Abstract
Purpose
Observational studies report conflicting results on the association between metformin exposure and prostate cancer outcomes. This meta-analysis summarizes studies reporting overall survival, prostate cancer-specific mortality, and biochemical recurrence.
Methods
PubMed and Embase were systematically reviewed to identify studies investigating the association between metformin use and clinical endpoints among men with prostate cancer while taking confounding by diabetes diagnosis into account. Pooled risk estimates (hazard ratios, HRs) and 95 % confidence intervals (CIs) were calculated using random-effects models. Sensitivity analyses for quality components and factors for heterogeneity were conducted.
Results
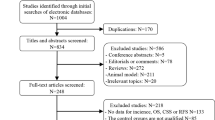
Of 549 articles identified, nine retrospective cohort studies representing 9,186 patients were included. There was significant heterogeneity between studies, and studies differed in quality. Metformin use was associated with improved overall survival in studies with clear risk window definition (HR 0.88, 95 % CI 0.86–0.90, p < 0.001) and in studies with potential immortal time bias (HR 0.52, 95 % CI 0.41–0.65, p < 0.001). No significant association with prostate cancer-specific mortality was detected (HR 0.76, 95 % CI 0.44–1.31, p = 0.33). Metformin use was associated with a decreased risk of biochemical recurrence (HR 0.79, 95 % CI 0.63–1.00, p = 0.047).
Conclusions
This meta-analysis suggests a benefit of metformin in men with diabetes and prostate cancer. However, further carefully designed studies are needed to confirm findings and to assess potential generalization to non-diabetic, non-white, and less aggressively treated men with prostate cancer.




Similar content being viewed by others
Abbreviations
- HR:
-
Hazard ratio
- OS:
-
Overall survival
- PCSM:
-
Prostate cancer-specific mortality
- PSA:
-
Prostate-specific antigen
- BCR:
-
Biochemical recurrence
- 95 % CI:
-
95 % Confidence interval
- RCT:
-
Randomized controlled trial
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61:69–90
Attner B, Landin-Olsson M, Lithman T, Noreen D, Olsson H (2012) Cancer among patients with diabetes, obesity and abnormal blood lipids: a population-based register study in Sweden. Cancer Causes Control 23:769–777
Kasper JS, Giovannucci E (2006) A meta-analysis of diabetes mellitus and the risk of prostate cancer. Cancer Epidemiol Biomark Prev 15:2056–2062
Bensimon L, Yin H, Suissa S, Pollak MN, Azoulay L (2014) Type 2 diabetes and the risk of mortality among patients with prostate cancer. Cancer Causes Control 25:329–338
Nathan DM, Buse JB, Davidson MB et al (2006) Management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 29:1963–1972
Flavin R, Zadra G, Loda M (2011) Metabolic alterations and targeted therapies in prostate cancer. J Pathol 223:283–294
Ben Sahra I, Laurent K, Loubat A et al (2008) The antidiabetic drug metformin exerts an antitumoral effect in vitro and in vivo through a decrease of cyclin D1 level. Oncogene 27:3576–3586
DeCensi A, Puntoni M, Goodwin P et al (2010) Metformin and cancer risk in diabetic patients: a systematic review and meta-analysis. Cancer Prev Res (Phila) 3:1451–1461
Zhang P, Li H, Tan X, Chen L, Wang S (2013) Association of metformin use with cancer incidence and mortality: a meta-analysis. Cancer Epidemiol 37:207–218
Yu H, Yin L, Jiang X et al (2014) Effect of metformin on cancer risk and treatment outcome of prostate cancer: a meta-analysis of epidemiological observational studies. PLoS ONE 9:e116327
Raval AD, Thakker D, Vyas A, Salkini M, Madhavan S, Sambamoorthi U (2015) Impact of metformin on clinical outcomes among men with prostate cancer: a systematic review and meta-analysis. Prostate Cancer Prostatic Dis 18:110–121
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6:e1000097
Saenz A, Fernandez-Esteban I, Mataix A, Ausejo M, Roque M, Moher D (2005) Metformin monotherapy for type 2 diabetes mellitus. Cochrane Database Syst Rev 20(3):CD002966
Stephenson AJ, Kattan MW, Eastham JA et al (2006) Defining biochemical recurrence of prostate cancer after radical prostatectomy: a proposal for a standardized definition. J Clin Oncol 24:3973–3978
Margel D, Urbach DR, Lipscombe LL et al (2013) Metformin use and all-cause and prostate cancer-specific mortality among men with diabetes. J Clin Oncol 31:3069–3075
Cochran WG (1954) The combination of estimates from different experiments. Biometrics 10:101–129
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
