Abstract
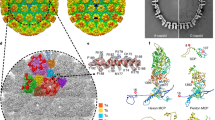
THREE-DIMENSIONAL structures of several spherical viruses have been determined by electron microscopy1–6 and X-ray crystallography7,8. We report here the first three-dimensional structure of the complex between an intact virus and Fab fragments of a neutralizing monoclonal antibody. The antibody is against VP4, one of the two outer capsid proteins of rotaviruses. These large icosahedral viruses cause gastroenteritis in children and young animals and account for over a million human deaths annually9. VP4 in these viruses has been implicated in several important functions such as cell penetration, haemagglutination, neutralization and virulence10–12. Here we demonstrate that the surface spikes on rotavirus particles are made up of VP4. Antigenic sites are located near the distal ends of the spikes and two Fab fragments bind to each of the sixty spikes. The mass of the spike indicates that it is a dimer of VP4. The bilobed structure at the distal end of the spike may be involved in both the attachment to the cell and in viral penetration. A novel feature in the virus–Fab complex is the structural difference between the two chemically equivalent Fab fragments on each spike, which could be indicative of variations in the Fab elbow angles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Crowther, R. A. & Klug, A. A. Rev. Biochem. 44, 161–182 (1975).
Vogel, R. H., Provencher, S. W., von-Bonsdorff, C.-H., Adrian, M. & Dubochet, J. Nature 320, 533–535 (1986).
Fuller, S. D. Cell 48, 923–934 (1987).
Prasad, B. V. V., Wang, G. J., Clerx, J. P. M. & Chiu, W. J. molec. Biol. 199, 269–275 (1988).
Baker, T. S., Drak, J. & Bina, M. Proc. natn. Acad. Sci. U.S.A. 85, 422–426 (1988).
Schrag, J. D., Prasad, B. V. V., Rixon, F. J. & Chiu, W. Cell 56, 651–660 (1989).
Rossmann, M. G. & Reuckert, R. R. Microbiol. Sci. 4, 206–214 (1987).
Acharya, R. et al. Nature 337, 709–716 (1989).
De Zoysa, I. & Feachem, R. G. Bull. WHO 63, 569–583 (1985).
Estes, M. K. & Cohen, J. Microbiol. Rev. 53, 410–449 (1989).
Kapikian, A. Z. & Chanock, R. M. in Virology (ed. Fields, B. N.) 1353–1404 (Raven, New York, 1990).
Kaljot, K. T., Shaw, R. D., Rubin, D. H. & Greenberg, H. B. J. Virol. 62, 1136–1144 (1988).
Dubochet, J. et al. Q. Rev. Biophys. 21, 129–228 (1988).
Davies, D. R. & Metzger, H. A. Rev. Immun. 1, 87–117 (1983).
Amit, A. G., Marriuzza, R. A., Phillips, S. E. V. & Poljak, R. J. Science 233, 747–753 (1986).
Colman, P. M. et al. Nature 326, 358–363 (1987).
Wilson, I. A., Skehel, J. J. & Wiley, D. C. Nature 289, 366–373 (1981).
Mackow, E. R., Barnett, J. W., Chan, H. & Greenberg, H. B. J. Virol. 63, 1661–1668 (1989).
Estes, M. K., Graham, D. Y. & Mason, B. B. J. Virol 39, 879–888 (1981).
Mackow, E. R. et al. Proc. natn. Acad. Sci. U.S.A. 85, 645–649 (1988).
Fukuhara, N., Yoshie, O., Kitaoka, S. & Konno, T. J. Virol 62, 2209–2218 (1988).
Matsuno, S. & Inouye, S. Infection and Immunity 39, 155–158 (1983).
Sabara, M., Gilchrist, J. E., Hudson, G. R. & Babiuk, L. A. J. Virol. 53, 58–66 (1985).
Mason, B. B., Graham, D. Y. & Estes, M. K. J. Virol. 46, 413–423 (1983).
Shaw, R. D., Vo, P. T., Offit, P. A., Coulson, B. S. & Greenberg, H. B. Virology 155, 434–451 (1986).
Burns, J. W., Greenberg, H. B., Shaw, R. D. & Estes, M. K. J. Virol. 62, 2164–2172 (1988).
Crowther, R. A. Phil. Trans. Roy. Soc. Lond B261, 221–230 (1971).
Vigers, G. P. A., Crowther, R. A. & Pearse, B. M. F. EMB0 J. 5, 2079–2085 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Prasad, B., Burns, J., Marietta, E. et al. Localization of VP4 neutralization sites in rotavirus by three-dimensional cryo-electron microscopy. Nature 343, 476–479 (1990). https://doi.org/10.1038/343476a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/343476a0
This article is cited by
Development and evaluation of two subunit vaccine candidates containing antigens of hepatitis E virus, rotavirus, and astrovirus
Scientific Reports (2016)
Comparative analysis of pentavalent rotavirus vaccine strains and G8 rotaviruses identified during vaccine trial in Africa
Scientific Reports (2015)
Fitting \(\alpha \) α \(\beta \) β -crystalline structure onto electron microscopy based on SO(3) rotation group theory
Journal of Combinatorial Optimization (2015)
Carbohydrate recognition by rotaviruses
Journal of Structural and Functional Genomics (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.