Abstract
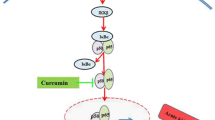
Curcumin, a polyphenol, has pharmacological effects including antioxidant, anti-inflammatory and anti-cancer features. In this study, we have performed comparative in vivo evaluations of CDF (curcumin difluorinated) and curcumin in cisplatin-induced nephrotoxicity in rats. Male Wistar rats were divided into four groups: (1) Control; (2) Cisplatin (7 mg/kg body wt, intraperitoneal as a single dose); (3) Cisplatin and CDF (50 mg/rat/day; for 12 days); (4) Cisplatin and curcumin (50 mg/rat/day), for 12 days). Cisplatin treated rats exhibited kidney injury manifested by increased serum N-urea and creatinine (P < 0.001). Kidney from cisplatin treated rats also exhibited significant increase in malondialdehyde (MDA) and 8-isoprostane levels (P < 0.001). Treatment with CDF and curcumin prevented the rise in serum N-urea, creatinine, MDA and 8-isoprostane as compared to experimental control group in kidney (P < 0.05). Compared to curcumin, CDF had greater potential in suppressing cisplatin-induced pro-inflammatory factors NF-κB and COX-2 as well as downstream markers Nrf2 and HO-1 (P < 0.05) in kidney. The analysis on anion transport markers (OAT1 and OAT3) showed a similar trend (CDF > curcumin). CDF could reduce the expression of multi-drug resistance markers OCT1, OCT2, MRP2 and MRP4 to a much greater extent than curcumin (P < 0.05). We also demonstrate that CDF influenced the expression of p-mTOR, p-p70S6K1, p-4E-BP1 and p-Akt. These data suggest that CDF can potentially be used to reduce the chemotherapy induced nephrotoxicity thereby enhancing the therapeutic window of cisplatin. The results also proved that compared to curcumin, CDF has superior protective effect in nephrotoxicity.



Similar content being viewed by others
References
Rosenberg B, VanCamp L, Trosko JE et al (1969) Platinum compounds: a new class of potent antitumour agents. Nature 222:385–386
Loehrer PJ, Einhorn LH (1984) Drugs five years later. Cisplatin. Ann Intern Med 100:704–713
Ali BH, Al Moundhri MS (2006) Agents ameliorating or augmenting the nephrotoxicity of cisplatin and other platinum compounds: a review of some recent research. Food Chem Toxicol 44:1173–1183
Saha S, Adhikary A, Bhattacharyya P et al (2012) Death by design: where curcumin sensitizes drug-resistant tumours. Anticancer Res 32:2567–2584
Mendonça LM, da Silva Machado C, Teixeira CC et al (2013) Curcumin reduces cisplatin-induced neurotoxicity in NGF-differentiated PC12 cells. Neurotoxicology 34:205–211
Goel A, Aggarwal BB (2010) Curcumin, the golden spice from Indian saffron, is a chemosensitizer and radiosensitizer for tumors and chemoprotector and radioprotector for normal organs. Nutr Cancer 62:919–930
Dhillon N, Aggarwal BB, Newman RA et al (2008) Phase II trial of curcumin in patients with advanced pancreatic cancer. Clin Cancer Res 14:4491–4499
Kanai M, Yoshimura K, Asada M et al (2010) A phase I/II study of gemcitabine-based chemotherapy plus curcumin for patients with gemcitabine-resistant pancreatic cancer. Cancer Chemother Pharmacol 68:157–1649
Bar-Sela G, Epelbaum R, Schaffer M (2010) Curcumin as an anti-cancer agent: review of the gap between basic and clinical applications. Curr Med Chem 17:190–197
Cen L, Hutzen B, Ball S et al (2009) New structural analogues of curcumin exhibit potent growth suppressive activity in human colorectal carcinoma cells. BMC Cancer 9:99
Padhye S, Banerjee S, Chavan D et al (2009) Fluorocurcumins as cyclooxygenase-2 inhibitor: molecular docking, pharmacokinetics and tissue distribution in mice. Pharm Res 26:2438–2445
Ali S, Ahmad A, Banerjee S et al (2010) Gemcitabine sensitivity can be induced in pancreatic cancer cells through modulation of miR-200 and miR-21 expression by curcumin or its analogue CDF. Cancer Res 70:3606–3617
