Abstract
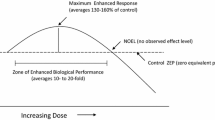
In order to survive living organisms have developed multiple mechanisms to deal with tough environmental conditions. Hormesis is defined as a process in which exposure to a low dose of a chemical agent or environmental factor that is damaging at higher doses induces an adaptive beneficial effect on the cell or organism. In this paper, we examine several ideas that might be taken into consideration before using hormesis as a therapeutic tool to improve health and life span, and hopefully will open the discussion for new and interesting debates regard hormesis. The first one is to understand that the same stressor or inductor can activate different pathways in a parallel or dual response, which might lead to diverse outcomes. Another idea is related to the mechanisms involved in activating Nrf2, which might be different and have diverse hormetic effects.
Last, we discuss mild oxidative stress in association to low-grade chronic inflammation as a stimulating avenue to be explored and the unexpected effects proposed by the obesity paradox theory. All the previous might help to clarify the reasons why centenarians are able to reach the extreme limits of human life span, which could probably be related to the way they deal with homeostasis maintenance, providing an opportunity for hormesis to intervene significantly.

Similar content being viewed by others
Abbreviations
- OCH :
-
Oxidative conditioning hormesis
- H 2 O 2 :
-
Hydrogen peroxide
- Nrf2 :
-
Nuclear factor erythroid-derived 2-like 2
- Keap1 :
-
Kelch-like ECH-associated protein 1
- ROS/RNS :
-
Reactive oxygen species/Reactive nitrogen species
- CAT :
-
Catalase
- SOD :
-
superoxide dismutase
- GSH :
-
Reduced glutathione
- PKC :
-
Protein kinase C
- AOS :
-
Alginate oligosachharide
- AGEs :
-
advanced glycation end products
- tBHQ :
-
tert-butylhydroquinone
- TBQ :
-
tertbutylbenzoquinone
- MPP+ :
-
1-methyl-4-phenylpyridinium
References
Abiko Y, Miura T, Phuc BH, Shinkai Y, Kumagai Y (2011) Participation of covalent modification of Keap1 in the activation of Nrf2 by tert-butylbenzoquinone, an electrophilic metabolite of butylated hydroxyanisole. Toxicol Appl Pharmacol 255:32–39
Alarcón-Aguilar A, Luna-López A, Ventura-Gallegos JL, Lazzarini R, Galván-Arzate S, González-Puertos VY, Morán J, Santamaría A, Königsberg M (2014) Primary cultured astrocytes from old rats are capable to activate the Nrf2 response against MPP + toxicity after tBHQ pretreatment. Neurobiol Aging 35:1901–1912
Barbieri M, Boccardi V, Papa M, Paolisso G (2009) Metabolic journey to healthy longevity. Horm Res 71:24–27
Bayod S, Del Valle J, Lalanza JF, Sanchez-Roige S, de Luxán-Delgado B, Coto-Montes A, Canudas AM, Camins A, Escorihuela RM, Pallàs M (2012) Long-term physical exercise induces changes in sirtuin 1 pathway and oxidative parameters in adult rat tissues. Exp Gerontol 47:925–935
Beddhu S (2004) The body mass index paradox and an obesity, inflammation, and atherosclerosis syndrome in chronic kidney disease. Semin Dial 17:229–232
Bluher M (2012) Are there still healthy obese patients? Curr Opin Endocrinol Diabetes Obes 19:341–346
Buelna-Chontal M, Guevara-Chávez JG, Silva-Palacios A, Medina-Campos ON, Pedraza-Chaverri J, Zazueta C (2014) Nrf2-regulated antioxidant response is activated by protein kinase C in postconditioned rat hearts. Free Radic Biol Med [Epub ahead of print]
