A Antero-posterior CXR showing diffuse bilateral interstitial-alveolar opacities (close view in B, corresponding to the white box in A, highlights predominant ground glass and reticular patterns). C, D Coronal reformatted and axial CT scan clarifies the radiologic pattern, showing extensive bilateral opacities with crazy-paving pattern
67-year-old male hospitalized one week before with COVID-19 pneumonia. A Antero-posterior CXR showing bilateral peripheral predominant GGO and subtle reticulation prevailing in the middle-lower regions (in the close view in B, corresponding to the white box in A, hazy opacities and reticular alteration are better depicted). C, D Coronal reformatted and axial CT scan highlight that the lung abnormalities clearly present a crazy-paving pattern, which could not be detected on the CXR
Consolidation
Consolidation is a homogeneous increase in lung density that obscures the margins of vessels and airways walls, the latter appearing as air bronchograms [6]. While CXR can accurately detect pulmonary consolidation, it commonly underestimates their extent [20]. For example, Choi et al. [20] compared a series of CXRs and CTs performed on the same day in the same patients with COVID-19, with CXR detecting only 25% of the lung abnormalities; the study concluded that the disease extension was the main factor driving the visibility of lung opacities on CXR. Similarly, Ippolito et al. [18] reported CXR sensitivity of 57% for COVID pneumonia, with higher values when the symptom onset was 5 days or more, typically when there is a greater lung involvement.
Despite this, an abnormal CXR has clinical utility and validity in quantifying the extent of COVID-19 pneumonia as a marker of severity, particularly when there is extensive and severe disease [21].
In patients with COVID-19, consolidation is a hallmark of organizing pneumonia, most commonly seen during progressive and peak stages of the disease, 5–13 days after symptom onset (Figs. 8 and 9) [17, 19, 22]. This seems to reflect the “organizing phase” of diffuse alveolar damage (DAD), which has been reported as the main histopathologic process in COVID-19 pneumonia [23, 24].
55-year-old male patient with COVID-19 pneumonia after 14 days of hospitalization in sub-intensive care unit. A Antero-posterior CXR showed bilateral interstitial–alveolar infiltrates with predominant consolidative pattern in the middle-lower lung zones (close view in B, corresponding to the white box in A). C, D Coronal reformatted and axial CT scan confirm the presence of bilateral patchy consolidation with predominant peribronchial distribution, consistent with radiologic pattern of organizing pneumonia
46-year-old male tested positive for SARS-CoV-2 with one-week history of fever and cough. He presented to the ED for the onset of dyspnea. Antero-posterior CXR, as well as CT, well documented the progression of COVID-19 pneumonia from diffuse ground glass opacities on A, D day 0 to consolidation with peribronchovascular distribution consistent with organizing pneumonia on B, E day 8. C, F On day 15, AP CXR and axial CT show more extensive consolidation with a gravity-dependent gradient
Reticular pattern
Reticular pattern is defined as a collection of innumerable small linear opacities that by summation produce an appearance resembling a net [6]. Reticulation is more easily seen at thin-section CT as interlobular and intralobular septal thickening. While reticulation has been described as more common than alveolar opacities on CXR in COVID-19 patients, these two patterns are usually seen together [18, 25]. When reticulation is the predominant pattern in patients with suspected or confirmed COVID-19, the main differential diagnosis is the acute interstitial edema, particularly in elderly patients with a high probability of having underlying heart disease.
Reticulation, in combination with other patterns, is more frequent in the advanced phase of COVID-19 pneumonia, often described as the “reabsorption phase,” characterized by the gradual decrease in density of GGO and consolidation and the presence of areas of parenchymal distortion (Figs. 10 and 11) [15, 19].