Dandawate PR, Vyas A, Ahmad A et al (2012) Inclusion complex of novel curcumin analogue CDF and β-cyclodextrin (1:2) and its enhanced in vivo anticancer activity against pancreatic cancer. Pharm Res 29:1775–1786
Azmi AS, Ali S, Banerjee S et al (2011) Network modeling of CDF treated pancreatic cancer cells reveals a novel c-myc-p73 dependent apoptotic mechanism. Am J Transl Res 3:374–382
Bao B, Ali S, Kong D et al (2011) Anti-tumor activity of a novel compound-CDF is mediated by regulating miR-21, miR-200, and PTEN in pancreatic cancer. Plos One 6, E17850
Kanwar SS, Yu Y, Nautiyal J et al (2011) Difluorinated-curcumin (CDF): a novel curcumin analog is a potent inhibitor of colon cancer stem-like cells. Pharm Res 28:827–838
Dandawate P, Khan E, Padhye S et al (2012) Synthesis, characterization, molecular docking and cytotoxic activity of novel plumbagin hydrazones against breast cancer cells. Bioorg Med Chem Lett 22:3104–3108
Li Y, Kong D, Wang Z et al (2011) Inactivation of AR/TMPRSS2-ERG/Wnt signaling networks attenuates the aggressive behavior of prostate cancer cells. Cancer Prev Res (Phila) 4:1495–1506
Bao B, Ali S, Banerjee S et al (2012) Curcumin analogue CDF inhibits pancreatic tumor growth by switching on suppressor microRNAs and attenuating EZH2 expression. Cancer Res 72:335–345
Ulu R, Dogukan A, Tuzcu M et al (2012) Regulation of renal organic anion and cation transporters by thymoquinone in cisplatin induced kidney injury. Food Chem Toxicol 50:1675–1679
Karatepe M (2004) Simultaneous determination of ascorbic acid and free malondialdehyde in human serum by HPLC/UV. LC-GC North Am 22:362–365
Wong YT, Ruan R, Tay FE (2006) Relationship between levels of oxidative DNA damage, lipid peroxidation and mitochondrial membrane potential in young and old F344 rats. Free Radic Res 40:393–402
Banerjee S, Kaseb AO, Wang Z et al (2009) Antitumor activity of gemcitabine and oxaliplatin is augmented by thymoquinone in pancreatic cancer. Cancer Res 69:5575–5583
Azmi AS, Philip PA, Beck FW et al (2011) MI-219–zinc combination: a new paradigm in MDM2 inhibitor-based therapy. Oncogene 30:117–126
Piccart MJ, Lamb H, Vermorken JB (2001) Current and future potential roles of the platinum drugs in the treatment of ovarian cancer. Ann Oncol 12:1195–1203
Surendiran A, Balamurugan N, Gunaseelan K et al (2010) Adverse drug reaction profile of cisplatin-based chemotherapy regimen in a tertiary care hospital in India: an evaluative study. Indian J Pharmacol 42:40–43
Hanigan MH, Devarajan P (2003) Cisplatin nephrotoxicity: molecular mechanisms. Cancer Ther 1:47–61
Alaikov T, Konstantinov SM, Tzanova T et al (2007) Antineoplastic and anticlastogenic properties of curcumin. Ann N Y Acad Sci 1095:355–370
Antunes LMG, Darin JDC, Bianchi NLP (2001) Effects of the antioxidants curcumin or selenium on cisplatin-induced nephrotoxicity and lipid peroxidation in rats. Pharmacol Res 43:145–150
Antunes LMG, Araújo MC, Darin JD et al (2000) Effects of the antioxidants curcumin and vitamin C on cisplatin-induced clastogenesis in Wistar rat bone marrow cells. Mutat Res 465:131–137
Kuhad A, Pilkhwal S, Sharma S et al (2007) Effect of curcumin on inflammation and oxidative stress in cisplatin-induced experimental nephrotoxicity. J Agric Food Chem 55:10150–10155
Ilbey YO, Ozbek E, Cekmen M et al (2009) Protective effect of curcumin in cisplatin-induced oxidative injury in rat testis: mitogen-activated protein kinase and nuclear factor-kappa B signaling pathways. Hum Reprod 24:1717–1725
Mendonça LM, dos Santos GC, dos Santos RA et al (2010) Evaluation of curcumin and cisplatin-induced DNA damage in PC12 cells by the alkaline comet assay. Hum Exp Toxicol 29:635–643
