Abstract
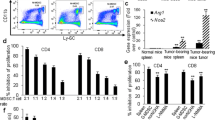
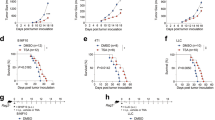
Astragaloside IV (AS-IV) has shown anti-tumorigenic properties in certain cancers for its effect of boosting the body’s immune system, but its role in colorectal cancer (CRC) remains unclear. In this study, we investigated the therapeutic effect of AS-IV in CRC and explored its underlying mechanism. CT26 colon cancer cells and mouse model by injection of CT26 cells subcutaneously were used as in vitro and in vivo model. M1 and M2 macrophage-associated markers, mRNA and protein expression levels were analyzed after AS-IV treatment. Inflammatory factors and cytokines in the tumors from mouse model were detected. Repolarization effect of AS-IV in vitro on bone-marrow-derived macrophages was also detected. In vitro, AS-IV inhibited the proliferation of CT26 cells and induced cell apoptosis dose-dependently, and significantly reduced M2 macrophages and increased M1 macrophages. In mouse model, it suppressed tumor growth and decreased the production of anti-inflammatory factors such as TGF-β, IL-10 and VEGF-A, while increased the production of pro-inflammatory factors like IFN-γ, IL-12 and TNF-α in tumor. Combination of AS-IV and checkpoint inhibitor aPD-1 exhibited synergistic antitumor effect by inhibiting tumor growth and increasing T cell infiltration. AS-IV could induce M2 macrophages polarization to the M1 phenotype. Its combination with immune checkpoint inhibitors could be expected to become a potential new strategy for the treatment of CRC.




Similar content being viewed by others
References
Auyeung KK, Han QB, Ko JK (2016) Astragalus membranaceus: a review of its protection against inflammation and gastrointestinal cancers. Am J Chin Med 44:1–22. https://doi.org/10.1142/S0192415X16500014
Ciombor KK, Wu C, Goldberg RM (2015) Recent therapeutic advances in the treatment of colorectal cancer. Annu Rev Med 66:83–95. https://doi.org/10.1146/annurev-med-051513-102539
Cook RS, Jacobsen KM, Wofford AM et al (2013) MerTK inhibition in tumor leukocytes decreases tumor growth and metastasis. J Clin Invest 123:3231–3242. https://doi.org/10.1172/JCI67655
Cuccarese MF, Dubach JM, Pfirschke C et al (2017) Heterogeneity of macrophage infiltration and therapeutic response in lung carcinoma revealed by 3D organ imaging. Nat Commun 8:14293. https://doi.org/10.1038/ncomms14293
Cully M (2018) Cancer: re-educating tumour-associated macrophages with nanoparticles. Nat Rev Drug Discov 17:468. https://doi.org/10.1038/nrd.2018.102
Dai PC, Liu DL, Zhang L et al (2017) Astragaloside IV sensitizes non-small cell lung cancer cells to gefitinib potentially via regulation of SIRT6. Tumour Biol 39:1010428317697555. https://doi.org/10.1177/1010428317697555
De Palma M, Lewis CE (2013) Macrophage regulation of tumor responses to anticancer therapies. Cancer Cell 23:277–286. https://doi.org/10.1016/j.ccr.2013.02.013
Franklin RA, Liao W, Sarkar A et al (2014) The cellular and molecular origin of tumor-associated macrophages. Science 344:921–925. https://doi.org/10.1126/science.1252510
He CS, Liu YC, Xu ZP et al (2016) Astragaloside IV enhances cisplatin chemosensitivity in non-small cell lung cancer cells through inhibition of B7–H3. Cell Physiol Biochem 40:1221–1229. https://doi.org/10.1159/000453175
Li L, Hou X, Xu R et al (2017) Research review on the pharmacological effects of astragaloside IV. Fundam Clin Pharmacol 31:17–36. https://doi.org/10.1111/fcp.12232
Li S, Yi Sun Y, Huang J et al (2020) Anti-tumor effects and mechanisms of Astragalus membranaceus (AM) and its specific immunopotentiation: status and prospect. J Ethnopharmacol 258:112797. https://doi.org/10.1016/j.jep.2020.112797
