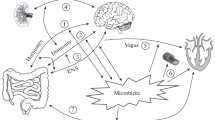
Abstract
The Gut-brain axis is a bidirectional neural and humoral signaling that plays an important role in mental disorders and intestinal health and connects them as well. Over the past decades, the gut microbiota has been explored as an important part of the gastrointestinal tract that plays a crucial role in the regulation of most functions of various human organs. The evidence shows several mediators such as short-chain fatty acids, peptides, and neurotransmitters that are produced by the gut may affect the brain’s function directly or indirectly. Thus, dysregulation in this microbiome community can give rise to several diseases such as Parkinson’s disease, depression, irritable bowel syndrome, and Alzheimer’s disease. So, the interactions between the gut and the brain are significantly considered, and also it provides a prominent subject to investigate the causes of some diseases. In this article, we reviewed and focused on the role of the largest and most repetitive bacterial community and their relevance with some diseases that they have mentioned previously.




Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Aizawa E, Tsuji H, Asahara T, Takahashi T, Teraishi T, Yoshida S, Ota M, Koga N, Hattori K, Kunugi H (2016) Possible association of Bifidobacterium and Lactobacillus in the gut microbiota of patients with major depressive disorder. J Affect Disord 202:254–257. https://doi.org/10.1016/j.jad.2016.05.038
Al-Jahmany A, Schultheiss G, Diener M (2004) Effects of dopamine on ion transport across the rat distal colon. Pflügers Archiv 448:605–612. https://doi.org/10.1007/s00424-004-1299-9
Alfonsetti M, Castelli V, d’Angelo M (2022) Are we what we eat? Impact of Diet on the gut-brain Axis in Parkinson’s Disease. Nutrients 14 https://doi.org/10.3390/nu14020380
Armstrong MJ, Okun MS (2020) Diagnosis and treatment of Parkinson Disease A Review. JAMA. https://doi.org/10.1001/jama.2019.22360. 323:548 – 60
Athauda D, Foltynie T (2016) Insulin resistance and Parkinson’s disease: a new target for disease modification? Prog Neurobiol 145:98–120. https://doi.org/10.1016/j.pneurobio.2016.10.001
Atri A (2019) The Alzheimer’s disease clinical spectrum: diagnosis and management. Med Clin 103:263–293. https://doi.org/10.1016/j.mcna.2018.10.009
Aziz MNM, Kumar J, Muhammad Nawawi KN, Raja Ali RA, Mokhtar NM (2021) Irritable bowel syndrome, Depression, and neurodegeneration: a bidirectional communication from gut to Brain. Nutrients 13. https://doi.org/10.3390/nu13093061
Baj A, Moro E, Bistoletti M, Orlandi V, Crema F, Giaroni C (2019) Glutamatergic signaling along the microbiota-gut-brain axis. Int J Mol Sci 20:1482. https://doi.org/10.3390/ijms20061482
Barichella M, Severgnini M, Cilia R, Cassani E et al (2019) Unraveling gut microbiota in Parkinson’s disease and atypical parkinsonism. Mov Disord 34:396–405. https://doi.org/10.1002/mds.27581
Barrett E, Ross RP, O’Toole PW, Fitzgerald GF, Stanton C (2012) γ-Aminobutyric acid production by culturable bacteria from the human intestine. J Appl Microbiol 113:411–417. https://doi.org/10.1111/j.1365-2672.2012.05344.x
Bear T, Dalziel J, Coad J, Roy N, Butts C, Gopal P (2021) The Microbiome-Gut-Brain Axis and Resilience to developing anxiety or depression under stress. Microorganisms 9. https://doi.org/10.3390/microorganisms9040723
Benakis C, Martin-Gallausiaux C, Trezzi JP, Melton P, Liesz A, Wilmes P (2020) The microbiome-gut-brain axis in acute and chronic brain diseases. Curr Opin Neurobiol 61:1–9. https://doi.org/10.1016/j.conb.2019.11.009
Berding K, Vlckova K, Marx W et al (2021) Diet and the microbiota–gut–brain Axis: sowing the seeds of good Mental Health. Adv Nutr 12:1239–1285. https://doi.org/10.1093/advances/nmaa181
Bibbò S, Ianiro G, Giorgio V et al (2016) The role of diet on gut microbiota composition. Eur Rev Med Pharmacol Sci 20:4742–4749
Bilen M, Dufour JC, Lagier JC et al (2018) The contribution of culturomics to the repertoire of isolated human bacterial and archaeal species. Microbiome 6:94. https://doi.org/10.1186/s40168-018-0485-5
Bourdu S, Dapoigny M, Chapuy E et al (2005) Rectal instillation of butyrate provides a novel clinically relevant model of noninflammatory colonic hypersensitivity in rats. Gastroenterology 128:1996–2008. https://doi.org/10.1053/j.gastro.2005.03.082
Brooks L, Viardot A, Tsakmaki A et al (2017) Fermentable carbohydrate stimulates FFAR2-dependent colonic PYY cell expansion to increase satiety. Mol Metab 6:48–60. https://doi.org/10.1016/j.molmet.2016.10.011
