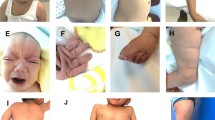
Abstract
Athelia is a very rare entity that is defined by the absence of the nipple–areola complex. It can affect either sex and is mostly part of syndromes including other congenital or ectodermal anomalies, such as limb-mammary syndrome, scalp–ear–nipple syndrome, or ectodermal dysplasias. Here, we report on three children from two branches of an extended consanguineous Israeli Arab family, a girl and two boys, who presented with a spectrum of nipple anomalies ranging from unilateral hypothelia to bilateral athelia but no other consistently associated anomalies except a characteristic eyebrow shape. Using homozygosity mapping after single nucleotide polymorphism (SNP) array genotyping and candidate gene sequencing we identified a homozygous frameshift mutation in PTPRF as the likely cause of nipple anomalies in this family. PTPRF encodes a receptor-type protein phosphatase that localizes to adherens junctions and may be involved in the regulation of epithelial cell–cell contacts, peptide growth factor signaling, and the canonical Wnt pathway. Together with previous reports on female mutant Ptprf mice, which have a lactation defect, and disruption of one allele of PTPRF by a balanced translocation in a woman with amastia, our results indicate a key role for PTPRF in the development of the nipple–areola region.



Similar content being viewed by others
References
Ausavarat S, Tongkobpetch S, Praphanphoj V, Mahatumarat C, Rojvachiranonda N, Snabboon T, Markello TC, Gahl WA, Suphapeetiporn K, Shotelersuk V (2011) PTPRF is disrupted in a patient with syndromic amastia. BMC Med Genet 12:46. doi:10.1186/1471-2350-12-46
Borck G, Rehman AU, Lee K, Pogoda HM, Kakar N, von Ameln S, Grillet N, Hildebrand MS, Ahmed ZM, Nürnberg G, Ansar M, Basit S, Javed Q, Morell RJ, Nasreen N, Shearer AE, Ahmad A, Kahrizi K, Shaikh RS, Ali RA, Khan SN, Goebel I, Meyer NC, Kimberling WJ, Webster JA, Stephan DA, Schiller MR, Bahlo M, Najmabadi H, Gillespie PG, Nürnberg P, Wollnik B, Riazuddin S, Smith RJ, Ahmad W, Müller U, Hammerschmidt M, Friedman TB, Riazuddin S, Leal SM, Ahmad J, Kubisch C (2011) Loss-of-function mutations of ILDR1 cause autosomal-recessive hearing impairment DFNB42. Am J Hum Genet 88(2):127–137. doi:10.1016/j.ajhg.2010.12.011
Cho YM, Lewis DA, Koltz PF, Richard V, Gocken TA et al (2004) Regulation of parathyroid hormone-related protein gene expression by epidermal growth factor-family ligands in primary human keratinocytes. J Endocrinol 181(1):179–190
Du WW, Fang L, Li M, Yang X, Liang Y, Peng C, Qian W, O’Malley YQ, Askeland RW, Sugg SL, Qian J, Lin J, Jiang Z, Yee AJ, Sefton M, Deng Z, Shan SW, Wang CH, Yang BB (2013) MicroRNA miR-24 enhances tumor invasion and metastasis by targeting PTPN9 and PTPRF to promote EGF signaling. J Cell Sci 126(Pt 6):1440–1453. doi:10.1242/jcs.118299
Dunah AW, Hueske E, Wyszynski M, Hoogenraad CC, Jaworski J, Pak DT, Simonetta A, Liu G, Sheng M (2005) LAR receptor protein tyrosine phosphatases in the development and maintenance of excitatory synapses. Nat Neurosci 8(4):458–467
Foley J, Dann P, Hong J, Cosgrove J, Dreyer B, Rimm D, Dunbar M, Philbrick W, Wysolmerski J (2001) Parathyroid hormone-related protein maintains mammary epithelial fate and triggers nipple skin differentiation during embryonic breast development. Development 128(4):513–525
Fraser FC (1956) Dominant inheritance of absent nipples and breasts. Novant’anni delle leggi mendeliane. Rome: Istituto Gregorio Mendel (pub) pp 360
Harrington RJ, Gutch MJ, Hengartner MO, Tonks NK, Chisholm AD (2002) The C. elegans LAR-like receptor tyrosine phosphatase PTP-3 and the VAB-1 Eph receptor tyrosine kinase have partly redundant functions in morphogenesis. Development 129(9):2141–2153
Hiremath M, Dann P, Fischer J, Butterworth D, Boras-Granic K, Hens J, Van Houten J, Shi W, Wysolmerski J (2012) Parathyroid hormone-related protein activates Wnt signaling to specify the embryonic mammary mesenchyme. Development 139(22):4239–4249. doi:10.1242/dev.080671
Incassati A, Chandramouli A, Eelkema R, Cowin P (2010) Key signaling nodes in mammary gland development and cancer: β-catenin. Breast Cancer Res 12(6):213. doi:10.1186/bcr2723
Ishida LH, Alves HR, Munhoz AM, Kaimoto C, Ishida LC, Saito FL, Gemperlli R, Ferreira MC (2005) Athelia: case report and review of the literature. Br J Plast Surg 58(6):833–837. doi:10.1016/j.bjps.2005.01.018
Kim WK, Jung H, Kim DH, Kim EY, Chung JW, Cho YS, Park SG, Park BC, Ko Y, Bae KH, Lee SC (2009) Regulation of adipogenic differentiation by LAR tyrosine phosphatase in human mesenchymal stem cells and 3T3-L1 preadipocytes. J Cell Sci 122(Pt 22):4160–4167. doi:10.1242/jcs.053009
King TD, Suto MJ, Li Y (2012) The Wnt/β-catenin signaling pathway: a potential therapeutic target in the treatment of triple negative breast cancer. J Cell Biochem 113(1):13–18. doi:10.1002/jcb.23350
