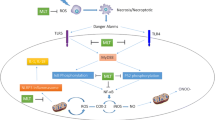
Abstract
It is estimated that more than half of cancer patients undergo radiotherapy during the course of their treatment. Despite its beneficial therapeutic effects on tumor cells, exposure to high doses of ionizing radiation (IR) is associated with several side effects. Although improvements in radiotherapy techniques and instruments could reduce these side effects, there are still important concerns for cancer patients. For several years, scientists have been trying to modulate tumor and normal tissue responses to IR, leading to an increase in therapeutic ratio. So far, several types of radioprotectors and radiosensitizers have been investigated in experimental studies. However, high toxicity of chemical sensitizers or possible tumor protection by radioprotectors creates a doubt for their clinical applications. On the other hand, the protective effects of these radioprotectors or sensitizer effects of radiosensitizers may limit some type of cancers. Hence, the development of some radioprotectors without any protective effect on tumor cells or low toxic radiosensitizers can help improve therapeutic ratio with less side effects. Melatonin as a natural body hormone is a potent antioxidant and anti-inflammatory agent that shows some anti-cancer properties. It is able to neutralize different types of free radicals produced by IR or pro-oxidant enzymes which are activated following exposure to IR and plays a key role in the protection of normal tissues. In addition, melatonin has shown the ability to inhibit long-term changes in inflammatory responses at different levels, thereby ameliorating late side effects of radiotherapy. Fortunately, in contrast to classic antioxidants, some in vitro studies have revealed that melatonin has a potent anti-tumor activity when used alongside irradiation. However, the mechanisms of its radiosensitive effect remain to be elucidated. Studies suggested that the activation of pro-apoptosis gene, such as p53, changes in the metabolism of tumor cells, suppression of DNA repair responses as well as changes in biosynthesis of estrogen in breast cancer cells are involved in this process. In this review, we describe the molecular mechanisms for radioprotection and radiosensitizer effects of melatonin. Furthermore, some other proposed mechanisms that may be involved are presented.


Similar content being viewed by others
References
Liauw SL, Connell PP, Weichselbaum RR. New paradigms and future challenges in radiation oncology: an update of biological targets and technology. Sci Transl Med. 2013;5(173):173sr2-sr2. https://doi.org/10.1126/scitranslmed.3005148.
Allison R, Dicker A. Minimizing morbidity in radiation oncology: a special issue from future oncology. Future Oncol. 2014;10(15):2303–5. https://doi.org/10.2217/fon.14.195.
Narmani A, Farhood B, Haghi-Aminjan H, Mortezazadeh T, Aliasgharzadeh A, Mohseni M, et al. Gadolinium nanoparticles as diagnostic and therapeutic agents: their delivery systems in magnetic resonance imaging and neutron capture therapy. J Drug Deliv Sci Technol. 2018;44:457–66. https://doi.org/10.1016/j.jddst.2018.01.011.
Bagheri H, Rezapour S, Najafi M, Motevaseli E, Shekarchi B, Cheki M et al. Protection against radiation-induced micronuclei in rat bone marrow erythrocytes by Curcumin and selenium l-methionine. Iran J Med Sci. 2018.
Najafi M, Motevaseli E, Shirazi A, Geraily Gh, Rezaeyan A, Norouzi F et al. Mechanisms of inflammatory responses to radiation and normal tissues toxicity: clinical implications. Int J Radiat Biol. 2018;94(4):335–356.
Bourhis J, Rosine D. Radioprotective effect of amifostine in patients with head and neck squamous cell carcinoma. Semin Oncol. 2002;29(6 Suppl 19):61–2. https://doi.org/10.1053/sonc.2002.37349.
Rosenthal DI, Chambers MS, Weber RS, Eisbruch A. A phase II study to assess the efficacy of amifostine for submandibular/sublingual salivary sparing during the treatment of head and neck cancer with intensity modulated radiation therapy for parotid salivary sparing. Semin Oncol. 2004;31(6 Suppl 18):25–8. https://doi.org/10.1053/j.seminoncol.2004.12.008.
Gu J, Zhu S, Li X, Wu H, Li Y, Hua F. Effect of amifostine in head and neck cancer patients treated with radiotherapy: a systematic review and meta-analysis based on randomized controlled trials. PLoS One. 2014;9(5):e95968. https://doi.org/10.1371/journal.pone.0095968.
Buntzel J, Glatzel M, Schuth J, Weinaug R, Kuttner K, Frohlich D. Cytoprotection with amifostine in the framework of radiochemotherapy in previously irradiated head and neck carcinoma. Strahlenther Onkol. 1999;175(Suppl 4):37–40.
Rades D, Fehlauer F, Bajrovic A, Mahlmann B, Richter E, Alberti W. Serious adverse effects of amifostine during radiotherapy in head and neck cancer patients. Radiother Oncol. 2004;70(3):261–4. https://doi.org/10.1016/j.radonc.2003.10.005.