Brown CT, Davis-Richardson AG, Giongo A et al (2011) Gut Microbiome Metagenomics Analysis suggests a functional model for the development of autoimmunity for type 1 diabetes. PLoS ONE 6:e25792. https://doi.org/10.1371/journal.pone.0025792
Çamcı G, Oğuz S (2016) Association between Parkinson’s Disease and Helicobacter Pylori. J Clin Neurol 12. https://doi.org/10.3988/jcn.2016.12.2.147. :147 – 50
Castillo RJ, Carlat DJ, Millon T, Millon CM, Meagher S, Grossman S, American Psychiatric Association (2007) Diagnostic and statistical manual of mental disorders. American Psychiatric Association Press, Washington, DC
Cattaneo A, Cattane N, Galluzzi S et al (2017) Association of brain amyloidosis with pro-inflammatory gut bacterial taxa and peripheral inflammation markers in cognitively impaired elderly. Neurobiol Aging 49:60–68. https://doi.org/10.1016/j.neurobiolaging.2016.08.019
Cersosimo MG, Raina GB, Pecci C et al (2013) Gastrointestinal manifestations in Parkinson’s disease: prevalence and occurrence before motor symptoms. J Neurol 260:1332–1338. https://doi.org/10.1007/s00415-012-6801-2
Chang C, Lin C, Lane HY (2020) D-glutamate and gut microbiota in Alzheimer’s Disease. Int J Mol Sci 21:2676. https://doi.org/10.3390/ijms21082676
Cherny I, Rockah L, Levy-Nissenbaum O, Gophna U, Ron EZ, Gazit E (2005) The formation of Escherichia coli curli amyloid fibrils is mediated by prion-like peptide repeats. J Mol Biol 352. https://doi.org/10.1016/j.jmb.2005.07.028. :245 – 52
Collins SM (2014) A role for the gut microbiota in IBS. Nat reviews Gastroenterol Hepatol 11:497–505. https://doi.org/10.1038/nrgastro.2014.40
Collins SM, Surette M, Bercik P (2012) The interplay between the intestinal microbiota and the brain. Nat Rev Microbiol 10:735–742. https://doi.org/10.1038/nrmicro2876
Cryan JF, Dinan TG (2012) Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci 13:701–712. https://doi.org/10.1038/nrn3346
Cryan JF, O’Mahony SM (2011) The microbiome-gut‐brain axis: from bowel to behavior. Neurogastroenterology & Motility. https://doi.org/10.1111/j.1365-2982.2010.01664.x. 23:187 – 92
Cussotto S, Sandhu KV, Dinan TG, Cryan JF (2018) The Neuroendocrinology of the Microbiota-Gut-Brain Axis: a behavioural perspective. Front Neuroendocrinol 51:80–101. https://doi.org/10.1016/j.yfrne.2018.04.002
De Kivit S, Tobin MC, Forsyth CB, Keshavarzian A, Landay AL (2014) Regulation of intestinal immune responses through TLR activation: implications for pro-and prebiotics. Front Immunol 5:60. https://doi.org/10.3389/fimmu.2014.00060
den Besten G, Bleeker A, Gerding A et al (2015) Short-chain fatty acids protect Against High-Fat Diet-Induced obesity via a PPARγ-Dependent switch from lipogenesis to Fat Oxidation. Diabetes 64:2398–2408. https://doi.org/10.2337/db14-1213
Desbonnet L, Garrett L, Clarke G, Kiely B, Cryan JF, Dinan TG (2010) Effects of the probiotic Bifidobacterium infantis in the maternal separation model of depression. Neuroscience 170:1179–1188. https://doi.org/10.1016/j.neuroscience.2010.08.005
Dinan TG, Cryan JF (2017a) Brain-gut-microbiota Axis and Mental Health. Psychosom Med 79:920–926. https://doi.org/10.1097/psy.0000000000000519
Dinan TG, Cryan JF (2017b) The Microbiome-Gut-Brain Axis in Health and Disease. Gastroenterol Clin 46:77–89. https://doi.org/10.1016/j.gtc.2016.09.007
Doifode T, Giridharan VV, Generoso JS et al (2021) The impact of the microbiota-gut-brain axis on Alzheimer’s disease pathophysiology. Pharmacol Res 164:105314. https://doi.org/10.1016/j.phrs.2020.105314
Dong S, Sun M, He C, Cheng H (2022) Brain-gut-microbiota axis in Parkinson’s disease: a historical review and future perspective. Brain Res Bull 183:84–93. https://doi.org/10.1016/j.brainresbull.2022.02.015
Dowlati Y, Herrmann N, Swardfager W, Liu H, Sham L, Reim EK, Lanctôt KL (2010) A meta-analysis of cytokines in major depression. Biological psychiatry 67 446 – 57. https://doi.org/10.1016/j.biopsych.2009.09.033
Du Y, Gao XR, Peng L, Ge JF (2020) Crosstalk between the microbiota-gut-brain axis and depression. Heliyon 6:e04097. https://doi.org/10.1016/j.heliyon.2020.e04097
Eisenhofer G, Åneman A, Friberg P et al (1997) Substantial production of dopamine in the human gastrointestinal tract. J Clin Endocrinol Metabolism 82:3864–3871. https://doi.org/10.1210/jcem.82.11.4339
Evrensel A, Ceylan ME (2015) The gut-brain Axis: the missing link in Depression. Clin Psychopharmacol Neurosci 13. https://doi.org/10.9758/cpn.2015.13.3.239. :239 – 44
Fasano A, Visanji NP, Liu LWC, Lang AE, Pfeiffer RF (2015) Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol 14. https://doi.org/10.1016/s1474-4422(15)00007-1. :625 – 39
