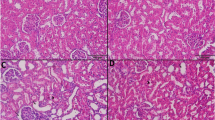
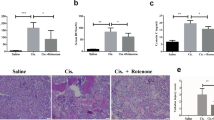
Abstract
Nephrotoxicity is the major dose-limiting factor of cisplatin chemotherapy. Reactive oxygen species generated in mitochondria are thought to be the main cause of cellular damage in such injury. The present study examined, in vivo, the protective potential of the hydroxyl radical scavenger dimethylthiourea (DMTU) against cisplatin-induced effects on renal mitochondrial bioenergetics, redox state and oxidative stress. Adult male Wistar rats (200 to 220 g) were divided into four groups of eight animals each. The control group was treated only with an intraperitoneal (i.p.) injection of saline solution (1 ml/100 g body weight). The second group was given only DMTU (500 mg/kg body weight, i.p, followed by 125 mg/Kg, i.p., twice a day until they were killed). The third group was given a single injection of cisplatin (10 mg/kg body weight, i.p.). The fourth group was given DMTU (500 mg/kg body weight, i.p.), just before the cisplatin injection (10 mg/kg body weight, i.p.), followed by injections of DMTU (125 mg/kg body weight, i.p.) twice a day until they were killed. Animals were killed 72 h after the treatment. Besides not presenting any direct effect on mitochondria, DMTU substantially inhibited cisplatin-induced mitochondrial injury and cellular death by apoptosis, suppressing the occurrence of acute renal failure. All the following cisplatin-induced effects were prevented by DMTU: (1) increased plasmatic levels of creatinine and blood urea nitrogen (BUN); (2) decreased ATP content, calcium uptake and electrochemical potential; (3) oxidation of lipids, including cardiolipin; and oxidation of proteins, including sulfhydryl, and aconitase enzyme, as well as accumulation of carbonyl proteins; (4) depletion of the antioxidant defense (NADPH and GSH) and (5) increased activity of the apoptosis executioner caspase-3. Our findings show the important role played by mitochondria and hydroxyl radicals in cisplatin-induced nephrotoxicity, as well as the effectiveness of DMTU in preventing the renal mitochondrial damage caused by cisplatin. These results strongly suggest that protection of mitochondria by hydroxyl radical scavengers may be an interesting approach to prevent the kidney tissue damage caused by cisplatin-chemotherapy.










Similar content being viewed by others
References
Taguchi T, Nazneen A, Abid MR, Razzaque MS (2005) Cisplatin-associated nephrotoxicity and pathological events. Contrib Nephrol 148:107–121
Baliga R, Zhang Z, Baliga M, Ueda N, Shah SV (1998) In vitro and in vivo evidence suggesting a role for iron in cisplatin-induced nephrotoxicity. Kidney Int 53:394–401
Ali BH, Al Moundhri MS (2006) Agents ameliorating or augmenting the nephrotoxicity of cisplatin and other platinum compounds: a review of some recent research. Food Chem Toxicol 44:1173–1183
Somani SM, Husain K, Whitworth C, Trammell GL, Malafa M, Rybak LP (2000) Dose-dependent protection by lipoic acid against cisplatin-induced nephrotoxicity in rats: antioxidant defense system. Pharmacol Toxicol 86:234–241
Chang B, Nishikawa M, Sato E, Utsumi K, Inoue M (2002) l-Carnitine inhibits cisplatin-induced injury of the kidney and small intestine. Arch Biochem Biophys 405:55–64
Matsushima H, Yonemura K, Ohishi K, Hishida A (1998) The role of oxygen free radicals in cisplatin-induced acute renal failure in rats. J Lab Clin Med 131:518–526
Turrens JF (2003) Mitochondrial formation of reactive oxygen species. J Physiol 552:335–344
Fariss MW, Chan CB, Patel M, Van Houten B, Orrenius S (2005) Role of mitochondria in toxic oxidative stress. Mol Interv 5:94–111
Sheu SS, Nauduri D, Anders MW (2006) Targeting antioxidants to mitochondria: a new therapeutic direction. Biochim Biophys Acta 1762:256–265
Lamson DW, Brignall MS (1999) Antioxidants in cancer therapy; their actions and interactions with oncologic therapies. Altern Med Rev 4:304–329
Lamson DW, Brignall MS (2000) Antioxidants and cancer therapy II: quick reference guide. Altern Med Rev 5:152–163
Moss RW (2006) Should patients undergoing chemotherapy and radiotherapy be prescribed antioxidants? Integr Cancer Ther 5:63–82