62-year-old male presented at the Emergency Department with recent history of fever and cough (for 2 weeks). He was tested positive for SARS-CoV-2 and hospitalized. A Antero-posterior CXR showing bilateral peripheral predominant reticulation, diffuse on the coronal plane (close view in B, corresponding to the white box in A, better shows opacities). C, D Coronal reformatted and axial CT scan confirm the presence of bilateral distortive reticulation, with peripheral predominant distribution. Low-density GGO areas are also present. Findings are suggestive of late “reabsorption” phase of COVID-19 pneumonia
57-year-old male hospitalized for 17 days due to COVID-19 pneumonia. A Antero-posterior CXR shows bilateral interstitial–alveolar infiltrates, with tendency to consolidation in the lower lobes. It is not possible to clearly differentiate the patterns on the basis of CXR. B, C Coronal reformatted and axial CT clarify the patterns: consolidation with aspect of organizing pneumonia are the prevalent pattern, in association with distortive reticular alterations. Findings are consistent with a late-phase COVID-19 pneumonia
Definition of a characteristic chest radiographic pattern
While the lung abnormalities described above are not pathognomonic of COVID-19 pneumonia, in the context of a pandemic with a high pre-test probability of SARS-CoV-2 infection, the presence of these findings is highly suggestive of COVID-19 pneumonia [26].
The pattern of bilateral patchy or confluent ground–glass opacities and/or consolidation in the lung periphery with a mid-to-lower lung distribution is the most common (Fig. 12) and is the radiographic equivalent of the CT typical pattern described in the joint statement on reporting by American College of Radiology, Radiologic Society of North America and Society of Thoracic Radiology [3].
63-year-old male patient tested positive for SARS-CoV-2 at the admission to the ED. He had cough and fever from 5 days and recent onset of dyspnea. A Antero-posterior CXR shows bilateral lung ground glass opacities with peripheral and patchy distribution. B, C Coronal reformatted and axial CT scan confirm the presence of bilateral lung infiltrates with predominant ground glass pattern: the aspect and distribution of the abnormalities is typical for COVID-19 pneumonia
Teaching point:
Chest radiography can help clinicians by raising a suspicion for COVID-19 infection in symptomatic patients.
In the setting of a high prevalence of COVID-19, the presence of bilateral patchy or confluent ground-glass opacities or consolidation in a peripheral and mid-to-lower lung zone distribution is highly suggestive for COVID-19 pneumonia.
Temporal changes of typical COVID-19 patterns on chest radiography reflect those reported by longitudinal studies of chest CT.
Chest radiography of COVID-19 complications
The clinical outcome of symptomatic patients with COVID-19 pneumonia generally correlates with the extent and severity of lung involvement, and patients with severe lung disease have a higher risk of developing complications [27, 28].
In particular, COVID-19 is a risk factor for pulmonary embolism (PE) or pulmonary in situ thrombosis, and it has been postulated that such complications may already be present at the moment of the hospital admission [29].
CT pulmonary angiography is the most accurate test for the diagnosis of PE, with an incidence of 17–35% in patients with COVID-19 who underwent CT angiography [30].
CXR has a limited value in the diagnosis of PE but it may raise the suspicion of it when there is evidence of pulmonary infarction: a triangular or dome-shaped opacity with the base abutting the pleural surface and the apex directed toward the hilum [6].
Acute respiratory distress syndrome (ARDS) is the most common cause for patient admission to intensive care unit (ICU), and the main cause of mortality in COVID-19 patients [5]. On CXR, rapid progression of bilateral alveolar opacities, which tend to have a dependent distribution, within 7–12 days after the onset of COVID-19 should raise the suspicion for ARDS (Figs. 13 and 14) [30, 31]. In the late phase of ARDS, imaging can depict signs of fibrotic organizing pneumonia, with architectural distortion and traction bronchiectasis [31].
