Abstract
Purpose
Glioblastoma multiforme (GBM) is the most common primary brain tumor and has a very poor overall prognosis. Multimodal treatment is still inefficient and one main reason is the invasive nature of GBM cells, enabling the tumor cells to escape from the treatment area causing tumor progression. This experimental study describes the effect of low- and high-LET irradiation on the invasion of primary GBM cells with a validation in established cell systems.
Methods
Seven patient derived primary GBM as well as three established cell lines (LN229, LN18 and U87) were used in this study. Invasion was investigated using Matrigel® coated transwell chambers. Irradiation was performed with low- (X-ray) and high-LET (alpha particles) radiation. The colony formation assay was chosen to determine the corresponding alpha particle dose equivalent to the X-ray dose.
Results
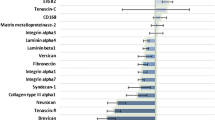
4 Gy X-ray irradiation increased the invasive potential of six patient derived GBM cells as well as two of the established lines. In contrast, alpha particle irradiation with an equivalent dose of 1.3 Gy did not show any effect on the invasive behavior. The findings were validated with established cell lines.
Conclusion
Our results show that in contrast to low-LET irradiation high-LET irradiation does not enhance the invasion of established and primary glioblastoma cell lines. We therefore suggest that high-LET irradiation could become an alternative treatment option. To fully exploit the benefits of high-LET irradiation concerning the invasion of GBM further molecular studies should be performed.





Similar content being viewed by others
Data availability
The datasets generated during and analysed during the current study are available from the corresponding author on reasonable request.
References
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131(6):803–820. https://doi.org/10.1007/s00401-016-1545-1
Hou LC, Veeravagu A, Hsu AR, Tse VC (2006) Recurrent glioblastoma multiforme: a review of natural history and management options. Neurosurg Focus 20(4):E5
Raychaudhuri B, Vogelbaum MA (2011) IL-8 is a mediator of NF-kappaB induced invasion by gliomas. J Neuro-oncol 101(2):227–235. https://doi.org/10.1007/s11060-010-0261-2
Averbeck NB, Topsch J, Scholz M, Kraft-Weyrather W, Durante M, Taucher-Scholz G (2016) Efficient rejoining of DNA double-strand breaks despite increased cell-killing effectiveness following spread-out Bragg peak carbon-ion irradiation. Front Oncol 6:28. https://doi.org/10.3389/fonc.2016.00028
Fitzek MM, Thornton AF, Harsh G, Rabinov JD, Munzenrider JE, Lev M, Ancukiewicz M, Bussiere M, Hedley-Whyte ET, Hochberg FH, Pardo FS (2001) Dose-escalation with proton/photon irradiation for Daumas-Duport lower-grade glioma: results of an institutional phase I/II trial. Int J Radiat Oncol Biol Phys 51(1):131–137
Krex D, Klink B, Hartmann C, von Deimling A, Pietsch T, Simon M, Sabel M, Steinbach JP, Heese O, Reifenberger G, Weller M, Schackert G (2007) Long-term survival with glioblastoma multiforme. Brain 130(Pt 10):2596–2606. https://doi.org/10.1093/brain/awm204
Curran Jr WJC, Scott CB, Weinstein AS, Martin LA, Nelson JS, Phillips TL, Murray K, Fischbach AJ, Yakar D, Schwade JG (1993) Survival comparison of radiosurgery-eligible and -ineligible malignant glioma patients treated with hyperfractionated radiation therapy and carmustine: a report of Radiation Therapy Oncology Group 83–02. J Clin Oncol 11(5):857–862. https://doi.org/10.1200/jco.1993.11.5.857