Jüni P, Altman DG, Egger M (2001) Systematic reviews in health care: assessing the quality of controlled clinical trials. BMJ 323:42–46
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634
Bensimon L, Yin H, Suissa S, Pollak MN, Azoulay L (2014) The use of metformin in patients with prostate cancer and the risk of death. Cancer Epidemiol Biomark Prev 23:2111–2118
Xu H, Aldrich MC, Chen Q et al (2015) Validating drug repurposing signals using electronic health records: a case study of metformin associated with reduced cancer mortality. J Am Med Inform Assoc 22(1):179–191
Spratt DE, Zhang C, Zumsteg ZS, Pei X, Zhang Z, Zelefsky MJ (2013) Metformin and prostate cancer: reduced development of castration-resistant disease and prostate cancer mortality. Eur Urol 63:709–716
Häggström C, Stocks T, Nagel G et al (2014) Prostate cancer, prostate cancer death, and death from other causes, among men with metabolic aberrations. Epidemiology 25:823–828
Lévesque LE, Hanley JA, Kezouh A, Suissa S (2010) Problem of immortal time bias in cohort studies: example using statins for preventing progression of diabetes. BMJ 340:b5087
He XX, Tu SM, Lee MH, Yeung SC (2011) Thiazolidinediones and metformin associated with improved survival of diabetic prostate cancer patients. Ann Oncol 22:2640–2645
Patel T, Hruby G, Badani K, Abate-Shen C, McKiernan JM (2010) Clinical outcomes after radical prostatectomy in diabetic patients treated with metformin. Urology 76:1240–1244
D’Amico AV, Cote K, Loffredo M, Renshaw AA, Chen MH (2003) Pretreatment predictors of time to cancer specific death after prostate specific antigen failure. J Urol 169:1320–1324
Margel D, Urbach D, Lipscombe LL et al (2013) Association between metformin use and risk of prostate cancer and its grade. J Natl Cancer Inst 105:1123–1131
Penney KL, Stampfer MJ (2013) The time is ripe for a randomized trial of metformin in clinically localized prostate cancer. J Clin Oncol 31:3054–3055
Popiolek M, Rider JR, Andren O et al (2013) Natural history of early, localized prostate cancer: a final report from three decades of follow-up. Eur Urol 63:428–435
Patorno E, Garry EM, Patrick AR et al (2015) Addressing limitations in observational studies of the association between glucose-lowering medications and all-cause mortality: a review. Drug Saf 38:295–310
Walker AM (1996) Confounding by indication. Epidemiology 7:335–336
Lee H, Kuk H, Byun SS, Lee SE, Hong SK (2015) Preoperative glycemic control status as a significant predictor of biochemical recurrence in prostate cancer patients after radical prostatectomy. PLoS ONE 10:e0124761
Suissa S, Azoulay L (2012) Metformin and the risk of cancer: time-related biases in observational studies. Diabetes Care 35:2665–2673
Ray WA (2003) Evaluating medication effects outside of clinical trials: new-user designs. Am J Epidemiol 158:915–920
IntHout J, Ioannidis JP, Borm GF (2014) The Hartung–Knapp–Sidik–Jonkman method for random effects meta-analysis is straightforward and considerably outperforms the standard DerSimonian–Laird method. BMC Med Res Methodol 14:25
Rothermundt C, Hayoz S, Templeton AJ et al (2014) Metformin in chemotherapy-naive castration-resistant prostate cancer: a multicenter phase 2 trial (SAKK 08/09). Eur Urol 66:468–474
Joshua AM, Zannella VE, Downes MR et al (2014) A pilot ‘window of opportunity’ neoadjuvant study of metformin in localised prostate cancer. Prostate Cancer Prostatic Dis 17:252–258
Allott EH, Abern MR, Gerber L et al (2013) Metformin does not affect risk of biochemical recurrence following radical prostatectomy: results from the SEARCH database. Prostate Cancer Prostatic Dis. 16:391–397
Kaushik D, Karnes RJ, Eisenberg MS, Rangel LJ, Carlson RE, Bergstralh EJ (2014) Effect of metformin on prostate cancer outcomes after radical prostatectomy. Urol Oncol 32(43):e1–e7
Spratt DE, Zhang Z, Zelefsky MJ (2013) Reply to Leah Bensimon, Samy Suissa, and Laurent Azoulay’s letter to the editor re: Daniel E. Spratt, Chi Zhang, Zachary S. Zumsteg, Xin Pei, Zhigang Zhang, Michael J. Zelefsky. metformin and prostate cancer: reduced development of castration-resistant disease and prostate cancer mortality. Eur Urol 2013;63:709–16. Eur Urol 64:e29–e30
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest to disclose.
Rights and permissions
About this article
Cite this article
Stopsack, K.H., Ziehr, D.R., Rider, J.R. et al. Metformin and prostate cancer mortality: a meta-analysis. Cancer Causes Control 27, 105–113 (2016). https://doi.org/10.1007/s10552-015-0687-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-015-0687-0