Thorstad WL, Chao KS, Haughey B. Toxicity and compliance of subcutaneous amifostine in patients undergoing postoperative intensity-modulated radiation therapy for head and neck cancer. Semin Oncol. 2004;31(6 Suppl 18):8–12. https://doi.org/10.1053/j.seminoncol.2004.12.005.
Chao KS, Ozyigit G, Thorsdad WL. Toxicity profile of intensity-modulated radiation therapy for head and neck carcinoma and potential role of amifostine. Semin Oncol. 2003;30(6 Suppl 18):101–8.
Singh VK, Fatanmi OO, Wise SY, Newman VL, Romaine PL, Seed TM. The potentiation of the radioprotective efficacy of two medical countermeasures, gamma-tocotrienol and amifostine, by a combination prophylactic modality. Radiat Prot Dosim. 2016;172(1–3):302–10.
Ghobadi A, Shirazi A, Najafi M, Kahkesh MH, Rezapoor S. Melatonin ameliorates radiation-induced oxidative stress at targeted and nontargeted lung tissue. J Med Phys. 2017;42(4):241.
Martínez-Campa C, Menéndez-Menéndez J, Alonso-González C, González A, Álvarez-García V, Cos S. What is known about melatonin, chemotherapy and altered gene expression in breast cancer. Oncol Lett. 2017;13(4):2003–14. https://doi.org/10.3892/ol.2017.5712.
Lissoni P. Biochemotherapy with immunomodulating pineal hormones other than melatonin: 5-methoxytryptamine as a new oncostatic pineal agent. Pathol Biol (Paris). 2007;55(3–4):198–200. https://doi.org/10.1016/j.patbio.2006.12.008.
Yahyapour R, Shabeeb D, Cheki M, Musa AE, Farhood B, Rezaeyan A et al. Radiation protection and mitigation by natural antioxidants and flavonoids; implications to radiotherapy and radiation disasters. Curr Mol Pharmacol. 2018. https://doi.org/10.2174/1874467211666180619125653.
Chen C-Q, Fichna J, Bashashati M, Li Y-Y, Storr M. Distribution, function and physiological role of melatonin in the lower gut. World J Gastroenterol WJG. 2011;17(34):3888–98. https://doi.org/10.3748/wjg.v17.i34.3888.
Talib WH. Melatonin and cancer hallmarks. Molecules. 2018. https://doi.org/10.3390/molecules23030518.
Yu G-M, Kubota H, Okita M, Maeda T. The anti-inflammatory and antioxidant effects of melatonin on LPS-stimulated bovine mammary epithelial cells. PLoS One. 2017;12(5):e0178525. https://doi.org/10.1371/journal.pone.0178525.
Favero G, Franceschetti L, Bonomini F, Rodella LF, Rezzani R. Melatonin as an anti-inflammatory agent modulating inflammasome activation. Int J Endocrinol. 2017;2017:1835195. https://doi.org/10.1155/2017/1835195.
Volt H, Garcia JA, Doerrier C, Diaz-Casado ME, Guerra-Librero A, Lopez LC, et al. Same molecule but different expression: aging and sepsis trigger NLRP3 inflammasome activation, a target of melatonin. J Pineal Res. 2016;60(2):193–205. https://doi.org/10.1111/jpi.12303.
Yu H, Dickson EJ, Jung S-R, Koh D-S, Hille B. High membrane permeability for melatonin. J Gen Physiol. 2016;147(1):63–76. https://doi.org/10.1085/jgp.201511526.
Vasin MV, Ushakov IB. Comparative efficacy and the window of radioprotection for adrenergic and serotoninergic agents and aminothiols in experiments with small and large animals. J Radiat Res. 2015;56(1):1–10. https://doi.org/10.1093/jrr/rru087.
Mozdarani H. Biological complexities in radiation carcinogenesis and cancer radiotherapy: impact of new biological paradigms. Genes (Basel). 2012;3(1):90–114. https://doi.org/10.3390/genes3010090.
Najafi M, Cheki M, Rezapoor S, Geraily G, Motevaseli E, Carnovale C, et al. Metformin: prevention of genomic instability and cancer: a review. Mutat Res. 2018;827:1–8. https://doi.org/10.1016/j.mrgentox.2018.01.007.
Manda K, Glasow A, Paape D, Hildebrandt G. Effects of ionizing radiation on the immune system with special emphasis on the interaction of dendritic and T cells. Front Oncol. 2012;2:102. https://doi.org/10.3389/fonc.2012.00102.
Hekim N, Cetin Z, Nikitaki Z, Cort A, Saygili EI. Radiation triggering immune response and inflammation. Cancer Lett. 2015;368(2):156–63. https://doi.org/10.1016/j.canlet.2015.04.016.