Combs SE, Kessel K, Habermehl D, Haberer T, Jakel O, Debus J (2013) Proton and carbon ion radiotherapy for primary brain tumors and tumors of the skull base. Acta Oncol 52(7):1504–1509. https://doi.org/10.3109/0284186x.2013.818255
Paw I, Carpenter RC, Watabe K, Debinski W, Lo HW (2015) Mechanisms regulating glioma invasion. Cancer Lett 362(1):1–7. https://doi.org/10.1016/j.canlet.2015.03.015
von Essen CF (1991) Radiation enhancement of metastasis: a review. Clin Exp Metastasis 9(2):77–104
Rieken S, Habermehl D, Mohr A, Wuerth L, Lindel K, Weber K, Debus J, Combs SE (2011) Targeting alphanubeta3 and alphanubeta5 inhibits photon-induced hypermigration of malignant glioma cells. Radiat Oncol 6:132. https://doi.org/10.1186/1748-717x-6-132
Edalat L, Stegen B, Klumpp L, Haehl E, Schilbach K, Lukowski R, Kuhnle M, Bernhardt G, Buschauer A, Zips D, Ruth P, Huber SM (2016) BK K + channel blockade inhibits radiation-induced migration/brain infiltration of glioblastoma cells. Oncotarget 7(12):14259–14278. https://doi.org/10.18632/oncotarget.7423
Kil WJ, Tofilon PJ, Camphausen K (2012) Post-radiation increase in VEGF enhances glioma cell motility in vitro. Radiat Oncol 7:25. https://doi.org/10.1186/1748-717x-7-25
Pribluda A, de la Cruz CC, Jackson EL (2015) Intratumoral heterogeneity: from diversity comes resistance. Clin Cancer Res 21(13):2916–2923. https://doi.org/10.1158/1078-0432.ccr-14-1213
Kleihues P, Louis DN, Scheithauer BW, Rorke LB, Reifenberger G, Burger PC, Cavenee WK (2002) The WHO classification of tumors of the nervous system. J Neuropathol Exp Neurol 61(3):215–225 (discussion 226–219)
Hirota Y, Masunaga S, Kondo N, Kawabata S, Hirakawa H, Yajima H, Fujimori A, Ono K, Kuroiwa T, Miyatake S (2014) High linear-energy-transfer radiation can overcome radioresistance of glioma stem-like cells to low linear-energy-transfer radiation. J Radiat Res 55(1):75–83. https://doi.org/10.1093/jrr/rrt095
Combs SE, Bruckner T, Mizoe JE, Kamada T, Tsujii H, Kieser M, Debus J (2013) Comparison of carbon ion radiotherapy to photon radiation alone or in combination with temozolomide in patients with high-grade gliomas: explorative hypothesis-generating retrospective analysis. Radiother Oncol 108(1):132–135. https://doi.org/10.1016/j.radonc.2013.06.026
Combs SE, Kieser M, Rieken S, Habermehl D, Jakel O, Haberer T, Nikoghosyan A, Haselmann R, Unterberg A, Wick W, Debus J (2010) Randomized phase II study evaluating a carbon ion boost applied after combined radiochemotherapy with temozolomide versus a proton boost after radiochemotherapy with temozolomide in patients with primary glioblastoma: the CLEOPATRA trial. BMC Cancer 10:478. https://doi.org/10.1186/1471-2407-10-478
Combs SE, Bohl J, Elsasser T, Weber KJ, Schulz-Ertner D, Debus J, Weyrather WK (2009) Radiobiological evaluation and correlation with the local effect model (LEM) of carbon ion radiation therapy and temozolomide in glioblastoma cell lines. Int J Radiat Biol 85(2):126–137. https://doi.org/10.1080/09553000802641151
Durante M, Loeffler JS (2010) Charged particles in radiation oncology. Nat Rev Clin Oncol 7(1):37–43. https://doi.org/10.1038/nrclinonc.2009.183
Roos H, Kellerer AM (1989) Design criteria and performance parameters of an alpha irradiation device for cell studies. Phys Med Biol 34(12):1823
Baulch JE, Geidzinski E, Tran KK, Yu L, Zhou YH, Limoli CL (2016) Irradiation of primary human gliomas triggers dynamic and aggressive survival responses involving microvesicle signaling. Environ Mol Mutagenesis 57(5):405–415. https://doi.org/10.1002/em.21988