Foster JA, McVey Neufeld KA (2013) Gut-brain axis: how the microbiome influences anxiety and depression. Trends Neurosci 36. https://doi.org/10.1016/j.tins.2013.01.005. :305 – 12
Friedland RP (2015) Mechanisms of molecular mimicry involving the microbiota in neurodegeneration. J Alzheimers Dis 45. https://doi.org/10.3233/jad-142841. :349 – 62
Friedland RP, Chapman MR (2017) The role of microbial amyloid in neurodegeneration. PLoS Pathog 13:e1006654. https://doi.org/10.1371/journal.ppat.1006654
Frost G, Sleeth ML, Sahuri-Arisoylu M et al (2014) The short-chain fatty acid acetate reduces appetite via a central homeostatic mechanism. Nat Commun 5:1–11. https://doi.org/10.1038/ncomms4611
Furness JB (2012) The enteric nervous system and neurogastroenterology. Nat reviews Gastroenterol Hepatol. https://doi.org/10.1038/nrgastro.2012.32. 9:286 – 94
Fuster MA, Llorens MM, Hernández F, Avila J (2013) Role of neuroinflammation in adult neurogenesis and Alzheimer disease: therapeutic approaches. Mediators Inflamm 2013:260925. https://doi.org/10.1155/2013/260925
Galloway S, Takechi R, Nesbit M et al (2019) The differential effects of fatty acids on enterocytic abundance of amyloid-beta. Lipids Health Dis 18:1–6. https://doi.org/10.1186/s12944-019-1162-9
García-Peña C, Álvarez-Cisneros T, Quiroz-Baez R, Friedland RP (2017) Microbiota and aging. A review and commentary. Arch Med Res. https://doi.org/10.1016/j.arcmed.2017.11.005. 48:681 – 89
Gareau MG, Wine E, Rodrigues DM et al (2011) Bacterial infection causes stress-induced memory dysfunction in mice. Gut 60:307–317. https://doi.org/10.1136/gut.2009.202515
Gargari G, Taverniti V, Gardana C et al (2018) Fecal Clostridiales distribution and short-chain fatty acids reflect bowel habits in irritable bowel syndrome. Environ Microbiol 20:3201–3213. https://doi.org/10.1111/1462-2920.14271
Ge X, Pan J, Liu Y, Wang H, Zhou W, Wang X (2018) Intestinal crosstalk between microbiota and serotonin and its impact on gut motility. Curr Pharm Biotechnol 19:190–195. https://doi.org/10.2174/1389201019666180528094202
Gibson GR, Hutkins R, Sanders ME et al (2017) Expert consensus document: the International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat reviews Gastroenterol Hepatol 14(8):491–502. https://doi.org/10.1038/nrgastro.2017.75
Gorecki AM, Preskey L, Bakeberg MC et al (2019) Altered gut microbiome in Parkinson’s Disease and the influence of Lipopolysaccharide in a human α-Synuclein Over-Expressing Mouse Model. Front NeuroSci 13. https://doi.org/10.3389/fnins.2019.00839
Grad S, Dumitrascu DL (2020) Irritable bowel syndrome subtypes: New Names for Old Medical Conditions. Dig Dis 38:122–127. https://doi.org/10.1159/000505287
Gulaj E, Pawlak K, Bien B, Pawlak D (2010) Kynurenine and its metabolites in Alzheimer’s disease patients. Adv Med Sci 55:204–211. https://doi.org/10.2478/v10039-010-0023-6
Gururajan A, Clarke G, Dinan TG, Cryan JF (2016) Molecular biomarkers of depression. Neurosci Biobehavioral Reviews. https://doi.org/10.1016/j.neubiorev.2016.02.011. 64:101 – 33
Hadjivasilis A, Tsioutis C, Michalinos A, Ntourakis D, Christodoulou DK, Agouridis AP (2019) New insights into irritable bowel syndrome: from pathophysiology to treatment. Ann Gastroenterol 32:554–564. https://doi.org/10.20524/aog.2019.0428
Harach T, Marungruang N, Duthilleul N et al (2017) Reduction of Abeta amyloid pathology in APPPS1 transgenic mice in the absence of gut microbiota. Sci Rep 7:1–15. https://doi.org/10.1038/srep41802
Hardy J, Allsop D (1991) Amyloid deposition as the central event in the aetiology of Alzheimer’s disease. Trends Pharmacol Sci 12:383–388. https://doi.org/10.1016/0165-6147(91)90609-v
He N, Langley J, Huddleston DE et al (2020) Increased iron-deposition in lateral-ventral substantia nigra pars compacta: a promising neuroimaging marker for Parkinson’s disease. NeuroImage: Clin 28:102391. https://doi.org/10.1016/j.nicl.2020.102391
Heintz-Buschart A, Pandey U, Wicke T et al (2018) The nasal and gut microbiome in Parkinson’s disease and idiopathic rapid eye movement sleep behavior disorder. Mov Disord 33:88–98. https://doi.org/10.1002/mds.27105
Helton SG, Lohoff FW (2015) Serotonin pathway polymorphisms and the treatment of major depressive disorder and anxiety disorders. Pharmacogenomics 16:541–553. https://doi.org/10.2217/pgs.15.15
Heneka MT, Carson MJ, El Khoury J et al (2015) Neuroinflammation in Alzheimer’s disease. Lancet Neurol 14:388–405. https://doi.org/10.1016/s1474-4422(15)70016-5