Liu G, Bi Y, Xue L et al (2015) Dendritic cell SIRT1-HIF1alpha axis programs the differentiation of CD4+ T cells through IL-12 and TGF-beta1. Proc Natl Acad Sci USA 112:E957-965. https://doi.org/10.1073/pnas.1420419112
Liu P, Zhao H, Luo Y (2017) Anti-aging implications of astragalus membranaceus (Huangqi): a well-known Chinese tonic. Aging Dis 8:868–886. https://doi.org/10.14336/AD.2017.0816
Mantovani A, Allavena P (2015) The interaction of anticancer therapies with tumor-associated macrophages. J Exp Med 212:435–445. https://doi.org/10.1084/jem.20150295
Mantovani A, Marchesi F, Malesci A et al (2017) Tumour-associated macrophages as treatment targets in oncology. Nat Rev Clin Oncol 14:399–416. https://doi.org/10.1038/nrclinonc.2016.217
Modest DP, Pant S, Sartore-Bianchi A (2019) Treatment sequencing in metastatic colorectal cancer. Eur J Cancer 109:70–83. https://doi.org/10.1016/j.ejca.2018.12.019
Ni YH, Ding L, Huang XF et al (2015) Microlocalization of CD68+ tumor-associated macrophages in tumor stroma correlated with poor clinical outcomes in oral squamous cell carcinoma patients. Tumour Biol 36:5291–5298. https://doi.org/10.1007/s13277-015-3189-5
Ostuni R, Kratochvill F, Murray PJ et al (2015) Macrophages and cancer: from mechanisms to therapeutic implications. Trends Immunol 36:229–239. https://doi.org/10.1016/j.it.2015.02.004
Pyonteck SM, Akkari L, Schuhmacher AJ et al (2013) CSF-1R inhibition alters macrophage polarization and blocks glioma progression. Nat Med 19:1264–1272. https://doi.org/10.1038/nm.3337
Qian BZ, Pollard JW (2010) Macrophage diversity enhances tumor progression and metastasis. Cell 141:39–51. https://doi.org/10.1016/j.cell.2010.03.014
Ruffell B, Coussens LM (2015) Macrophages and therapeutic resistance in cancer. Cancer Cell 27:462–472. https://doi.org/10.1016/j.ccell.2015.02.015
Sica A, Schioppa T, Mantovani A et al (2006) Tumour-associated macrophages are a distinct M2 polarised population promoting tumour progression: potential targets of anti-cancer therapy. Eur J Cancer 42:717–727. https://doi.org/10.1016/j.ejca.2006.01.003
Sun P, Liu Y, Wang Q et al (2019) Astragaloside IV inhibits human colorectal cancer cell growth. Front Biosci 24:597–606
Wang S, Mou J, Cui L et al (2018) Astragaloside IV inhibits cell proliferation of colorectal cancer cell lines through down-regulation of B7–H3. Biomed Pharmacother 102:1037–1044. https://doi.org/10.1016/j.biopha.2018.03.127
Wynn TA, Chawla A, Pollard JW (2013) Macrophage biology in development, homeostasis and disease. Nature 496:445–455. https://doi.org/10.1038/nature12034
Xu F, Cui WQ, Wei Y et al (2018) Astragaloside IV inhibits lung cancer progression and metastasis by modulating macrophage polarization through AMPK signaling. J Exp Clin Cancer Res 37:207. https://doi.org/10.1186/s13046-018-0878-0
Ying W, Cheruku PS, Bazer FW et al (2013) Investigation of macrophage polarization using bone marrow derived macrophages. J Vis Exp 76:50323. https://doi.org/10.3791/50323
Zhang L, Zhou J, Qin X et al (2019) Astragaloside IV inhibits the invasion and metastasis of SiHa cervical cancer cells via the TGFbeta1mediated PI3K and MAPK pathways. Oncol Rep 41:2975–2986. https://doi.org/10.3892/or.2019.7062
Zhu Y, Knolhoff BL, Meyer MA et al (2014) CSF1/CSF1R blockade reprograms tumor-infiltrating macrophages and improves response to T-cell checkpoint immunotherapy in pancreatic cancer models. Cancer Res 74:5057–5069. https://doi.org/10.1158/0008-5472.CAN-13-3723
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Liu, F., Ran, F., He, H. et al. Astragaloside IV Exerts Anti-tumor Effect on Murine Colorectal Cancer by Re-educating Tumor-Associated Macrophage. Arch. Immunol. Ther. Exp. 68, 33 (2020). https://doi.org/10.1007/s00005-020-00598-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00005-020-00598-y