Calabrese EJ (2008) Converging concepts: adaptive response, preconditioning, and the Yerkes–Dodson law are manifestations of hormesis. J Ageing Res Rev 7:8–20
Calabrese EJ, Baldwin LA (2003) Hormesis at the National Toxicology Program (NTP): evidence of hormetic dose responses in NTP dose-range studies. Nonlinear Biol Toxicol Med 1:455–467
Calabrese V, Cornelius C, Mancuso C, Lentile R, Stella AM, Butterfield DA (2010) Redox homeostasis and cellular stress response in aging and neurodegeneration. Methods Mol Biol 610:285–308
Choi VW, Cheung AL, Cheng SH, Yu KN (2012) Hormetic effect induced by alpha-particle-induced stress communicated in vivo between zebrafish embryos. Environ Sci Technol 46:11678–11683
Correa F, Buelna-Chontal M, Hernández-Reséndiz S, García-Niño WJ, Roldán F, Soto V, Silva-Palacios A, Amador A, Pedraza-Chaverrí J, Tapia E, Zazueta C (2013) Curcumin maintains cardiac and mitochondrial function in chronic kidney disease. Free Radic Biol Med 61C:119–129
Cui H, Kong Y, Zhang H (2011) Oxidative stress, mitochondrial dysfunction, and aging. J Signal Transduct 2012:646354
de Magalhaes JP, Curado J, Church GM (2009) Meta-analysis of age-related gene expression profiles identifies common signatures of aging. Bioinformatics 25:875–881
Demirovic D, Rattan SI (2011) Curcumin induces stress response and hermetically modulates wound healing ability of human skin fibroblasts undergoing ageing in vitro. Biogerontology 12:437–444
Epel ES, Lithgow GJ (2014) Stress biology and aging mechanisms: toward understanding the deep connection between adaptation to stress and longevity. J Gerontol A Biol Sci Med Sci 69:S10–S16
Erlank H, Elmann A, Kohen R, Kanner J (2011) Polyphenols activate Nrf2 in astrocytes via H2O2, semiquinones, and quinones. Free Radic Biol Med 51:2319–2327
Flegal KM, Kalantar-Zadeh K (2013) Overweight, mortality and survival. Obesity 21:1744–1745
Hayflick L (1998) How and why we age. Exp Gerontol 33:639–653
García-Niño WR, Pedraza-Chaverrí J (2014) Protective effect of curcumin against heavy metals-induced liver damage. Food Chem Toxicol 69C:182–201
Gopalakrishna R, Jaken S (2000) Protein kinase C signaling and oxidative stress. Free Radic Biol Med 28:1349–1361
Gopalakrishna R, McNeill TH, Elhiani AA, Gundimeda U (2013) Methods for studying oxidative regulation of protein kinase C. Meth Enzymol 528:79–98
Grynkiewicz G, Ślifirski P (2012) Curcumin and curcuminoids in quest for medicinal status. Acta Biochem Pol 59:201–212
Hashmi MZ, Khan KY, Hu J, Naveedullah, Su X, Abbas G, Yu C, Shen C (2014) Hormetic effects of noncoplanar PCB exposed to human lung fibroblast cells (HELF) and possible role of oxidative stress. Environ Toxicol [Epub ahead of print]
Hayden A, Douglas J, Sommerlad M, Andrews L, Gould K, Hussain S, Thomas GJ, Packham G, Crabb SJ (2014) The Nrf2 transcription factor contributes to resistance to cisplatin in bladder cancer. Urol Oncol [Epub ahead of print]
Hernández-Bautista RJ, Alarcón-Aguilar FJ, Del C, Escobar-Villanueva M, Almanza-Pérez JC, Merino-Aguilar H, Fainstein MK, López-Diazguerrero NE (2014) Biochemical alterations during the obese-aging process in female and Male Monosodium Glutamate (MSG)-treated mice. Int J Mol Sci 15:11473–11494
Hine CM, Mitchell JR (2012) NRF2 and the phase II response in acute stress resistance induced by dietary restriction. J Clin Exp Pathol S4:7329