Hill C, Guarner F, Reid G et al (2014) Expert consensus document: the International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nature reviews Gastroenterology & hepatology. http://www.nature.com/doifinder/https://doi.org/10.1038/nrgastro.2014.66
Hsieh TH, Kuo CW, Hsieh KH et al (2020) Probiotics alleviate the progressive deterioration of Motor Functions in a mouse model of Parkinson’s Disease. Brain Sci 10:206. https://doi.org/10.3390/brainsci10040206
Jameson KG, Hsiao EY (2018) Linking the gut microbiota to a brain neurotransmitter. Trends Neurosci 41:413–414. https://doi.org/10.1016/j.tins.2018.04.001
Jeffery IB, O’toole PW, Öhman L et al (2012a) An irritable bowel syndrome subtype defined by species-specific alterations in faecal microbiota. Gut 61:997–1006. https://doi.org/10.1136/gutjnl-2011-301501
Jeffery IB, Quigley EMM, Öhman L, Simrén M, O’Toole PW (2012b) The microbiota link to irritable bowel syndrome: an emerging story. Gut Microbes 3:572–576. https://doi.org/10.4161/gmic.21772
Jiang H, Ling Z, Zhang Y et al (2015) Altered fecal microbiota composition in patients with major depressive disorder. Brain Behav Immun 48:186–194. https://doi.org/10.1016/j.bbi.2015.03.016
Kamath PS, Phillips SF, Zinsmeister AR (1988) Short-chain fatty acids stimulate ileal motility in humans. Gastroenterology 95. https://doi.org/10.1016/s0016-5085(88)80068-4. :1496 – 502
Karabiyik C, Lee MJ, Rubinsztein DC (2017) Autophagy impairment in Parkinson’s disease. Essays Biochem 61:711–720. https://doi.org/10.1042/ebc20170023
Kelly JR, Kennedy PJ, Cryan JF et al (2015) Breaking down the barriers: the gut microbiome, intestinal permeability and stress-related psychiatric disorders. Front Cell Neurosci 392. https://doi.org/10.3389/fncel.2015.00392
Kelly JR, Borre Y, O’Brien C et al (2016) Transferring the blues: depression-associated gut microbiota induces neurobehavioural changes in the rat. J Psychiatr Res 82. https://doi.org/10.1016/j.jpsychires.2016.07.019. :109 – 18
Kelly JR, Allen AP, Temko A et al (2017) Lost in translation? The potential psychobiotic Lactobacillus rhamnosus (JB-1) fails to modulate stress or cognitive performance in healthy male subjects. Brain Behav Immun 61:50–59. https://doi.org/10.1016/j.bbi.2016.11.018
Kennedy PJ, Cryan JF, Dinan TG, Clarke G (2017) Kynurenine pathway metabolism and the microbiota-gut-brain axis. Neuropharmacology 112:399–412. https://doi.org/10.1016/j.neuropharm.2016.07.002
Kessler RC, Bromet EJ (2013) The epidemiology of depression across cultures. Annu Rev Public Health 34:119. https://doi.org/10.1146/annurev-publhealth-031912-114409
Kincaid HJ, Nagpal R, Yadav H (2021) Diet-Microbiota-Brain Axis in Alzheimer’s Disease. Ann Nutr Metab 77 Suppl 221–27. https://doi.org/10.1159/000515700
Klein C, Westenberger A (2012) Genetics of Parkinson’s disease. Cold Spring Harb Perspect Med 2:a008888. https://doi.org/10.1101/cshperspect.a008888
Koloski NA, Jones M, Talley NJ (2016) Evidence that independent gut-to-brain and brain-to-gut pathways operate in the irritable bowel syndrome and functional dyspepsia: a 1-year population-based prospective study. Aliment Pharmacol Ther 44:592–600. https://doi.org/10.1111/apt.13738
König J, Brummer RJ (2014) Alteration of the intestinal microbiota as a cause of and a potential therapeutic option in irritable bowel syndrome. Benef Microbes 5:247–261. https://doi.org/10.3920/bm2013.0033
Kowalski K, Mulak A (2019) Brain-gut-microbiota axis in Alzheimer’s disease. J Neurogastroenterol Motil 25:48. https://doi.org/10.5056/jnm18087
Kujawska M, Jodynis-Liebert J (2018) What is the evidence that Parkinson’s Disease is a prion disorder, which originates in the gut? Int J Mol Sci 19. https://doi.org/10.3390/ijms19113573
Kundu P, Blacher E, Elinav E, Pettersson S (2017) Our gut Microbiome: the evolving inner self. Cell 171:1481–1493. https://doi.org/10.1016/j.cell.2017.11.024
Kusich TH (2018) A functional application of the Gut-Brain Axis: a proposed Nutrition intervention for the treatment of Depression. https://doi.org/10.15760/honors.589
Labus JS, Hollister EB, Jacobs J et al (2017) Differences in gut microbial composition correlate with regional brain volumes in irritable bowel syndrome. Microbiome 5:49. https://doi.org/10.1186/s40168-017-0260-z
Lach G, Schellekens H, Dinan TG, Cryan JF (2018) Anxiety, Depression, and the Microbiome: a role for gut peptides. Neurotherapeutics 15:36–59. https://doi.org/10.1007/s13311-017-0585-0
Larraufie P, Martin-Gallausiaux C, Lapaque N, Dore J, Gribble FM, Reimann F, Blottiere HM (2018) SCFAs strongly stimulate PYY production in human enteroendocrine cells. Sci Rep 8:74. https://doi.org/10.1038/s41598-017-18259-0