Rodel F, Frey B, Multhoff G, Gaipl U. Contribution of the immune system to bystander and non-targeted effects of ionizing radiation. Cancer Lett. 2015;356(1):105–13. https://doi.org/10.1016/j.canlet.2013.09.015.
Yahyapour R, Amini P, Rezapour S, Cheki M, Rezaeyan A, Farhood B, et al. Radiation-induced inflammation and autoimmune diseases. Mil Med Res. 2018;5(1):9. https://doi.org/10.1186/s40779-018-0156-7.
Yahyapour R, Motevaseli E, Rezaeyan A, Abdollahi H, Farhood B, Cheki M, et al. Mechanisms of radiation bystander and non-targeted effects: implications to radiation carcinogenesis and radiotherapy. Curr Radiopharm. 2018;11(1):34–45. https://doi.org/10.2174/1874471011666171229123130.
Yahyapour R, Motevaseli E, Rezaeyan A, Abdollahi H, Farhood B, Cheki M et al. Reduction–oxidation (redox) system in radiation-induced normal tissue injury: molecular mechanisms and implications in radiation therapeutics. Clin Transl Oncol. 2018;20(8):975–988. https://doi.org/10.1007/s12094-017-1828-6.
Yahyapour R, Amini P, Rezapoor S, Rezaeyan A, Farhood B, Cheki M et al. Targeting of inflammation for radiation protection and mitigation. Curr Mol Pharmacol. 2018;11(3):203–210
Holley AK, Miao L, St. Clair DK, St. Clair WH. Redox-modulated phenomena and radiation therapy: the central role of superoxide dismutases. Antioxid Redox Signal. 2014;20(10):1567–89. https://doi.org/10.1089/ars.2012.5000.
Miao L, Holley AK, Zhao Y, St. Clair DK, St. Clair WH. Redox-mediated and ionizing-radiation-induced inflammatory mediators in prostate cancer development and treatment. Antioxid Redox Signal. 2014;20(9):1481–500. https://doi.org/10.1089/ars.2013.5637.
Castellani P, Balza E, Rubartelli A. Inflammation, DAMPs, tumor development, and progression: a vicious circle orchestrated by redox signaling. Antioxid Redox Signal. 2014;20(7):1086–97. https://doi.org/10.1089/ars.2012.5164.
Vijayalaxmi, Reiter RJ, Herman TS, Meltz ML. Melatonin reduces gamma radiation-induced primary DNA damage in human blood lymphocytes. Mutat Res. 1998;397(2):203–8.
Vijayalaxmi, Reiter RJ, Meltz ML, Herman TS. Melatonin: possible mechanisms involved in its ‘radioprotective’ effect. Mutat Res Fundam Mol Mech Mutagen. 1998;404(1):187–9. https://doi.org/10.1016/S0027-5107(98)00112-2.
Vijayalaxmi, Reiter RJ, Tan DX, Herman TS, Thomas CR Jr. Melatonin as a radioprotective agent: a review. Int J Radiat Oncol Biol Phys. 2004;59(3):639–53. https://doi.org/10.1016/j.ijrobp.2004.02.006.
Galano A, Tan D-X, Reiter RJ. Melatonin: a versatile protector against oxidative DNA damage. Molecules. 2018;23(3):530.
Reiter RJ, Tan DX, Manchester LC, Qi W. Biochemical reactivity of melatonin with reactive oxygen and nitrogen species: a review of the evidence. Cell Biochem Biophys. 2001;34(2):237–56. https://doi.org/10.1385/cbb:34:2:237.
Karbownik M, Reiter RJ. Antioxidative effects of melatonin in protection against cellular damage caused by ionizing radiation. Proc Soc Exp Biol Med. 2000;225(1):9–22.
Tan DX, Reiter RJ, Manchester LC, Yan MT, El-Sawi M, Sainz RM, et al. Chemical and physical properties and potential mechanisms: melatonin as a broad spectrum antioxidant and free radical scavenger. Curr Top Med Chem. 2002;2(2):181–97.
Karslioglu I, Ertekin MV, Taysi S, Kocer I, Sezen O, Gepdiremen A, et al. Radioprotective effects of melatonin on radiation-induced cataract. J Radiat Res. 2005;46(2):277–82.
Cakmak Karaer I, Simsek G, Yildiz A, Vardi N, Polat A, Tanbek K, et al. Melatonin’s protective effect on the salivary gland against ionized radiation damage in rats. J Oral Pathol Med. 2016;45(6):444–9. https://doi.org/10.1111/jop.12386.
Koc M, Taysi S, Buyukokuroglu ME, Bakan N. Melatonin protects rat liver against irradiation-induced oxidative injury. J Radiat Res. 2003;44(3):211–5.
Sener G, Jahovic N, Tosun O, Atasoy BM, Yegen BC. Melatonin ameliorates ionizing radiation-induced oxidative organ damage in rats. Life Sci. 2003;74(5):563–72.