Tsuruya K, Tokumoto M, Ninomiya T, Hirakawa M, Masutani K, Taniguchi M, Fukuda K, Kanai H, Hirakata H, Iida K (2003) Antioxidant ameliorates cisplatin-induced renal tubular cell death through inhibition of death receptor-mediated pathways. Am J Renal Physiol 285:F208–F218
Cameron NE, Tuck Z, McCabe L, Cotter MA (2001) Effect of the hydroxyl radical scavenger, dimethylthiourea, on peripheral nerve tissue perfusion, conduction velocity and nociception in experimental diabetes. Diabetologia 44:1161–1191
Roychoudhury S, Ghosh SK, Chakraborti T, Chakraborti S (1996) H2O2-induced lipoperoxidation in mitochondria of pulmonary vascular smooth muscle tissue and its modification by DFO, DMTU and DIDS. Indian J Exp Biol 34:1220–1230
Baek SM, Kwon CH, Kim JH, Jung JS, Kim YK (2003) Differencial roles of hydrogen peroxide and hydroxyl radical in cisplatin-induced cell death in renal proximal tubular epithelial cells. J Lab Clin Med 142:178–186
Berry MN, Edwards AM, Barritt GJ (1991) High-yield preparation of isolated hepatocytes from rat liver. In: Burdon RH, Knippenberg PH (eds) Laboratory techniques in biochemistry and molecular biology. solated hepatocytes preparation, properties and applications. Elsevier, Amsterdam, pp 15–58
Pedersen PL, Grenawalt JW, Reynafarje B, Hullihen J, Decker GL, Soper JW, Bustamente E (1978) Preparation and characterization of mitochondria and submitochondrial particles of rat liver-derived tissues. Methods Cell Biol 20:411–481
Santos AC, Uyemura SA, Santos NA, Mingatto FE, Curti C (1997) Hg (II)-induced renal cytotoxicity: in vitro and in vivo implications for the bioenergetic and oxidative status of mitochondria. Mol Cell Biochem 177:53–59
O’Connor JE, Vargas JL, Kimler BF, Hernandez-Yago J, Grisolia S (1988) Use of rhodamine 123 to investigate alterations in mitochondrial activity in isolated mouse liver mitochondria. Biochem Biophys Res Commun 151:568–573
Emaus RK, Grunwald R, Lemasters JJ (1986) Rhodamine-123 as a probe of transmembrane potential in isolated rat-liver mitochondria-spectral and metabolic properties. Biochim Biophys Acta 850:436–448
Scarpa A (1979) Measurements of cation transport with metallochromic indicators. In: Fleisher S, Packer L (eds) Methods in Enzimology. Academic Press, New York, pp 301–352
Cardoso SM, Pereira C, Oliveira R (1999) Mitochondrial function is differentially affected upon oxidative stress. Free Radic Biol Med 26:3–13
Masubuchi Y, Suda C, Horie T (2005) Involvement of mitochondrial permeability transition in acetaminophen-induced liver injury in mice. J Hepatol 42:110–116
Petit JM, Maftah A, Ratinaud MH, Julien R (1992) 10-N-nonyl acridine orange interacts with cardiolipin and allows the quantification of this phospholipid in isolated mitochondria. Eur J Biochem 209:267–273
Gallet PF, Maftah A, Petit JM, Denis-Gay M, Julien R (1995) Direct cardiolipin assay in yeast using the red fluorescence emission of 10-N-nonyl acridine orange. Eur J Biochem 15:113–119
Grattagliano I, Vendemiale G, Sabbá C, Buonamico P, Altomare E (1996) Oxidation of circulating proteins in alcoholics: role of acetaldehyde and xantine oxidase. J Hepatol 25:28–36
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:192–205
Zheng W, Sean R, Graziano JH (1998) Manganese inhibits mitochondrial aconitase: a mechanism of manganese neurotoxicity. Brain Res 799:334–342
Lund BO, Miller DM, Woods JS (1993) Studies on Hg (II)- induced H2O2 formation and oxidative stress in vivo and in vitro in rat kidney mitochondria. Biochem Pharmacol 45:2017–2024
Tietze F (1969) Enzimatic method for quantitative determination of nanogram amounts of total and oxidized gluthathione: applications to mammaliam blood and others tissues. Anal Biochem 27:502–522
Singh G (1989) A possible mechanism of cisplatin-induced nephrotoxicity. Toxicology 58:71–80
Sueishi K, Mishima K, Makino K, Itoh Y, Tsuruya K, Hirakata H, Oishi R (2002) Protection by a radical scavenger edaravone against cisplatin-induced nephrotoxicity in rats. Eur J Pharmacol 451:203–208
Kim YH, Kim YW, Oh YJ, Back NI, Chung SA, Chung HG, Jeong TS, Choi MS, Lee KT (2006) Protective effect of the ethanol extract of the roots of Brassica rapa on cisplatin-induced nephrotoxicity in LLC-PK1 cells and rats. Biol Pharm Bull 29:2436–2441