Latorre R, Sternini C, De Giorgio R, Greenwood-Van B, Meerveld (2016) Enteroendocrine cells: a review of their role in brain–gut communication. Neurogastroenterology & Motility 28(5):620–630. https://doi.org/10.1111/nmo.12754
Le Page A, Dupuis G, Frost EH, Larbi A, Pawelec G, Witkowski JM, Fulop T (2018) Role of the peripheral innate immune system in the development of Alzheimer’s disease. Exp Gerontol 107:59–66. https://doi.org/10.1016/j.exger.2017.12.019
Ledford H (2014) Medical research: if depression were cancer. Nature 515:182–184. https://doi.org/10.1038/515182a
Lee SE, Lee Y, Lee GH (2019) The regulation of glutamic acid decarboxylases in GABA neurotransmission in the brain. Arch Pharm Res 42:1031–1039. https://doi.org/10.1007/s12272-019-01196-z
Liang S, Wang T, Hu X, Luo J, Li W, Wu X, Duan Y, Jin F (2015) Administration of Lactobacillus helveticus NS8 improves behavioral, cognitive, and biochemical aberrations caused by chronic restraint stress. Neuroscience 310:561–577. https://doi.org/10.1016/j.neuroscience.2015.09.033
Long-Smith C, O’Riordan KJ, Clarke G, Stanton C, Dinan TG, Cryan JF (2020) Microbiota-Gut-Brain Axis: New Therapeutic Opportunities. Annu Rev Pharmacol Toxicol 60:477–502. https://doi.org/10.1146/annurev-pharmtox-010919-023628
Lu Y, Fan C, Li P, Lu Y, Chang X, Qi K (2016) Short chain fatty acids prevent high-fat-diet-induced obesity in mice by regulating G protein-coupled receptors and gut microbiota. Sci Rep 6:37589. https://doi.org/10.1038/srep37589
Luk KC (2019) Oxidative stress and α-synuclein conspire in vulnerable neurons to promote Parkinson’s disease progression. J Clin Investig 129:3530–3531. https://doi.org/10.1172/jci130351
Lyte M (2011) Probiotics function mechanistically as delivery vehicles for neuroactive compounds: microbial endocrinology in the design and use of probiotics. BioEssays 33:574–581. https://doi.org/10.1002/bies.201100024
Lyte M (2013) Microbial endocrinology in the microbiome-gut-brain axis: how bacterial production and utilization of neurochemicals influence behavior. PLoS Pathog 9:e1003726. https://doi.org/10.1371/journal.ppat.1003726
Maes M, Kubera M, Leunis JC, Berk M (2012) Increased IgA and IgM responses against gut commensals in chronic depression: further evidence for increased bacterial translocation or leaky gut. J Affect Disord 141:55–62
Margolis KG, Cryan JF, Mayer EA (2021) The Microbiota-Gut-Brain Axis: from motility to Mood. Gastroenterology 160:1486–1501. https://doi.org/10.1053/j.gastro.2020.10.066
Masato A, Plotegher N, Boassa D, Bubacco L (2019) Impaired dopamine metabolism in Parkinson’s disease pathogenesis. Mol neurodegeneration 14:1–21. https://doi.org/10.1186/s13024-019-0332-6
Mass M, Kubera M, Leunis JC (2008) The gut-brain barrier in major depression: intestinal mucosal dysfunction with an increased translocation of LPS from gram negative enterobacteria (leaky gut) plays a role in the inflammatory pathophysiology of depression. Neuroendocrinol Lett 29:117–124
Mayer EA (2011) Gut feelings: the emerging biology of gut-brain communication. Nat Rev Neurosci 12:453–466. https://doi.org/10.1038/nrn3071
Mayer EA, Knight R, Mazmanian SK, Cryan JF, Tillisch K (2014) Gut microbes and the brain: paradigm shift in neuroscience. J Neurosci 34:15490–15496. https://doi.org/10.1523/jneurosci.3299-14.2014
McCutcheon RA, Abi-Dargham A, Howes OD (2019) Schizophrenia, dopamine and the striatum: from biology to symptoms. Trends Neurosci 42. https://doi.org/10.1016/j.tins.2018.12.004. :205 – 20
McGee DJ, Lu XH, Disbrow EA (2018) Stomaching the possibility of a pathogenic role for Helicobacter pylori in Parkinson’s Disease. J Parkinsons Dis 8:367–374. https://doi.org/10.3233/JPD-181327
Megur A, Baltriukienė D, Bukelskienė V et al (2021) The microbiota–gut–brain Axis and Alzheimer’s Disease: Neuroinflammation is to blame? Nutrients 13:37. https://doi.org/10.3390/nu13010037
Mehrabadi S, Sadr SS (2020) Assessment of Probiotics mixture on memory function, inflammation markers, and oxidative stress in an Alzheimer’s Disease Model of rats. Iran Biomed J 24:220–228. https://doi.org/10.29252/ibj.24.4.220
Menozzi E, Macnaughtan J, Schapira AH (2021) The gut-brain axis and Parkinson disease: clinical and pathogenetic relevance. Ann Med 53:611–625. https://doi.org/10.1080/07853890.2021.1890330
Messaoudi M, Lalonde R, Violle N et al (2011) Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br J Nutr 105:755–764. https://doi.org/10.1017/S0007114510004319