74-year-old female patient with COVID-19 pneumonia after 8 days of hospitalization in intensive care unit. A Antero-posterior CXR showing bilateral interstitial–alveolar with predominant consolidative pattern (in the close view in B, corresponding to the white box in A, a consolidation with partial air bronchogram is well detected). C, D Coronal reformatted and axial CT scan show bilateral consolidation with gravity dependent distribution: the radiographic pattern is suggestive of initial evolution into acute respiratory distress syndrome (ARDS)
45-year-old female patient with COVID-19 pneumonia admitted to the ICU two days before images were acquired. A Antero-posterior CXR showing extensive bilateral consolidation with lower predominance. B, C Axial CT scan acquired in supine (B) and prone (C) position well demonstrated the dependent distribution of consolidation: note the ventral distribution in the prone acquisition
Patients with severe COVID-19 on mechanical ventilation with high-positive pressure or continuous positive airway pressure (cPAP) can develop barotrauma events including pneumothorax and pneumomediastinum (Fig. 15) [32, 33].
79-year-old male admitted to the hospital with COVID-19 pneumonia, requiring ventilation support on continuous positive airway pressure (CPAP) for two weeks. A Antero-posterior CXR showed bilateral interstitial–alveolar infiltrates and a hyperlucent line on the right profile of the upper mediastinum which raised the suspicion of pneumomediastinum (close view in B, corresponding to the white box in A, better displays the hyperlucent line–arrowheads). C, D Coronal reformatted and axial CT scan confirmed the presence of pneumomediastinum (arrowheads)
Despite the low frequency of coinfections described in patients with COVID-19, early diagnosis of superimposed pneumonia is crucial due to the association with poor outcomes [34]. Hospital-acquired bacterial or fungal infections are frequent in ICU patients [35]. CXR can help in diagnosing a superimposed bacterial infection showing typical radiographic features such as unilateral lobar or segmental air-space opacities with or without air bronchograms (Fig. 16).
53-year-old female patient presented at the ED with fever and chest pain. She was tested positive for SARS-CoV-2 infection and also had a positive urinary antigen test for pneumococcal pneumonia. A Postero-anterior CXR shows an extensive consolidation involving the mid-lower left lung; in the middle zone of the right lung, patchy consolidation with adjacent hazy opacities are seen. B, C Axial CT scan confirm the presence of consolidation involving the entire left lower lobe, suggesting a bacterial pneumonia. In the right lung patchy confluent consolidation with hazy margins are detected, possible expression of bilateral extension of bacterial pneumonia or COVID-pneumonia
Furthermore, the evidence on CXR of multiple pulmonary nodules or lung cavitation should promptly raise the suspicion of superimposed fungal and/or mycobacterial infection or necrotizing bacterial infection (Fig. 17).
72-year-old female patient with kidney transplantation history, currently under immunosuppressive treatment with Tacrolimus. She was admitted to the ED for the recent onset of dyspnea and tested positive for SARS-CoV-2 infection. During the hospitalization she had positive PCR test for Aspergillus on BAL. A Antero-posterior supine CXR acquired on the 5th day from hospitalization. Bilateral interstitial–alveolar infiltrates are present, involving the mid-lower regions of the lungs. In the lower zone of the right lung a round consolidation is detected (arrowhead). B Axial CT scan acquired on the same day of the CXR in A. It confirms the presence of extensive bilateral ground glass opacities and a round consolidation in the middle lobe (arrow), the latter is not a typical CT sign of COVID-19 pneumonia. C Antero-posterior supine CXR acquired on the 8th day after hospitalization. There is a mild worsening of the radiographic findings compared to the previous CXR. However, the round consolidation (arrowhead) seems to be less dense than in the prior control. D Axial CT scan acquired two days after the CXR in C. The CT clarifies the aspect of the middle lobe consolidation, which is now excavated (arrow), raising the suspicion of a fungal infection. In the left lung an anterior pneumothorax is present, which could not be clearly detected on the supine CXR
Teaching point:
Chest radiography represents a valid imaging tool for monitoring COVID-19 patients with severe pulmonary disease suspected of developing complications, particularly those with clinical deterioration and for detecting barotrauma in patients under mechanical ventilation or cPAP.