Sener G, Atasoy BM, Ersoy Y, Arbak S, Sengoz M, Yegen BC. Melatonin protects against ionizing radiation-induced oxidative damage in corpus cavernosum and urinary bladder in rats. J Pineal Res. 2004;37(4):241–6. https://doi.org/10.1111/j.1600-079X.2004.00161.x.
Koc M, Taysi S, Emin Buyukokuroglu M, Bakan N. The effect of melatonin against oxidative damage during total-body irradiation in rats. Radiat Res. 2003;160(2):251–5.
Barlas AM, Sadic M, Atilgan HI, Bag YM, Onalan AK, Yumusak N, et al. Melatonin: a hepatoprotective agent against radioiodine toxicity in rats. Bratisl Lek Listy. 2017;118(2):95–100. https://doi.org/10.4149/bll_2017_020.
McDonald JT, Kim K, Norris AJ, Vlashi E, Phillips TM, Lagadec C, et al. Ionizing radiation activates the Nrf2 antioxidant response. Cancer Res. 2010;70(21):8886–95. https://doi.org/10.1158/0008-5472.can-10-0171.
Jiang Y, Chen X, Tian W, Yin X, Wang J, Yang H. The role of TGF-beta1-miR-21-ROS pathway in bystander responses induced by irradiated non-small-cell lung cancer cells. Br J Cancer. 2014;111(4):772–80. https://doi.org/10.1038/bjc.2014.368.
Tian W, Yin X, Wang L, Wang J, Zhu W, Cao J, et al. The key role of miR-21-regulated SOD2 in the medium-mediated bystander responses in human fibroblasts induced by alpha-irradiated keratinocytes. Mutat Res. 2015;780:77–85. https://doi.org/10.1016/j.mrfmmm.2015.08.003.
Janjetovic Z, Jarrett SG, Lee EF, Duprey C, Reiter RJ, Slominski AT. Melatonin and its metabolites protect human melanocytes against UVB-induced damage: involvement of NRF2-mediated pathways. Sci Rep. 2017;7(1):1274. https://doi.org/10.1038/s41598-017-01305-2.
Guo Y, Sun J, Li T, Zhang Q, Bu S, Wang Q, et al. Melatonin ameliorates restraint stress-induced oxidative stress and apoptosis in testicular cells via NF-κB/iNOS and Nrf2/HO-1 signaling pathway. Sci Rep. 2017;7:9599. https://doi.org/10.1038/s41598-017-09943-2.
Amini P, Mirtavoos-Mahyari H, Motevaseli E, Shabeeb D, Musa AE, Cheki M et al. Mechanisms for radioprotection by melatonin; can it be used as a radiation countermeasure?. Curr Mol Pharmacol. 2018. https://doi.org/10.2174/1874467211666180802164449.
Brand RM, Epperly MW, Stottlemyer JM, Skoda EM, Gao X, Li S, et al. A topical mitochondria-targeted redox-cycling nitroxide mitigates oxidative stress-induced skin damage. J Investig Dermatol. 2017;137(3):576–86. https://doi.org/10.1016/j.jid.2016.09.033.
Kam WW, Banati RB. Effects of ionizing radiation on mitochondria. Free Radic Biol Med. 2013;65:607–19. https://doi.org/10.1016/j.freeradbiomed.2013.07.024.
Farhood B, Goradel NH, Mortezaee K, Khanlarkhani N, Salehi E, Nashtaei MS, et al. Intercellular communications-redox interactions in radiation toxicity; potential targets for radiation mitigation. J Cell Commun Signal. 2018. https://doi.org/10.1007/s12079-018-0473-3.
Atkinson J, Kapralov AA, Yanamala N, Tyurina YY, Amoscato AA, Pearce L, et al. A mitochondria-targeted inhibitor of cytochrome c peroxidase mitigates radiation-induced death. Nat Commun. 2011;2:497. https://doi.org/10.1038/ncomms1499.
Rwigema J-CM, Beck B, Wang W, Doemling A, Epperly MW, Shields D, et al. Two strategies for the development of mitochondrial-targeted small molecule radiation damage mitigators. Int J Radiat Oncol Biol Phys. 2011;80(3):860–8. https://doi.org/10.1016/j.ijrobp.2011.01.059.
Reiter RJ, Tan DX, Mayo JC, Sainz RM, Leon J, Czarnocki Z. Melatonin as an antioxidant: biochemical mechanisms and pathophysiological implications in humans. Acta Biochim Pol. 2003;50(4):1129–46 (0350041129).
Najafi M, Shirazi A, Motevaseli E, Rezaeyan AH, Salajegheh A, Rezapoor S. Melatonin as an anti-inflammatory agent in radiotherapy. Inflammopharmacology. 2017;25(4):403–13. https://doi.org/10.1007/s10787-017-0332-5.
Vijayalaxmi, Reiter RJ, Herman TS, Meltz ML. Melatonin and radioprotection from genetic damage: in vivo/in vitro studies with human volunteers. Mutat Res. 1996;371(3–4):221–8.