Park CM, Park MJ, Kwak HJ, Lee HC, Kim MS, Lee SH, Park IC, Rhee CH, Hong SI (2006) Ionizing radiation enhances matrix metalloproteinase-2 secretion and invasion of glioma cells through Src/epidermal growth factor receptor-mediated p38/Akt and phosphatidylinositol 3-kinase/Akt signaling pathways. Cancer Res 66(17):8511–8519. https://doi.org/10.1158/0008-5472.can-05-4340
Cordes N, Hansmeier B, Beinke C, Meineke V, van Beuningen D (2003) Irradiation differentially affects substratum-dependent survival, adhesion, and invasion of glioblastoma cell lines. Br J Cancer 89(11):2122–2132. https://doi.org/10.1038/sj.bjc.6601429
Rieken S, Habermehl D, Wuerth L, Brons S, Mohr A, Lindel K, Weber K, Haberer T, Debus J, Combs SE (2012) Carbon ion irradiation inhibits glioma cell migration through downregulation of integrin expression. Int J Radiat Oncol Biol Phys 83(1):394–399. https://doi.org/10.1016/j.ijrobp.2011.06.2004
Akino Y, Teshima T, Kihara A, Kodera-Suzumoto Y, Inaoka M, Higashiyama S, Furusawa Y, Matsuura N (2009) Carbon-ion beam irradiation effectively suppresses migration and invasion of human non-small-cell lung cancer cells. Int J Radiat Oncol Biol Phys 75(2):475–481. https://doi.org/10.1016/j.ijrobp.2008.12.090
Ogata T, Teshima T, Inaoka M, Minami K, Tsuchiya T, Isono M, Furusawa Y, Matsuura N (2011) Carbon Ion Irradiation Suppresses Metastatic Potential of Human Non-small Cell Lung Cancer A549 Cells through the Phosphatidylinositol-3-Kinase/Akt Signaling Pathway. J Radiat Res 52(3):374–379. https://doi.org/10.1269/jrr.10102
Babu R, Komisarow JM, Agarwal VJ, Rahimpour S, Iyer A, Britt D, Karikari IO, Grossi PM, Thomas S, Friedman AH, Adamson C (2016) Glioblastoma in the elderly: the effect of aggressive and modern therapies on survival. J Neurosurg 124(4):998–1007. https://doi.org/10.3171/2015.4.jns142200
Polivka J Jr, Polivka J, Holubec L, Kubikova T, Priban V, Hes O, Pivovarcikova K, Treskova I (2017) Advances in experimental targeted therapy and immunotherapy for patients with glioblastoma multiforme. Anticancer Res 37(1):21–33. https://doi.org/10.21873/anticanres.11285
Barazzuol L, Jena R, Burnet NG, Jeynes JC, Merchant MJ, Kirkby KJ, Kirkby NF (2012) In vitro evaluation of combined temozolomide and radiotherapy using X rays and high-linear energy transfer radiation for glioblastoma. Radiat Res 177(5):651–662
Benzina S, Altmeyer A, Malek F, Dufour P, Denis J-M, Gueulette J, Bischoff P (2008) High-LET radiation combined with oxaliplatin induce autophagy in U-87 glioblastoma cells. Cancer Lett 264(1):63–70. https://doi.org/10.1016/j.canlet.2008.01.023
Mizoe JE, Tsujii H, Hasegawa A, Yanagi T, Takagi R, Kamada T, Tsuji H, Takakura K (2007) Phase I/II clinical trial of carbon ion radiotherapy for malignant gliomas: combined X-ray radiotherapy, chemotherapy, and carbon ion radiotherapy. Int J Radiat Oncol Biol Phys 69(2):390–396. https://doi.org/10.1016/j.ijrobp.2007.03.003
Dreher C, Habermehl D, Jakel O, Combs SE (2017) Effective radiotherapeutic treatment intensification in patients with pancreatic cancer: higher doses alone, higher RBE or both? Radiat Oncol 12(1):203. https://doi.org/10.1186/s13014-017-0945-2
Acknowledgements
The authors thank Friederike Lämmer for help and guidance in primary culture techniques.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Every author of this manuscript declares that he/she has no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Wank, M., Schilling, D., Reindl, J. et al. Evaluation of radiation-related invasion in primary patient-derived glioma cells and validation with established cell lines: impact of different radiation qualities with differing LET. J Neurooncol 139, 583–590 (2018). https://doi.org/10.1007/s11060-018-2923-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-2923-4