Mika A, Day HE, Martinez A et al (2017) Early life diets with prebiotics and bioactive milk fractions attenuate the impact of stress on learned helplessness behaviours and alter gene expression within neural circuits important for stress resistance. Eur J Neurosci 45:342–357. https://doi.org/10.1111/ejn.13444
Minter MR, Zhang C, Leone V et al (2016) Antibiotic-induced perturbations in gut microbial diversity influences neuro-inflammation and amyloidosis in a murine model of Alzheimer’s disease. Sci Rep 6:1–12. https://doi.org/10.1038/srep30028
Minter MR, Hinterleitner R, Meisel M et al (2017) Antibiotic-induced perturbations in microbial diversity during post-natal development alters amyloid pathology in an aged APPSWE/PS1∆E9 murine model of Alzheimer’s disease. Sci Rep 7:1–18. https://doi.org/10.1038/s41598-017-11047-w
Mittal R, Debs LH, Patel AP et al (2017) Neurotransmitters: the critical modulators regulating gut–brain Axis. J Cell Physiol 232:2359–2372. https://doi.org/10.1002/jcp.25518
Moloney RD, Johnson AC, O’mahony SM et al (2016) Stress and the Microbiota-Gut-Brain Axis in Visceral Pain: relevance to irritable bowel syndrome. CNS Neurosci Ther 22:102–117. https://doi.org/10.1111/cns.12490
Montiel-Castro AJ, González-Cervantes RM, Bravo-Ruiseco G et al (2013) The microbiota-gut-brain axis: neurobehavioral correlates, health and sociality. Front Integr Nuerosci 7:70. https://doi.org/10.3389/fnint.2013.00070
Mou Z, Yuan YH, Zhang Z et al (2020) Endoplasmic reticulum stress, an important factor in the development of Parkinson’s disease. Toxicol Lett 324:20–29. https://doi.org/10.1016/j.toxlet.2020.01.019
Naomi R, Hashim E, Fezah O et al (2022) Probiotics for Alzheimer & rsquo;s Disease: A Systematic Review. In Nutrients. https://doi.org/10.3390/nu14010020
Naseribafrouei A, Hestad K, Avershina E et al (2014) Correlation between the human fecal microbiota and depression. Neurogastroenterology & Motility 26:1155–1162. https://doi.org/10.1111/nmo.12378
Nuzum ND, Loughman A, Szymlek-Gay EA et al (2020) Gut microbiota differences between healthy older adults and individuals with Parkinson’s disease: a systematic review’. Neurosci Biobehavioral Reviews 112:227–241. https://doi.org/10.1016/j.neubiorev.2020.02.003
O’Brien SM, Scully P, Scott LV et al (2006) Cytokine profiles in bipolar affective disorder: focus on acutely ill patients. J Affect Disord 90. https://doi.org/10.1016/j.jad.2005.11.015. :263 – 67
Öhman L, Simren M (2007) New insights into the pathogenesis and pathophysiology of irritable bowel syndrome. Digestive and liver disease 39:201 – 15. https://doi.org/10.1016/j.dld.2006.10.014
Oświęcimska J, Szymlak A, Roczniak W et al (2017) New insights into the pathogenesis and treatment of irritable bowel syndrome. Adv Med Sci 62:17–30. https://doi.org/10.1016/j.advms.2016.11.001
Parkes GC, Brostoff J, Whelan K et al (2008) Gastrointestinal microbiota in irritable bowel syndrome: their role in its pathogenesis and treatment. Official J Am Coll Gastroenterology| ACG 103:1557–1567. https://doi.org/10.1111/j.1572-0241.2008.01869.x
Penke B, Bogár F, Fülöp L (2017) β-Amyloid and the Pathomechanisms of Alzheimer’s Disease: a Comprehensive View’. Molecules 22. https://doi.org/10.3390/molecules22101692
Petra AI, Panagiotidou S, Hatziagelaki E et al (2015) Gut-Microbiota-Brain Axis and its Effect on Neuropsychiatric Disorders with suspected Immune Dysregulation. Clin Ther 37:984–995. https://doi.org/10.1016/j.clinthera.2015.04.002
Pfeiffer RF (2011) Gastrointestinal dysfunction in Parkinson’s disease. Parkinsonism Relat Disord 17:10–15. https://doi.org/10.1016/j.parkreldis.2010.08.003
Picciotto MR, Higley MJ, Mineur YS (2012) Acetylcholine as a neuromodulator: cholinergic signaling shapes nervous system function and behavior. Neuron 76:116–129. https://doi.org/10.1016/j.neuron.2012.08.036
Pistollato F, Sumalla Cano S, Elio I et al (2016) Role of gut microbiota and nutrients in amyloid formation and pathogenesis of Alzheimer disease. Nutr Rev 74:624–634. https://doi.org/10.1093/nutrit/nuw023
Pittayanon R, Lau JT, Yuan Y et al (2019) Gut microbiota in patients with irritable bowel Syndrome—A systematic review. Gastroenterology 157:97–108. https://doi.org/10.1053/j.gastro.2019.03.049
Popa TA, Ladea M (2012) Nutrition and depression at the forefront of progress. J Med Life 5:414
Posserud I, Ersryd A, Simrén M (2006) Functional findings in irritable bowel syndrome. World J gastroenterology: WJG 12:2830. https://doi.org/10.3748/wjg.v12.i18.2830
Potter H, Granic A, Caneus J (2016) Role of trisomy 21 mosaicism in sporadic and familial Alzheimer’s disease. Curr Alzheimer Res 13:7–17. https://doi.org/10.2174/156720501301151207100616