Rostami A, Moosavi SA, Dianat Moghadam H, Bolookat ER. Micronuclei assessment of the radioprotective effects of melatonin and vitamin C in human lymphocytes. Cell J. 2016;18(1):46–51.
Rezapoor S, Shirazi A, Abbasi S, Bazzaz JT, Izadi P, Rezaeejam H, et al. Modulation of radiation-induced base excision repair pathway gene expression by melatonin. J Med Phys. 2017;42(4):245–50.
Valizadeh M, Shirazi A, Izadi P, Tavakkoly Bazzaz J, Rezaeejam H. Expression levels of two DNA repair-related genes under 8 Gy ionizing radiation and 100 mg/kg melatonin delivery in rat peripheral blood. J Biomed Phys Eng. 2017;7(1):27–36.
Valizadeh M, Shirazi A, Izadi P, Bazzaz JT, Rezaeejam H, Tabesh GA. Effects of melatonin on repair of DNA double strand breaks caused by ionizing radiation in rat peripheral blood. 2016.
Rezaeejam H, Shirazi A, Izadi P, Bazzaz JT, Ghazi-Khansari M, Valizadeh M, et al. Radioprotective effect of melatonin on expression of Cdkn1a and Rad50 genes in rat peripheral blood. 2018.
Cui YF, Ding YQ, Zhang Y, Xu H, Jin W, Liu XL, et al. Apoptotic characteristics of spleen lymphocyte in mice irradiated by lethal dose and its relationship to the expression of Bax and Bcl-XL proteins. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue. 2005;17(2):109–12.
Mohseni M, Mihandoost E, Shirazi A, Sepehrizadeh Z, Bazzaz JT, Ghazi-khansari M. Melatonin may play a role in modulation of bax and bcl-2 expression levels to protect rat peripheral blood lymphocytes from gamma irradiation-induced apoptosis. Mutat Res. 2012;738–739:19–27. https://doi.org/10.1016/j.mrfmmm.2012.08.006.
Jang SS, Kim WD, Park WY. Melatonin exerts differential actions on X-ray radiation-induced apoptosis in normal mice splenocytes and Jurkat leukemia cells. J Pineal Res. 2009;47(2):147–55. https://doi.org/10.1111/j.1600-079X.2009.00694.x.
Khan S, Adhikari JS, Rizvi MA, Chaudhury NK. Melatonin attenuates (60) Co gamma-ray-induced hematopoietic, immunological and gastrointestinal injuries in C57BL/6 male mice. Environ Toxicol. 2017;32(2):501–18. https://doi.org/10.1002/tox.22254.
Erol FS, Topsakal C, Ozveren MF, Kaplan M, Ilhan N, Ozercan IH, et al. Protective effects of melatonin and vitamin E in brain damage due to gamma radiation: an experimental study. Neurosurg Rev. 2004;27(1):65–9. https://doi.org/10.1007/s10143-003-0291-8.
Fernandez-Gil B, Moneim AE, Ortiz F, Shen YQ, Soto-Mercado V, Mendivil-Perez M, et al. Melatonin protects rats from radiotherapy-induced small intestine toxicity. PLoS One. 2017;12(4):e0174474. https://doi.org/10.1371/journal.pone.0174474.
Ortiz F, Acuna-Castroviejo D, Doerrier C, Dayoub JC, Lopez LC, Venegas C, et al. Melatonin blunts the mitochondrial/NLRP3 connection and protects against radiation-induced oral mucositis. J Pineal Res. 2015;58(1):34–49. https://doi.org/10.1111/jpi.12191.
Onseng K, Johns NP, Khuayjarernpanishk T, Subongkot S, Priprem A, Hurst C, et al. Beneficial effects of adjuvant melatonin in minimizing oral mucositis complications in head and neck cancer patients receiving concurrent chemoradiation. J Altern Complement Med. 2017;23(12):957–63. https://doi.org/10.1089/acm.2017.0081.
Ersahin M, Toklu HZ, Cetinel S, Yuksel M, Yegen BC, Sener G. Melatonin reduces experimental subarachnoid hemorrhage-induced oxidative brain damage and neurological symptoms. J Pineal Res. 2009;46(3):324–32. https://doi.org/10.1111/j.1600-079X.2009.00664.x.
Najafi M, Shirazi A, Motevaseli E, Geraily G, Norouzi F, Heidari M, et al. The melatonin immunomodulatory actions in radiotherapy. Biophys Rev. 2017;9(2):139–48. https://doi.org/10.1007/s12551-017-0256-8.
Gurses I, Ozeren M, Serin M, Yucel N, Erkal HS. Histopathological evaluation of melatonin as a protective agent in heart injury induced by radiation in a rat model. Pathol Res Pract. 2014;210(12):863–71. https://doi.org/10.1016/j.prp.2014.08.006.