COVID-19 pneumonia follow-up
The risk of developing long-term pulmonary sequelae after COVID-19 is still subject to debate. CXR features in the follow-up of patients recovering from COVID-19 pneumonia are not well described and deserve more extensive research. Vice versa, longitudinal studies of COVID-19 survivors discharged from the hospital are focused on chest CT findings. They show that lung abnormalities are relatively common after SARS-CoV-2 and may persist up to 12 months after discharge [28, 36]. The lung abnormalities tend to reduce in extent at 1-year follow-up; the improvement in radiologic findings over time is in line with data from SARS-CoV-1 in which the prevalence of lung abnormalities also decreased with time. Parenchymal bands and reticulations are among the most common CT findings at follow-up and seem to be sign of organizing pneumonia rather than markers of fibrosis [37, 38].
Conclusion
In patients with suspicion of COVID-19 pneumonia, the use of the terms ground–glass opacities, consolidation, and reticular pattern is suggested on CXR. Then, location of these findings should be reported, with peripheral and mid-to-lower lung zone distribution being a characteristic CXR pattern of COVID-19 pneumonia and highly suggestive for the diagnosis particularly in population with high prevalence of COVID-19 infection. Other typical findings of COVID-19 pneumonia, such as a crazy-paving pattern, can be easily shown on chest CT, but are difficult to be detected or differentiated from ground–glass, consolidative, and reticular abnormalities on CXR.
Similarly to CT, CXR findings and their extent can help to define the phase of this viral pneumonia, to monitor the disease, and potentially to predict outcomes.
References
Wong HYF, Lam HYS, Fong AH-T, et al (2020) Frequency and distribution of chest radiographic findings in COVID-19 positive patients. Radiology 201160https://doi.org/10.1148/radiol.2020201160
Nair A, Rodrigues JCL, Hare S et al (2020) A British Society of Thoracic Imaging statement: considerations in designing local imaging diagnostic algorithms for the COVID-19 pandemic. Clin Radiol 75:329–334. https://doi.org/10.1016/j.crad.2020.03.008
Simpson S, Kay FU, Abbara S et al (2020) Radiological Society of North America Expert Consensus document on reporting chest CT findings related to COVID-19: endorsed by the Society of Thoracic Radiology, the American College of Radiology, and RSNA. Radiol Cardiothorac Imaging 2:e200152. https://doi.org/10.1148/ryct.2020200152
Chung M, Bernheim A, Mei X et al (2020) CT imaging features of 2019 novel coronavirus (2019-nCoV). Radiology 295:202–207. https://doi.org/10.1148/radiol.2020200230
Salehi S, Abedi A, Balakrishnan S, Gholamrezanezhad A (2020) Coronavirus Disease 2019 (COVID-19): a systematic review of imaging findings in 919 patients. Am J Roentgenol 215:87–93. https://doi.org/10.2214/AJR.20.23034
Hansell DM, Bankier AA, Mcloud TC et al (2008) Fleischner Society : glossary of terms for thoracic imaging. Radiology 246:697–722. https://doi.org/10.1148/radiol.2462070712
Rubin GD, Ryerson CJ, Haramati LB et al (2020) The role of chest imaging in patient management during the COVID-19 pandemic: a multinational consensus statement from the Fleischner Society. Radiology 296:172–180. https://doi.org/10.1148/radiol.2020201365
Akl EA, Blažić I, Yaacoub S et al (2021) Use of chest imaging in the diagnosis and management of COVID-19: a WHO rapid advice guide. Radiology 298:E63–E69. https://doi.org/10.1148/radiol.2020203173