Queipo-Ortuño MI, Seoanen LM, Murri M et al (2013) Gut microbiota composition in male rat models under different nutritional status and physical activity and its association with serum leptin and ghrelin levels. PLoS ONE 8:e65465. https://doi.org/10.1371/journal.pone.0065465
Quigley EM (2017) Microbiota-brain-gut axis and neurodegenerative diseases. Curr Neurol Neurosci Rep 17:1–9. https://doi.org/10.1007/s11910-017-0802-6
Rajilić–Stojanović M, Biagi E, Heilig HG et al (2011) Global and deep molecular analysis of microbiota signatures in fecal samples from patients with irritable bowel syndrome. Gastroenterology 141:1792–1801. https://doi.org/10.1053/j.gastro.2011.07.043
Reichmann H, Schneider C, Löhle M (2009) Non-motor features of Parkinson’s disease: depression and dementia. Parkinsonism Relat Disord 15:S87–S92. https://doi.org/10.1016/S1353-8020(09)70789-8
Rizvi SJ, Pizzagalli DA, Sproule BA et al (2016) Assessing anhedonia in depression: potentials and pitfalls. Neurosci Biobehavioral Reviews 65:21–35. https://doi.org/10.1016/j.neubiorev.2016.03.004
Roesler R, Schwartsmann G (2012) Gastrin-releasing peptide receptors in the central nervous system: role in brain function and as a drug target. Front Endocrinol 3:159. https://doi.org/10.3389/fendo.2012.0015
Ruddick JP, Evans AK, Nutt DJ et al (2006) Tryptophan metabolism in the central nervous system: medical implications. Expert Rev Mol Med 8:1–27. https://doi.org/10.1017/S1462399406000068
Salminen S, Bouley C, Boutron MC et al (1998) Functional food science and gastrointestinal physiology and function. Br J Nutr 80:S147–S71. https://doi.org/10.1079/BJN19980108
Salonen A, de Vos WM, Palva A (2010) Gastrointestinal microbiota in irritable bowel syndrome: present state and perspectives. Microbiology 156:3205–3215. https://doi.org/10.1099/mic.0.043257-0
Scheperjans F, Aho V, Pereira PA et al (2015) Gut microbiota are related to Parkinson’s disease and clinical phenotype. Mov Disord 30:350–358. https://doi.org/10.1002/mds.26069
Schneider RB, Iourinets J, Richard IH (2017) Parkinson’s disease psychosis: presentation, diagnosis and management. Neurodegener Dis Manag 7:365–376. https://doi.org/10.2217/nmt-2017-0028
Sharma MK, Jalewa J, Hölscher C (2014) Neuroprotective and anti-apoptotic effects of liraglutide on SH‐SY 5Y cells exposed to methylglyoxal stress. J Neurochem 128:459–471. https://doi.org/10.1111/jnc.12469
Singhrao SK, Harding A, Poole S et al (2015) <i> Porphyromonas gingivalis</i> Periodontal Infection and Its Putative Links with Alzheimer’s Disease. Mediat Inflamm 2015:137357. https://doi.org/10.1155/2015/137357
Sochocka M, Diniz BS, Leszek J (2017) Inflammatory response in the CNS: friend or foe? Mol Neurobiol 54:8071–8089. https://doi.org/10.1007/s12035-016-0297-1
Sochocka M, Donskow-Łysoniewska K, Diniz BS et al (2019) The gut microbiome alterations and inflammation-driven pathogenesis of Alzheimer’s Disease-a critical review. Mol Neurobiol 56:1841–1851. https://doi.org/10.1007/s12035-018-1188-4
Spacova I, Dodiya HB, Happel AU et al (2020) Future of probiotics and prebiotics and the implications for early career researchers. Front Microbiol 11:1400. https://doi.org/10.3389/fmicb.2020.01400
Strawbridge R, Young AH, Cleare AJ (2018) Biomarkers for depression: recent insights, current challenges and future prospects. Focus 16:194–209. https://doi.org/10.1176/appi.focus.16206
Su WJ, ZhangY, Chen Y et al (2017) NLRP3 gene knockout blocks NF-κB and MAPK signaling pathway in CUMS-induced depression mouse model. Behav Brain Res 322:1–8. https://doi.org/10.1016/j.bbr.2017.01.018
Sun Q, Jia Q, Song L, Duan L (2019) Alterations in fecal short-chain fatty acids in patients with irritable bowel syndrome: a systematic review and meta-analysis. Medicine 98(7). https://doi.org/10.1097%2FMD.0000000000014513
Sundin J, Rangel I, Fuentes S et al (2015) Altered faecal and mucosal microbial composition in post-infectious irritable bowel syndrome patients correlates with mucosal lymphocyte phenotypes and psychological distress. Aliment Pharmacol Ther 41:342–351. https://doi.org/10.1111/apt.13055
Tap J, Derrien M, Törnblom H et al (2017) Identification of an intestinal microbiota signature associated with severity of irritable bowel syndrome. Gastroenterology. 152:111 – 23. e8 https://doi.org/10.1053/j.gastro.2016.09.049
Testerman TL, Morris J (2014) Beyond the stomach: an updated view of Helicobacter pylori pathogenesis, diagnosis, and treatment. World J Gastroenterol 20:12781–12808. https://doi.org/10.3748/wjg.v20.i36.12781