Aricigil M, Dundar MA, Yucel A, Eryilmaz MA, Aktan M, Alan MA, et al. Melatonin prevents possible radiotherapy-induced thyroid injury. Int J Radiat Biol. 2017;93(12):1350–6. https://doi.org/10.1080/09553002.2017.1397296.
Escames G, Fernández-Gil BI, Guerra-Librero A, Shen Y, García-López S, Florido J et al. PO-089: Melatonin enhances the toxicity of radio- and chemotherapy in head and neck cancer cells. Radiother Oncol.122:43. https://doi.org/10.1016/s0167-8140(17)30223-2.
Gavande NS, VanderVere-Carozza PS, Hinshaw HD, Jalal SI, Sears CR, Pawelczak KS, et al. DNA repair targeted therapy: the past or future of cancer treatment? Pharmacol Ther. 2016;160:65–83. https://doi.org/10.1016/j.pharmthera.2016.02.003.
Alonso-Gonzalez C, Gonzalez A, Martinez-Campa C, Gomez-Arozamena J, Cos S. Melatonin sensitizes human breast cancer cells to ionizing radiation by downregulating proteins involved in double-strand DNA break repair. J Pineal Res. 2015;58(2):189–97. https://doi.org/10.1111/jpi.12205.
Zheng JIE. Energy metabolism of cancer: glycolysis versus oxidative phosphorylation (Review). Oncol Lett. 2012;4(6):1151–7. https://doi.org/10.3892/ol.2012.928.
Yadav N, Chandra D. Mitochondrial DNA mutations and breast tumorigenesis. Biochim Biophys Acta. 2013;1836(2):336–44. https://doi.org/10.1016/j.bbcan.2013.10.002.
Shimura T, Noma N, Sano Y, Ochiai Y, Oikawa T, Fukumoto M, et al. AKT-mediated enhanced aerobic glycolysis causes acquired radioresistance by human tumor cells. Radiother Oncol. 2014;112(2):302–7. https://doi.org/10.1016/j.radonc.2014.07.015.
Yu L, Chen X, Sun X, Wang L, Chen S. The glycolytic switch in tumors: how many players are involved? J Cancer. 2017;8(17):3430–40. https://doi.org/10.7150/jca.21125.
Robinson GL, Dinsdale D, MacFarlane M, Cain K. Switching from aerobic glycolysis to oxidative phosphorylation modulates the sensitivity of mantle cell lymphoma cells to TRAIL. Oncogene. 2012;31:4996. https://doi.org/10.1038/onc.2012.13.
Cao K, Li J, Chen J, Qian L, Wang A, Chen X, et al. microRNA-33a-5p increases radiosensitivity by inhibiting glycolysis in melanoma. Oncotarget. 2017;8(48):83660–72. https://doi.org/10.18632/oncotarget.19014.
Escames G, Guerra-Librero A, Shen Y, Florido J, Sayed R, Molina-Navarro M et al. PO-090: Oncostatic effect of melatonin in head and neck cancer: role of mitochondrial function. Radiother Oncol. 122:43–4. https://doi.org/10.1016/s0167-8140(17)30224-4.
Yue W, Wang J-P, Li Y, Fan P, Liu G, Zhang N, et al. Effects of estrogen on breast cancer development: role of estrogen receptor independent mechanisms. Int J Cancer J Int Cancer. 2010;127(8):1748–57. https://doi.org/10.1002/ijc.25207.
Russo J, Hasan Lareef M, Balogh G, Guo S, Russo IH. Estrogen and its metabolites are carcinogenic agents in human breast epithelial cells. J Steroid Biochem Mol Biol. 2003;87(1):1–25.
Travis RC, Allen DS, Fentiman IS, Key TJ. Melatonin and breast cancer: a prospective study. JNCI. 2004;96(6):475–82. https://doi.org/10.1093/jnci/djh077.
Sabzichi M, Samadi N, Mohammadian J, Hamishehkar H, Akbarzadeh M, Molavi O. Sustained release of melatonin: a novel approach in elevating efficacy of tamoxifen in breast cancer treatment. Colloids Surf B. 2016;145:64–71. https://doi.org/10.1016/j.colsurfb.2016.04.042.
Kiefer T, Ram PT, Yuan L, Hill SM. Melatonin inhibits estrogen receptor transactivation and cAMP levels in breast cancer cells. Breast Cancer Res Treat. 2002;71(1):37–45.
Lopes J, Arnosti D, Trosko JE, Tai M-H, Zuccari D. Melatonin decreases estrogen receptor binding to estrogen response elements sites on the OCT4 gene in human breast cancer stem cells. Genes Cancer. 2016;7(5–6):209–17. https://doi.org/10.18632/genesandcancer.107.
del Rio B, Garcia Pedrero JM, Martinez-Campa C, Zuazua P, Lazo PS, Ramos S. Melatonin, an endogenous-specific inhibitor of estrogen receptor alpha via calmodulin. J Biol Chem. 2004;279(37):38294–302. https://doi.org/10.1074/jbc.M403140200.