Anand P, Kunnumakkara AB, Newman RA et al (2007) Bioavailability of curcumin: problems and promises. Mol Pharm 4:807–818
Kim SH, Hong KO, Chung WY et al (2004) Abrogation of cisplatin-induced hepatotoxicity in mice by xanthorrhizol is related to its effect on the regulation of gene transcription. Toxicol Appl Pharmacol 196:346–355
Banning A, Brigelius-Flohé R (2005) NF-kappaB, Nrf2, and HO-1 interplay in redox-regulated VCAM-1 expression. Antioxid Redox Signal 7:889–899
Trujillo J, Chirino YI, Molina-Jijón E et al (2013) Renoprotective effect of the antioxidant curcumin: recent findings. Redox Biol 17:448–456
Soetikno V, Sari FR, Lakshmanan AP et al (2013) Curcumin alleviates oxidative stress, inflammation, and renal fibrosis in remnant kidney through the Nrf2-keap1 pathway. Mol Nutr Food Res 57:1649–1659
Ramadoss J, Stewart RH, Cudd TA (2011) Acute renal response to rapid onset respiratory acidosis. Can J Physiol Pharmacol 89:227–231
Pritchard JB, Miller DS (1996) Renal secretion of organic anions and cations. Kidney Int 49:1649–1654
Srimaroeng C, Perry JL, Pritchard JB (2008) Physiology, structure, and regulation of the cloned organic anion transporters. Xenobiotica 38:889–935
Ciarimboli G (2008) Organic cation transporters. Xenobiotica 38:936–971
Aleksunes LM, Augustine LM, Scheffer GL et al (2008) Renal xenobiotic transporters are differentially expressed in mice following cisplatin treatment. Toxicology 250:82–88
Tirkey N, Kaur G, Vij G et al (2005) Curcumin, a diferuloylmethane, attenuates cyclosporine-induced renal dysfunction and oxidative stress in rat kidneys. BMC Pharmacol 5:15
Cole SP, Sparks KE, Fraser K et al (1994) Pharmacological characterization of multidrug resistant MRP-transfected human tumor cells. Cancer Res 54:5902–5910
Altenberg GA (2004) Structure of multidrug-resistance proteins of the ATP-binding cassette (ABC) superfamily. Curr Med Chem Anticancer Agents 4:53–62
Reid G, Wielinga P, Zelcer N et al (2003) The human multidrug resistance protein MRP4 functions as a prostaglandin efflux transporter and is inhibited by nonsteroidal antiinflammatory drugs. Proc Natl Acad Sci U S A 100:9244–9249
Reid G, Wielinga P, Zelcer N et al (2003) Characterization of the transport of nucleoside analog drugs by the human multidrug resistance proteins MRP4 and MRP5. Mol Pharmacol 63:1094–1103
Wortelboer HM, Usta M, van der Velde AE et al (2003) Interplay between MRP inhibition and metabolism of MRP inhibitors: the case of curcumin. Chem Res Toxicol 16:1642–1651
Laplante M, Sabatini DM (2012) mTOR signaling in growth control and disease. Cell 149:274–293
Laplante M, Sabatini DM (2012) mTOR Signaling. Cold Spring Harb Perspect Biol 4:1–3
Guertin DA, Sabatini DM (2007) Defining the role of mTOR in cancer. Cancer Cell 12:9–22
Johnson SM, Gulhati P, Rampy BA et al (2010) Novel expression patterns of PI3K/Akt/mTOR signaling pathway components in colorectal cancer. J Am Coll Surg 210:767–778
Johnson SM, Gulhati P, Arrieta I (2009) Curcumin inhibits proliferation of colorectal carcinoma by modulating Akt/mTOR signaling. Anticancer Res 29:3185–3190
Acknowledgments
The authors thank the Veterinary Control and Research Institute of Elazig for providing the experimental facility and the Turkish Academy of Sciences (TUBA) for providing the fund.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sahin, K., Orhan, C., Tuzcu, M. et al. Comparative In Vivo Evaluations of Curcumin and Its Analog Difluorinated Curcumin Against Cisplatin-Induced Nephrotoxicity. Biol Trace Elem Res 157, 156–163 (2014). https://doi.org/10.1007/s12011-014-9886-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-014-9886-x