Gemba M, Yamaguchi T, Kambara K, Suzuki A, Kawai Y (1992) Increases in urinary enzyme excretion in rats depleted of glutathione inhibited by scavenger of oxygen free radicals. J Pharmacobiodyn 15:513–518
Hanigan MH, Devarajan P (2003) Cisplatin nephrotoxicity: molecular mechanisms. Cancer Ther 1:47–61
Iguchi T, Nishikawa M, Chang B, Muroya O, Sato EF, Nakatani T, Inoue M (2004) Edaravone inhibits acute renal injury and cyst formation in cisplatin-treated rat kidney. Free Radic Res 38:333–341
Kharbangar A, Khynriam D, Prasad SB (2000) Effect of cisplatin on mitochondrial protein, glutathione, and succinate dehydrogenase in Dalton lymphoma-bearing mice. Cell Biol Toxicol 16:363–373
Hauff KD, Hatch GM (2006) Cardiolipin metabolism and Barth Syndrome. Prog Lipid Res 45:91–101
Antunes LM, Darin JD, Bianchi M de L (2001) Effects of the antioxidants curcumin or selenium on cisplatin-induced nephrotoxicity and lipid peroxidation in rats. Pharmacol Res 43:145–150
Mora L de O, Antunes LM, Francescato HD, Bianchi M de L (2003) The effects of oral glutamine on cisplatin-induced nephrotoxicity in rats. Pharmacol Res 47:517–522
Tirosh O, Katzhendler Y, Barenholz Y, Ginsburg I, Kohen R (1996) Antioxidant properties of amidothionophosphates: novel antioxidant molecules. Free Radic Biol Med 20:421–432
Al-Harbi MM, Osman AM, al-Gharably NM, al-Bekairi AM, al-Shabanah OA, Sabah DM, Raza M (1995) Effect of desferrioxamine on cisplatin-induced nephrotoxicity in normal rats. Chemotherapy 41:448–454
Adwankar M, Banerji A, Ghosh S (1991) Differential response of retinoic acid pretreated human synovial sarcoma cell line to anticancer drugs. Tumori 77:391–394
Kurbacher CM, Wagner U, Kolster B, Andreotti PE, Krebs D, Bruckner HW (1996) Ascorbic acid (vitamin C) improves the antineoplastic activity of doxorubicin, cisplatin, and paclitaxel in human breast carcinoma cells in vitro. Cancer Lett 103:183–189
Marverti G, Andrews PA (1996) Stimulation of cisdiamminedichloroplatinum(II) accumulation by modulation of passive permeability with genistein: an altered response in accumulationdefective resistant cells. Clin Cancer Res 2:991–999
Hofmann J, Fiebig HH, Winterhalter BR, Berger DP, Grunicke H (1990) Enhancement of the antiproliferative activity of cis-diamminedichloroplatinum(II) by quercetin. Int J Cancer 45:536–539
Weijl NI, Cleton FJ, Osanto S (1997) Free radicals and antioxidants in chemotherapy-induced toxicity. Cancer Treat Rev 23:209–240
Sastry J, Kellie SJ (2005) Severe neurotoxicity, ototoxicity and nephrotoxicity following high-dose cisplatin and amifostine. Pediatr Hematol Oncol 22:441–445
Wang D, Lippard SJ (2005) Cellular processing of platinum anticancer drugs. Nat Rev Drug Discov 4:307–320
Saillenfait AM, Sabate JP, Langonne I, de Ceaurriz J (1991) Difference in the developmental toxicity of ethylenethiourea and three N,N′-substituted thiourea derivatives in rats. Fundam Appl Toxicol 17:399–408
Beehler CJ, Ely ME, Rutledge KS, Simchuk ML, Reiss OK, Shanley PF, Repine JE (1994) Toxic effects of dimethylthiourea in rats. J Lab Clin Med 123:73–80
Kim SG, Kim HJ, Yang CH (1999) Thioureas differentially induce rat hepatic microsomal epoxide hydrolase and rGSTA2 irrespective of their oxygen radical scavenging effect: effects on toxicant-induced liver injury. Chem Biol Interact 117:117–134
Milner LS, Wei SH, Houser MT (1993) Enhancement of renal and hepatic glutathione metabolism by dimethylthiourea. Toxicol Lett 66:117–123
Otoikhian A, Simoyi RH, Petersen JL (2005) Oxidation of a dimethylthiourea metabolite by iodine and acidified iodate: N,N′-dimethylaminoiminomethanesulfinic acid (1). Chem Res Toxicol 18:1167–1177
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Santos, N.A.G., Bezerra, C.S.C., Martins, N.M. et al. Hydroxyl radical scavenger ameliorates cisplatin-induced nephrotoxicity by preventing oxidative stress, redox state unbalance, impairment of energetic metabolism and apoptosis in rat kidney mitochondria. Cancer Chemother Pharmacol 61, 145–155 (2008). https://doi.org/10.1007/s00280-007-0459-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-007-0459-y