Hoffmann GR (2009) A perspective on the scientific, philosophical, and policy dimensions of hormesis. Dose Response 7:1–51
Hoffmann GR, Moczula AV, Laterza AM, Macneil LK, Tartaglione JP (2013) Adaptive response to hydrogen peroxide in yeast: induction, time course, and relationship to dose–response models. Environ Mol Mutagen 54:384–396
Hotamisligil GS, Erbay E (2008) Nutrient sensing and inflammation in metabolic diseases. Nat Rev Immunol 8:923–934
Hsu YY, Chen CS, Wu SN, Jong YJ, Lo YC (2012) Berberine activates Nrf2 nuclear translocation and protects against oxidative damage via a phosphatidylinositol 3-kinase/Akt-dependent mechanism in NSC34 motor neuron-like cells. Eur J Pharm Sci 46:415–425
Imhoff BR, Hansen JM (2010) Tert-butylhydroquinone induces mitochondrial oxidative stress causing Nrf2 activation. Cell Biol Toxicol 26:541–5
Ishii T, Mann GE (2014) Redox status in mammalian cells and stem cells during culture in vitro: critical roles of Nrf2 and cystine transporter activity in the maintenance of redox balance. Redox Biol 2:786–794. doi:10.1016/j.redox.2014.04.008
Itoh K, Tong KI, Yamamoto M (2004) Molecular mechanism activating Nrf2-Keap1 pathway in regulation of adaptive response to electrophiles. Free Radic Biol Med 36:1208–1213
Jacobs AT, Marnett LJ (2007) Heat shock factor 1 attenuates 4-Hydroxynonenal-mediated apoptosis: critical role for heat shock protein 70 induction and stabilization of Bcl-XL. J Biol Chem 282:33412–33421
Jin H, Kanthasamy A, Anantharam V, Rana A, Kanthasamy AG (2011) Transcriptional regulation of pro-apoptotic protein kinase C delta: implications for oxidative stress-induced neuronal cell death. J Biol Chem 286:19840–19859
Kahn SE, Zinman B, Haffner SM, O’Neill MC, Kahn SE1, Zinman B, Haffner SM, O’Neill MC, Kravitz BG, Yu D, Freed MI, Herman WH, Holman RR, Jones NP, Lachin JM, Viberti GC. Obesity is a major determinant of the association of C-reactive protein levels and the metabolic syndrome in type 2 diabetes. Diabetes 55:2357–2364
Khoramian TS, Khalaj L, Ashabi G, Kiaei M, Khodagholi F (2011) Alginate oligosaccharide protects against endoplasmic reticulum- and mitochondrial-mediated apoptotic cell death and oxidative stress. Biomaterials 32:5438–545
Kleszczyński K, Ernst IM, Wagner AE, Kruse N, Zillikens D, Rimbach G, Fischer TW (2013) Sulforaphane and phenylethyl isothiocyanate protect human skin against UVR-induced oxidative stress and apoptosis: role of Nrf2-dependent gene expression and antioxidant enzymes. Pharmacol Res 78:28–40
Kobayashi A, Yamamoto M (2006) Nrf2-Keap1 regulation of cellular defense mechanisms against electrophiles and reactive oxygen species. Adv Enzym Regul 46:113–140
Kobayashi A, Kang M, Watai Y, Tong KI, Shibata T, Uchida K, Yamamoto M (2006) Oxidative and electrophilicstresses activate Nrf2 through inhibition of ubiquitination activity of Keap. Mol Cel Biol 26:221–229
Komatsu M, Kurokawa H, Waguri S, Taguchi K, Kobayashi A, Ichimura Y, Sou YS, Ueno I, Sakamoto A, Tong KI, Kim M, Nishito Y, Iemura S, Natsume T, Ueno T, Kominami E, Motohashi H, Tanaka K, Yamamoto M (2010) The selective autophagy substrate p62 activates the stress responsive transcription factor Nrf2 through inactivation of Keap1. Nat Cell Biol 12:213–223
Kregel KC, Zhang HJ (2007) An integrated view of oxidative stress in aging: basic mechanisms, functional effects, and pathological considerations. Am J Physiol Regul Integr Comp Physiol 292:18–36