Kobayashi T, Kronenberg HM, Foley J (2005) Reduced expression of the PTH/PTHrP receptor during development of the mammary gland influences the function of the nipple during lactation. Dev Dyn 233(3):794–803
Kowlessar M, Orti E (1968) Complete breast absence in siblings. Am J Dis Child 115(1):91–92. doi:10.1001/archpedi.1968.02100010093018
Koyama S, Wu HJ, Easwaran T, Thopady S, Foley J (2013) The nipple: a simple intersection of mammary gland and integument, but focal point of organ function. J Mammary Gland Biol Neoplasia 18(2):121–131. doi:10.1007/s10911-013-9289-1
Machide M, Hashigasako A, Matsumoto K, Nakamura T (2006) Contact inhibition of hepatocyte growth regulated by functional association of the c-Met/hepatocyte growth factor receptor and LAR protein-tyrosine phosphatase. J Biol Chem 281(13):8765–8772
Mander A, Hodgkinson CP, Sale GJ (2005) Knock-down of LAR protein tyrosine phosphatase induces insulin resistance. FEBS Lett 579(14):3024–3028
Moore KL, Persaud TVN (1998) The developing human, clinically oriented embryology, 5th edn. Saunders, Philadelphia, pp 448–449
Nelson MM, Cooper CK (1982) Congenital defects of the breast—an autosomal dominant trait. S Afr Med J 61(12):434–436
Nickerson NK, Mohammad KS, Gilmore JL, Crismore E, Bruzzaniti A, Guise TA, Foley J (2012) Decreased autocrine EGFR signaling in metastatic breast cancer cells inhibits tumor growth in bone and mammary fat pad. PLoS One 7(1):e30255. doi:10.1371/journal.pone.0030255
Schaapveld RQ, Schepens JT, Robinson GW, Attema J, Oerlemans FT, Fransen JA, Streuli M, Wieringa B, Hennighausen L, Hendriks WJ (1997) Impaired mammary gland development and function in mice lacking LAR receptor-like tyrosine phosphatase activity. Dev Biol 188(1):134–146
Schaapveld RQ, Schepens JT, Bächner D, Attema J, Wieringa B, Jap PH, Hendriks WJ (1998) Developmental expression of the cell adhesion molecule-like protein tyrosine phosphatases LAR, RPTPdelta and RPTPsigma in the mouse. Mech Dev 77(1):59–62
Stewart K, Uetani N, Hendriks W, Tremblay ML, Bouchard M (2013) Inactivation of LAR family phosphatase genes Ptprs and Ptprf causes craniofacial malformations resembling Pierre-Robin sequence. Development 140(16):3413–3422. doi:10.1242/dev.094532
Symons JR, LeVea CM, Mooney RA (2002) Expression of the leucocyte common antigen-related (LAR) tyrosine phosphatase is regulated by cell density through functional E-cadherin complexes. Biochem J 365(Pt 2):513–519
Tautz L, Critton DA, Grotegut S (2013) Protein tyrosine phosphatases: structure, function, and implication in human disease. Methods Mol Biol 1053:179–221. doi:10.1007/978-1-62703-562-0_13
Tonks NK (2013) Protein tyrosine phosphatases—from housekeeping enzymes to master regulators of signal transduction. FEBS J 280(2):346–378. doi:10.1111/febs.12077
Toribio RE, Brown HA, Novince CM, Marlow B, Hernon K, Lanigan LG, Hildreth BE 3rd, Werbeck JL, Shu ST, Lorch G, Carlton M, Foley J, Boyaka P, McCauley LK, Rosol TJ (2010) The midregion, nuclear localization sequence, and C terminus of PTHrP regulate skeletal development, hematopoiesis, and survival in mice. FASEB J 24(6):1947–1957. doi:10.1096/fj.09-147033
Um JW, Ko J (2013) LAR-RPTPs: synaptic adhesion molecules that shape synapse development. Trends Cell Biol 23(10):465–475. doi:10.1016/j.tcb.2013.07.004
Wang X, Weng LP, Yu Q (2000) Specific inhibition of FGF-induced MAPK activation by the receptor-like protein tyrosine phosphatase LAR. Oncogene 19(19):2346–2353
Wilson MG, Hall EB, Ebbin AJ (1972) Dominant inheritance of absence of the breast. Humangenetik 15(3):268–270
Zabolotny JM, Kim YB, Peroni OD, Kim JK, Pani MA, Boss O, Klaman LD, Kamatkar S, Shulman GI, Kahn BB, Neel BG (2001) Overexpression of the LAR (leukocyte antigen-related) protein-tyrosine phosphatase in muscle causes insulin resistance. Proc Natl Acad Sci USA 98(9):5187–5192
Zheng W, Lennartsson J, Hendriks W, Heldin CH, Hellberg C (2011) The LAR protein tyrosine phosphatase enables PDGF β-receptor activation through attenuation of the c-Abl kinase activity. Cell Signal 23(6):1050–1056. doi:10.1016/j.cellsig.2011.01.024
Author information
Authors and Affiliations
Corresponding authors
Additional information
G. Borck and L. de Vries contributed equally.
C. Kubisch and L. Basel-Vanagaite jointly supervised this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1. Eyebrow shape in a patient with intellectual disability, normal nipples, and no PTPRF mutation.
Rights and permissions
About this article
Cite this article
Borck, G., de Vries, L., Wu, HJ. et al. Homozygous truncating PTPRF mutation causes athelia. Hum Genet 133, 1041–1047 (2014). https://doi.org/10.1007/s00439-014-1445-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-014-1445-1