Martinez-Campa C, Alonso-Gonzalez C, Mediavilla MD, Cos S, Gonzalez A, Ramos S, et al. Melatonin inhibits both ER alpha activation and breast cancer cell proliferation induced by a metalloestrogen, cadmium. J Pineal Res. 2006;40(4):291–6. https://doi.org/10.1111/j.1600-079X.2006.00315.x.
Alonso-Gonzalez C, Gonzalez A, Martinez-Campa C, Menendez-Menendez J, Gomez-Arozamena J, Garcia-Vidal A, et al. Melatonin enhancement of the radiosensitivity of human breast cancer cells is associated with the modulation of proteins involved in estrogen biosynthesis. Cancer Lett. 2016;370(1):145–52. https://doi.org/10.1016/j.canlet.2015.10.015.
Martinez-Campa C, Gonzalez A, Mediavilla MD, Alonso-Gonzalez C, Alvarez-Garcia V, Sanchez-Barcelo EJ, et al. Melatonin inhibits aromatase promoter expression by regulating cyclooxygenases expression and activity in breast cancer cells. Br J Cancer. 2009;101(9):1613–9. https://doi.org/10.1038/sj.bjc.6605336.
Cheki M, Yahyapour R, Farhood B, Rezaeyan A, Shabeeb D, Amini P, Rezapoor S, Najafi M. COX-2 in radiotherapy; a potential target for radioprotection and radiosensitization. Curr Mol Pharmacol. 2018;11(3):173–183
Bizzarri M, Proietti S, Cucina A, Reiter RJ. Molecular mechanisms of the pro-apoptotic actions of melatonin in cancer: a review. Expert Opin Ther Targets. 2013;17(12):1483–96. https://doi.org/10.1517/14728222.2013.834890.
Cheng Y, Cai L, Jiang P, Wang J, Gao C, Feng H, et al. SIRT1 inhibition by melatonin exerts antitumor activity in human osteosarcoma cells. Eur J Pharmacol. 2013;715(1–3):219–29. https://doi.org/10.1016/j.ejphar.2013.05.017.
Cucina A, Proietti S, D’Anselmi F, Coluccia P, Dinicola S, Frati L, et al. Evidence for a biphasic apoptotic pathway induced by melatonin in MCF-7 breast cancer cells. J Pineal Res. 2009;46(2):172–80. https://doi.org/10.1111/j.1600-079X.2008.00645.x.
Kosar PA, Naziroglu M, Ovey IS, Cig B. Synergic effects of doxorubicin and melatonin on apoptosis and mitochondrial oxidative stress in MCF-7 breast cancer cells: involvement of TRPV1 channels. J Membr Biol. 2016;249(1–2):129–40. https://doi.org/10.1007/s00232-015-9855-0.
Alonso-Gonzalez C, Menendez-Menendez J, Gonzalez-Gonzalez A, Gonzalez A, Cos S, Martinez-Campa C. Melatonin enhances the apoptotic effects and modulates the changes in gene expression induced by docetaxel in MCF7 human breast cancer cells. Int J Oncol. 2018;52(2):560–70. https://doi.org/10.3892/ijo.2017.4213.
Uguz AC, Cig B, Espino J, Bejarano I, Naziroglu M, Rodriguez AB, et al. Melatonin potentiates chemotherapy-induced cytotoxicity and apoptosis in rat pancreatic tumor cells. J Pineal Res. 2012;53(1):91–8. https://doi.org/10.1111/j.1600-079X.2012.00974.x.
Gao Y, Xiao X, Zhang C, Yu W, Guo W, Zhang Z, et al. Melatonin synergizes the chemotherapeutic effect of 5-fluorouracil in colon cancer by suppressing PI3K/AKT and NF-kappaB/iNOS signaling pathways. J Pineal Res. 2017. https://doi.org/10.1111/jpi.12380.
Gore E. Celecoxib and radiation therapy in non-small-cell lung cancer. Oncology (Williston Park). 2004;18(14 Suppl 14):10–4.
Gore E, Bae K, Langer C, Extermann M, Movsas B, Okunieff P, et al. Phase I/II trial of a COX-2 inhibitor with limited field radiation for intermediate prognosis patients who have locally advanced non-small-cell lung cancer: radiation therapy oncology group 0213. Clin Lung Cancer. 2011;12(2):125–30. https://doi.org/10.1016/j.cllc.2011.03.007.
Ganswindt U, Budach W, Jendrossek V, Becker G, Bamberg M, Belka C. Combination of celecoxib with percutaneous radiotherapy in patients with localised prostate cancer—a phase I study. Radiat Oncol (London, England). 2006;1:9. https://doi.org/10.1186/1748-717x-1-9.