Kubicova L1, Hadacek F, Chobot V (2013) Quinolinic acid: neurotoxin or oxidative stress modulator? Int J Mol Sci 14(11):21328–38
Lainscak M, von Haehling S, Doehner W, Anker SD (2012) The obesity paradox in chronic diseases:facts and numbers. J Cachex Sarcopenia Muscle 3:1–4
Landbo C, Prescott E, Lange P, Vestbo J, Almdal TP (1999) Prognostic value of nutritional status in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 160:1856–1861
Le Bourg E, Rattan SI (2009) Is hormesis applicable as a pro-healthy aging intervention in mammals and human beings, and how? Introduction to a special issue of Dose-Response. Dose Response 8:1–3. doi:10.2203/dose-response.09-052
Lee MJ, Wu Y, Fried SK (2010) Adipose tissue remodeling in pathophysiology of obesity. Curr Opin Clin Nutr Metab Care 13:371–376
Lee SK, Shehzad A, Jung JC, Sonn JK, Lee JT, Park JW, Lee YS (2012) Protein kinase Cα protects against multidrug resistance in human colon cancer cells. Mol Cells 34:61–69
Lerner C, Bitto A, Pulliam D, Nacarelli T, Konigsberg M, Van Remmen H, Torres C, Sell C (2013) Reduced mammalian target of rapamycin activity facilitates mitochondrial retrograde signaling and increases life span in normal human fibroblasts. Aging Cell 12:966–977
Luna-López A, Triana-Martínez F, López-Diazguerrero NE, Ventura-Gallegos JL, Gutiérrez-Ruiz MC, Damián-Matsumura P, Zentella A, Gómez-Quiroz LE, Königsberg M (2010) Bcl-2 sustains hormetic response by inducing Nrf-2 nuclear translocation in L929 mouse fibroblasts. Free Radic Biol Med 49:1192–1204. doi:10.1016/j.freeradbiomed.2010.07.004
Matsuda M, Shimomura I (2013) Increased oxidative stress in obesity: implications for metabolic syndrome, diabetes, hypertension, dyslipidemia, atherosclerosis, and cancer. Obes Res Clin Pract 7:330–341
Mattson MP (2008) Hormesis and disease resistance: activation of cellular stress response pathways. Hum Exp Toxicol 27:155–162
Meakin PJ, Chowdhry S, Sharma RS, Ashford FB, Walsh SV, McCrimmon RJ, Dinkova-Kostova AT, Dillon JF, Hayes JD, Ashford ML (2014) Susceptibility of Nrf2-null mice to steatohepatitis and cirrhosis upon consumption of a high-fat diet is associated with oxidative stress, perturbation of the unfolded protein response, and disturbance in the expression of metabolic enzymes but not with insulin resistance. Mol Cell Biol 34:3305–3320
Mendelsohn AR, Larrick JW (2014) Paradoxical effects of antioxidants on cancer. Rejuvenation Res 17:306–311
Mendoza-Núñez VM, Sánchez-Rodríguez MA, Correa-Muñoz E (2009) Undernutrition and oxidative stress as risk factors for high blood pressure in older Mexican adults. Ann Nutr Metab 54:119–123
Mishra S, Vinayak M (2014) Ellagic acid inhibits PKC signaling by improving antioxidant defense system in murine T cell lymphoma. Mol Biol Rep 4:4187–4197
Morimoto RI, Cuervo AM (2014) Proteostasis and the aging proteome in health and disease. J Gerontol A Biol Sci Med Sci 69(Suppl 1):S33–8
Mukherjee S, Ghosh S, Choudhury S, Adhikary A, Manna K, Dey S, Sa G, Das T, Chattopadhyay S (2013) Pomegranate reverses methotrexate-induced oxidative stress and apoptosis in hepatocytes by modulating Nrf2-NF-κB pathways. J Nutr Biochem 24:2040–2050
Namani A, Li Y, Wang XJ, Tang X (2014) Modulation of NRF2 signaling pathway by nuclear receptors: implications for cancer. Biochim Biophys Acta 1843:1875–1885