Loftus TM, Wessling EG, Cruz DS et al (2022) Impact of the COVID pandemic on emergency department CT utilization: where do we go from here? Emerg Radiol 29:879–885. https://doi.org/10.1007/s10140-022-02071-z
Lopes N, Vernuccio F, Costantino C et al (2020) An Italian guidance model for the management of suspected or confirmed COVID-19 patients in the primary care setting. Front Public Health 8:1–9. https://doi.org/10.3389/fpubh.2020.572042
Kuo BJ, Lai YK, Tan MLM, Goh X-YC (2021) Utility of screening chest radiographs in patients with asymptomatic or minimally symptomatic COVID-19 in Singapore. Radiology 298:E131–E140. https://doi.org/10.1148/radiol.2020203496
Tsikala Vafea M, Atalla E, Kalligeros M et al (2020) Chest CT findings in asymptomatic cases with COVID-19: a systematic review and meta-analysis. Clin Radiol 75:876.e33-876.e39. https://doi.org/10.1016/j.crad.2020.07.025
Weinstock MB, Echenique A, Russel JW et al (2020) Chest x-ray findings in 636 ambulatory patients with COVID-19 presenting to an urgent care center: a normal chest x-ray is no guarantee. J Urgent Care Med 14:13–18
Martinez-Jimenez S, Pettavel PP (2018) Pathologic patterns of injury: diffuse alveolar damage. In: Martinez-Jimenez S, Rosado-de-Christenson ML, Carter BW (eds) Specialty imaging: HRCT of the lung (Second Edition). Elsevier Inc., pp 62–65
Kligerman SJ, Franks TJ, Galvin JR (2013) From the radiologic pathology archives: organization and fibrosis as a response to lung injury in diffuse alveolar damage, organizing pneumonia, and acute fibrinous and organizing pneumonia. Radiographics 33:1951–1975. https://doi.org/10.1148/rg.337130057
Stephanie S, Shum T, Cleveland H et al (2020) Determinants of chest radiography sensitivity for COVID-19: a multi-institutional study in the United States. Radiol Cardiothorac Imaging 2:e200337. https://doi.org/10.1148/ryct.2020200337
Bernheim A, Mei X, Huang M, et al (2020) Chest CT findings in coronavirus disease-19 (COVID-19): relationship to duration of infection. Radiology 200463https://doi.org/10.1148/radiol.2020200463
Ippolito D, Pecorelli A, Maino C et al (2020) Diagnostic impact of bedside chest X-ray features of 2019 novel coronavirus in the routine admission at the emergency department: case series from Lombardy region. Eur J Radiol 129:109092. https://doi.org/10.1016/j.ejrad.2020.109092
Pan F, Ye T, Sun P, et al (2020) Time course of lung changes on chest CT during recovery from 2019 novel coronavirus (COVID-19) pneumonia. Radiology 200370https://doi.org/10.1148/radiol.2020200370
Choi H, Qi X, Yoon SH et al (2020) Extension of coronavirus disease 2019 on chest CT and implications for chest radiographic interpretation. Radiol Cardiothorac Imaging 2:e200107. https://doi.org/10.1148/ryct.2020200107
Toussie D, Voutsinas N, Finkelstein M et al (2020) Clinical and chest radiography features determine patient outcomes in young and middle-aged adults with COVID-19. Radiology 297:E197–E206. https://doi.org/10.1148/radiol.2020201754
Rousan LA, Elobeid E, Karrar M, Khader Y (2020) Chest x-ray findings and temporal lung changes in patients with COVID-19 pneumonia. BMC Pulm Med 20:245. https://doi.org/10.1186/s12890-020-01286-5
Borczuk AC, Salvatore SP, Seshan SV et al (2020) COVID-19 pulmonary pathology: a multi-institutional autopsy cohort from Italy and New York City. Mod Pathol 33:2156–2168. https://doi.org/10.1038/s41379-020-00661-1
Henkel M, Weikert T, Marston K et al (2020) Lethal COVID-19: radiologic-pathologic correlation of the lungs. Radiol Cardiothorac Imaging 2:e200406. https://doi.org/10.1148/ryct.2020200406