Tolhurst G, Heffron H, Lam YS et al (2012) Short-chain fatty acids stimulate glucagon-like peptide-1 secretion via the G-protein-coupled receptor FFAR2. Diabetes 61:364–371. https://doi.org/10.2337/db11-1019
Tsigos C, Chrousos GP (2002) Hypothalamic-pituitary-adrenal axis, neuroendocrine factors and stress. J Psychosom Res 53. https://doi.org/10.1016/S0022-3999(02)00429-4. :865 – 71
Unger MM, Spiegel J, Dillmann KU, Fa beta bender K, Schwiertz A, Schafer KH et al (2016) Short chain fatty acids and gut microbiota differ between patients with Parkinson’s disease and age-matched controls. Parkinsonism Relat Disord 32:66–72. https://doi.org/10.1016/j.parkreldis.2016.08.019
Van Kessel SP, El Aidy S (2019) Contributions of gut Bacteria and Diet to Drug Pharmacokinetics in the treatment of Parkinson’s disease. Front Neurol 10. https://doi.org/10.3389/fneur.2019.01087
Vaughan CJ, Aherne AM, Lane E et al (2000) Identification and regional distribution of the dopamine D1A receptor in the gastrointestinal tract. Am J Physiology-Regulatory Integr Comp Physiol 279:R599–R609. https://doi.org/10.1152/ajpregu.2000.279.2.R599
Vogt NM, Kerby RL, Dill-McFarland KA et al (2017) Gut microbiome alterations in Alzheimer’s disease. Sci Rep 7:13537. https://doi.org/10.1038/s41598-017-13601-y
Wachsmuth HR, Weninger SN, Duca FA (2022) Role of the gut-brain axis in energy and glucose metabolism. Exp Mol Med 54:377–392. https://doi.org/10.1038/s12276-021-00677-w
Wallace CJ, Milev R (2017) The effects of probiotics on depressive symptoms in humans: a systematic review. Ann Gen Psychiatry 16:14. https://doi.org/10.1186/s12991-017-0138-2
Wang Q, Luo Y, Ray Chaudhuri K et al (2021) The role of gut dysbiosis in Parkinson’s disease: mechanistic insights and therapeutic options. Brain 144:2571–2593. https://doi.org/10.1093/brain/awab156
Westfall S, Lomis N, Kahouli I et al (2017) Microbiome, probiotics and neurodegenerative diseases: deciphering the gut brain axis. Cell Mol Life Sci 74:3769–3787. https://doi.org/10.1007/s00018-017-2550-9
Widner B, Leblhuber F, Walli J et al (2000) Tryptophan degradation and immune activation in Alzheimer’s disease. J Neural Transm (Vienna) 107:343–353. https://doi.org/10.1007/s007020050029
Yang X, Qian Y, Xu S et al (2018) Longitudinal analysis of fecal microbiome and pathologic processes in a Rotenone Induced mice Model of Parkinson’s Disease. Front Aging Neurosci 9. https://doi.org/10.3389/fnagi.2017.00441
Yang C, Fang X, Zhan G et al (2019) Key role of gut microbiota in anhedonia-like phenotype in rodents with neuropathic pain. Translational psychiatry 9:1–11. https://doi.org/10.1038/s41398-019-0379-8
Yang J, Zheng P, Li Y (2020) Landscapes of bacterial and metabolic signatures and their interaction in major depressive disorders. Sci Adv 6:eaba8555. https://doi.org/10.1126/sciadv.aba8555
Yu M, Jia H, Zhou C et al (2017) Variations in gut microbiota and fecal metabolic phenotype associated with depression by 16S rRNA gene sequencing and LC/MS-based metabolomics. J Pharm Biomed Anal 138:231–239. https://doi.org/10.1016/j.jpba.2017.02.008
Zhao Y, Lukiw WJ (2015) Microbiome-generated amyloid and potential impact on amyloidogenesis in Alzheimer’s disease (AD). J Nat Sci 1. https://doi.org/10.3389/fnagi.2015.00009
Zhao Y, Dua P, Lukiw WJ (2015) Microbial sources of amyloid and relevance to amyloidogenesis and Alzheimer’s disease (AD). J Alzheimer’s disease Parkinsonism 5:177. https://doi.org/10.4172/2161-0460.1000177
Zheng P, Zeng B, Zhou C et al (2016) Gut microbiome remodeling induces depressive-like behaviors through a pathway mediated by the host’s metabolism. Mol Psychiatry 21:786–796. https://doi.org/10.1038/mp.2016.44
Acknowledgements
We acknowledge the Research Institute for Gastroenterology and Liver Diseases, Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Funding
No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Writing - original draft preparation: Kimia Basiji, and Azade Aghamohammadi Sendani; Writing - review and editing: Maryam Farmani, and Nesa Kazemifard; Drawing of figure: Kimia Basiji; Resources: Hamid Asadzadeh Aghdaei, and Ensieh Lotfali; Supervision: Shaghayegh Baradaran Ghavami, and Amir Sadeghi.
Corresponding author
Ethics declarations
Non-financial interests
None.
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Data transparency
Not applicable
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Basiji, K., Sendani, A.A., Ghavami, S.B. et al. The critical role of gut-brain axis microbiome in mental disorders. Metab Brain Dis 38, 2547–2561 (2023). https://doi.org/10.1007/s11011-023-01248-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-023-01248-w