Niture SK, Jaiswal AK (2012) Nrf2 protein up-regulates antiapoptotic protein Bcl-2 and prevents cellular apoptosis. J Biol Chem 287:9873–9886
Niture SK, Jain AK, Jaiswal AK (2009) Antioxidant-induced modification of INrf2 cysteine 151 and PKC-delta-mediated phosphorylation of Nrf2 serine 40 are both required for stabilization and nuclear translocation of Nrf2 and increased drug resistance. J Cell Sci 122:4452–4464
Nunn AVW, Bell JD, Guy GW (2009) Lifestyle-induced metabolic inflexibility and accelerated ageing syndrome: insulin resistance, friend or foe? kNutr Metab 6:16
Okin D, Medzhitov R (2012) Evolution of inflammatory diseases. Curr Biol 22:733–740
Park SH, Jang JH, Chen CY, Na HK, Surh YJ (2010) A formulated red ginseng extract rescues PC12 cells from PCB-induced oxidative cell death through Nrf2-mediated upregulation of heme oxygenase-1 and glutamate cysteine ligase. Toxicology 278:131–139
Pulliam DA, Deepa SS, Liu Y, Hill S, Lin AL, Bhattacharya A, Shi Y, Sloane L, Viscomi C, Zeviani M, Van Remmen H (2014) Complex IV deficient surf1−/− mice initiate mitochondrial stress responses. Biochem J. [Epub ahead of print]
Queisser N, Oteiza PI, Link S, Hey V, Stopper H, Schupp N (2014) Aldosterone activates transcription factor Nrf2 in kidney cells both in vitro and in vivo. Antioxid Redox Signal. [Epub ahead of print]
Rattan SI (2006) Hormetic modulation of aging and longevity by mild heat stress. Dose Response 3:533–546
Ravuri C1, Svineng G, Huseby NE (2013) Differential regulation of γ-glutamyltransferase and glutamate cysteine ligase expression after mitochondrial uncoupling: γ-glutamyltransferase is regulated in an Nrf2- and NFκB-independent manner. Free Radic Res 47:394–403
Ristow M, Schmeisser S (2011) Extending life span by increasing oxidative stress. Free Radic Biol Med 51:327–336
Rohrer PR, Rudraiah S, Goedken MJ, Manautou JE (2014) Is nuclear factor erythroid 2-related factor 2 responsible for sex differences in susceptibility to acetaminophen-induced hepatotoxicity in mice? Drug Metab Dispos 42:1663–1674
Rubio V, Valverde M, Rojas E (2010) Effects of atmospheric pollutants on the Nrf2 survival pathway. Environ Sci Pollut Res Int 17:369–382
Santana-Martínez RA, Galván-Arzáte S, Hernández-Pando R, Chánez-Cárdenas ME, Avila-Chávez E, López-Acosta G, Pedraza-Chaverrí J, Santamaría A, Maldonado PD (2014) Sulforaphane reduces the alterations induced by quinolinic acid: modulation of glutathione levels. Neuroscience 272:188–198
Satoh T, McKercher SR, Lipton SA (2013) Nrf2/ARE-mediated antioxidant actions of pro-electrophilic drugs. Free Radic Biol Med 65:645–57
Scannapieco AC, Sørensen JG, Loeschcke V, Norry FM (2007) Heat-induced hormesis in longevity of two sibling drosophila species. Biogerontology 8:315–325
Shaw PX, Werstuck G, Chen Y (2014) Oxidative medicine and cellular longevity. Oxi Med Cel Longev 569146
Shoelson SE, Lee J, Goldfine AB (2006) Inflammation and insulin resistance. J Clin Investig 7:1793–1801
Singh B, Shoulson R, Chatterjee A, Ronghe A, Bhat NK, Dim DC, Bhat HK (2014) Resveratrol inhibits estrogen-induced breast carcinogenesis through induction of NRF2-mediated protective pathways. Carcinog. [Epub ahead of print]
Song HJ, Oh S, Quan S, Ryu OH, Jeong JY, Hong KS, Kim DH (2014) Gender differences in adiponectin levels and body composition in older adults: hallym aging study. BMC Geriatr 14:8
Speciale A, Chirafisi J, Saija A, Cimino F (2011) Nutritional antioxidants and adaptive cell responses: an update. Curr Mol Med 11:770–789