Li W, Wang Z, Chen Y, Wang K, Lu T, Ying F, et al. Melatonin treatment induces apoptosis through regulating the nuclear factor-κB and mitogen-activated protein kinase signaling pathways in human gastric cancer SGC7901 cells. Oncol Lett. 2017;13(4):2737–44. https://doi.org/10.3892/ol.2017.5785.
Shrestha S, Zhu J, Wang Q, Du X, Liu F, Jiang J, et al. Melatonin potentiates the antitumor effect of curcumin by inhibiting IKKbeta/NF-kappaB/COX-2 signaling pathway. Int J Oncol. 2017;51(4):1249–60. https://doi.org/10.3892/ijo.2017.4097.
Yi C, Zhang Y, Yu Z, Xiao Y, Wang J, Qiu H, et al. Melatonin enhances the anti-tumor effect of fisetin by inhibiting COX-2/iNOS and NF-kappaB/p300 signaling pathways. PLoS One. 2014;9(7):e99943. https://doi.org/10.1371/journal.pone.0099943.
Wang J, Guo W, Chen W, Yu W, Tian Y, Fu L, et al. Melatonin potentiates the antiproliferative and pro-apoptotic effects of ursolic acid in colon cancer cells by modulating multiple signaling pathways. J Pineal Res. 2013;54(4):406–16. https://doi.org/10.1111/jpi.12035.
Lu JJ, Fu L, Tang Z, Zhang C, Qin L, Wang J, et al. Melatonin inhibits AP-2beta/hTERT, NF-kappaB/COX-2 and Akt/ERK and activates caspase/Cyto C signaling to enhance the antitumor activity of berberine in lung cancer cells. Oncotarget. 2016;7(3):2985–3001. https://doi.org/10.18632/oncotarget.6407.
Woo SM, Min KJ, Kwon TK. Melatonin-mediated Bim up-regulation and cyclooxygenase-2 (COX-2) down-regulation enhances tunicamycin-induced apoptosis in MDA-MB-231 cells. J Pineal Res. 2015;58(3):310–20. https://doi.org/10.1111/jpi.12217.
Pioli C, Caroleo MC, Nistico G, Doria G. Melatonin increases antigen presentation and amplifies specific and non specific signals for T-cell proliferation. Int J Immunopharmacol. 1993;15(4):463–8.
Poon AM, Liu ZM, Pang CS, Brown GM, Pang SF. Evidence for a direct action of melatonin on the immune system. Biol Signals. 1994;3(2):107–17.
Bonilla E, Rodon C, Valero N, Pons H, Chacin-Bonilla L, Garcia Tamayo J, et al. Melatonin prolongs survival of immunodepressed mice infected with the Venezuelan equine encephalomyelitis virus. Trans R Soc Trop Med Hyg. 2001;95(2):207–10.
Currier NL, Miller SC. Echinacea purpurea and melatonin augment natural-killer cells in leukemic mice and prolong life span. J Altern Complement Med. 2001;7(3):241–51. https://doi.org/10.1089/107555301300328115.
Christopher FL, Dussault I, Miller SC. Population dynamics of natural killer cells in the spleen and bone marrow of normal and leukemic mice during in vivo exposure to interleukin-2. Immunobiology. 1991;184(1):37–52. https://doi.org/10.1016/s0171-2985(11)80570-x.
Leon J, Casado J, Jimenez Ruiz SM, Zurita MS, Gonzalez-Puga C, Rejon JD, et al. Melatonin reduces endothelin-1 expression and secretion in colon cancer cells through the inactivation of FoxO-1 and NF-kappabeta. J Pineal Res. 2014;56(4):415–26. https://doi.org/10.1111/jpi.12131.
Cos S, Blask DE. Melatonin modulates growth factor activity in MCF-7 human breast cancer cells. J Pineal Res. 1994;17(1):25–32.
Cos S, Fernandez R, Guezmes A, Sanchez-Barcelo EJ. Influence of melatonin on invasive and metastatic properties of MCF-7 human breast cancer cells. Cancer Res. 1998;58(19):4383–90.
Jardim-Perassi BV, Lourenco MR, Doho GM, Grigolo IH, Gelaleti GB, Ferreira LC, et al. Melatonin regulates angiogenic factors under hypoxia in breast cancer cell lines. Anticancer Agents Med Chem. 2016;16(3):347–58.
Raben D, Helfrich B. Angiogenesis inhibitors: a rational strategy for radiosensitization in the treatment of non-small-cell lung cancer? Clin Lung Cancer. 2004;6(1):48–57. https://doi.org/10.3816/CLC.2004.n.021.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent for this article is not required.
Rights and permissions
About this article
Cite this article
Farhood, B., Goradel, N.H., Mortezaee, K. et al. Melatonin as an adjuvant in radiotherapy for radioprotection and radiosensitization. Clin Transl Oncol 21, 268–279 (2019). https://doi.org/10.1007/s12094-018-1934-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-018-1934-0