Vancheri SG, Savietto G, Ballati F et al (2020) Radiographic findings in 240 patients with COVID-19 pneumonia: time-dependence after the onset of symptoms. Eur Radiol 30:6161–6169. https://doi.org/10.1007/s00330-020-06967-7
Smith DL, Grenier J-P, Batte C, Spieler B (2020) A characteristic chest radiographic pattern in the setting of the COVID-19 pandemic. Radiol Cardiothorac Imaging 2:e200280. https://doi.org/10.1148/ryct.2020200280
Han X, Fan Y, Alwalid O et al (2021) Six-month follow-up chest CT findings after severe COVID-19 pneumonia. Radiology 299:E177–E186. https://doi.org/10.1148/radiol.2021203153
Wu X, Liu X, Zhou Y et al (2021) 3-month, 6-month, 9-month, and 12-month respiratory outcomes in patients following COVID-19-related hospitalisation: a prospective study. Lancet Respir Med 9:747–754. https://doi.org/10.1016/S2213-2600(21)00174-0
Lodigiani C, Iapichino G, Carenzo L et al (2020) Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb Res 191:9–14. https://doi.org/10.1016/j.thromres.2020.04.024
Kwee TC, Kwee RM (2020) Chest CT in COVID-19: What the radiologist needs to know. Radiographics 40:1848–1865. https://doi.org/10.1148/rg.2020200159
Gosangi B, Rubinowitz AN, Irugu D et al (2022) COVID-19 ARDS: a review of imaging features and overview of mechanical ventilation and its complications. Emerg Radiol 29:23–34. https://doi.org/10.1007/s10140-021-01976-5
Revzin MV, Raza S, Warshawsky R et al (2020) Multisystem imaging manifestations of COVID-19, part 1: viral pathogenesis and pulmonary and vascular system complications. Radiographics 40:1574–1599. https://doi.org/10.1148/rg.2020200149
McGuinness G, Zhan C, Rosenberg N et al (2020) Increased incidence of barotrauma in patients with COVID-19 on invasive mechanical ventilation. Radiology 297:E252–E262. https://doi.org/10.1148/radiol.2020202352
Rodriguez-Nava G, Yanez-Bello MA, Trelles-Garcia DP, et al (2020) A retrospective study of coinfection of SARS-CoV-2 and Streptococcus pneumoniae in 11 hospitalized patients with severe COVID-19 pneumonia at a single center. Medical Science Monitor 26:. https://doi.org/10.12659/MSM.928754
Søgaard KK, Baettig V, Osthoff M et al (2021) Community-acquired and hospital-acquired respiratory tract infection and bloodstream infection in patients hospitalized with COVID-19 pneumonia. J Intensive Care 9:10. https://doi.org/10.1186/s40560-021-00526-y
Vijayakumar B, Tonkin J, Devaraj A et al (2022) CT lung abnormalities after COVID-19 at 3 months and 1 year after hospital discharge. Radiology 303:444–454. https://doi.org/10.1148/RADIOL.2021211746
Zhao Y, Yang C, An X et al (2021) Follow-up study on COVID-19 survivors one year after discharge from hospital. Int J Infect Dis 112:173–182. https://doi.org/10.1016/j.ijid.2021.09.017
Poerio A, Carlicchi E, Lotrecchiano L, et al (2022) Evolution of COVID-19 pulmonary fibrosis–like residual changes over time — longitudinal chest CT up to 9 months after disease onset: a single-center case series. SN Compr Clin Med 4https://doi.org/10.1007/s42399-022-01140-1
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Flor, N., Fusco, S., Blazic, I. et al. Interpretation of chest radiography in patients with known or suspected SARS-CoV-2 infection: what we learnt from comparison with computed tomography. Emerg Radiol 30, 363–376 (2023). https://doi.org/10.1007/s10140-022-02105-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10140-022-02105-6