Taguchi K, Fujii S, Yamano S, Cho AK, Kamisuki S, Nakai Y, Sugawara F, Froines JR, Verhagen H, Furnee C, Schutte B, Hermans R, Kumagai Y (2007) An approach to evaluate two-electron reduction of 9,10-phenanthraquinone and redox activity of the hydroquinone associatedwith oxidativestress. Free Radic Biol Med 43:789–799
Trujillo J1, Chirino YI, Molina-Jijón E, Andérica-Romero AC, Tapia E, Pedraza-Chaverrí J (2013) Renoprotective effect of the antioxidant curcumin: recent findings. Redox Biol 1:448–456
Uretsky S, Messerli FH, Bangalore S, Champion A, Cooper-Dehoff RM, Zhou Q, Pepine CJ (2007) Obesity paradox in patients with hypertension and coronary artery disease. Am J Med 120:863–870
Vida C, Gonzalez EM, Fuente MD (2014) Increase of oxidation and inflammation in nervous and immune systems withaging and anxiety. Curr Pharm Des [Epub ahead of print]
Wakabayashi N, Itoh K, Wakabayashi J, Motohashi H, Noda S, Takahashi S, Imakado S, Kotsuji T, Otsuka F, Roop DR, Harada T, Engel JD, Yamamoto M (2003) Keap1-null mutation leads to postnatal lethality due to constitutive Nrf2 activation. Nat Genet 35:238–245
Wang W, Wu Y, Zhang G, Fang H, Wang H, Zang H, Xie T, Wang W (2014a) Activation of Nrf2-ARE signal pathway protects the brain from damage induced by epileptic seizure. Brain Res 1544:54–61
Wang XJ, Li Y, Luo L, Wang H, Chi Z, Xin A, Li X, Wu J, Tang X (2014b) Oxaliplatin activates the Keap1/Nrf2 antioxidant system conferring protection against the cytotoxicity of anticancer drugs. Free Radic Biol Med 70:68–77. doi:10.1016/j.freeradbiomed.2014.02.010
Wiese AG, Pacifici RE, Davies KJA (1995) Transcient adaptation to oxidative stress in mammalian cells. Arch Biochem Biophys 318:231–240
Xu J, Wang H, Ding K, Zhang L, Wang C, Li T, Wei W, Lu X (2014) Luteolin provides neuroprotection in models of traumatic brain injury via the Nrf2–ARE pathway. Free Radic Biol Med 71:186–195
Yadav A, Kataria MA, Saini V, Yadav A (2013) Role of leptin and adiponectin in insulin resistance. Clin Chim Acta 417:80–84
Yan L (2014) Positive oxidative stress in aging and aging-related disease tolerance. Redox Biol 2:165–169
Zanotto-Filho A, Gelain DP, Schröder R, Souza LF, Pasquali MA, Klamt F, Moreira JC (2009) The NF kappa B-mediated control of RS and JNK signaling in vitamin A-treated cells: duration of JNK-AP-1 pathway activation may determine cell death or proliferation. Biochem Pharmacol 77:1291–1301
Zucker SN, Fink EE1, Bagati A, Mannava S, Bianchi-Smiraglia A, Bogner PN, Wawrzyniak JA, Foley C, Leonova KI, Grimm MJ, Moparthy K, Ionov Y, Wang J, Liu S, Sexton S, Kandel ES, Bakin AV, Zhang Y, Kaminski N, Segal BH, Nikiforov MA (2014) Nrf2 amplifies oxidative stress via induction of Klf9. Mol Cell 53:916–928
Acknowledgments
The authors want to acknowledge M. in BE Luis A. Maciel for his comments to this paper. This work was supported by CONACyT’s grant CB-2012-1-178349. As well as the “Red Temática de Investigación en Salud y Desarrollo Social” from CONACYT and INGER DI-PI004/2012.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luna–López, A., González-Puertos, V.Y., López-Diazguerrero, N.E. et al. New considerations on hormetic response against oxidative stress. J. Cell Commun. Signal. 8, 323–331 (2014). https://doi.org/10.1007/s12079-014-0248-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-014-0248-4