Abstract
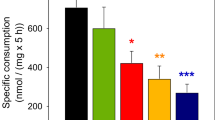
It is well established that 3-hydroxybutyrate can serve as an energy source for the brain. Since substrate utilization may be regulated in part by transport across the cellular membrane, we investigated the uptake of 3-hydroxybutyrate by primary cultures of rat brain astrocytes. Measurement of the net uptake indicated a saturable system and a Lineweaver-Burke type plot was consistent with a single carrier-mediated mechanism with a Km of 6.03 mM and a Vmax of 32.7 nmol/30 seconds/mg protein. The rate of uptake at pH 6.2 was more than ten times the rate at pH 8.2, with the rate at pH 7.4 being intermediate between these values, suggesting the possibility of cotransport with H+ or exchange with OH− (antiport). Mersalyl had only a slight effect on the transport of 3-hydroxybutyrate, suggesting that sulfhydryl groups are not involved in the transport of this monocarboxylic acid. Phenylpyruvate and α-ketoisocaproate also attenuated the transport, but lactate had only a marginal effect. These results suggest that the utilization of 3-hydroxybutyrate as an energy source by astrocytes is regulated in part by carrier-mediated transport and that the uptake system is different from the lactate transport system.
Similar content being viewed by others
References
Kraus, H., Schlenker, S., and Schwedesky, D. 1974. Developmental changes of cerebral ketone body utilization in human infants. Hoppe-Seyler's Z. Physiol. Chem. 355:164–170.
Persson, B., Settergsen, G., and Dahlquist, G. 1975. Cerebral arteriovenous difference of acetoacetate and 3-hydroxybutyrate. Acta Paediatr. Scand. 61:273–278.
Robinson, A. M. and Williamson, D. H. 1980. Physiological roles of ketone bodies as substrates and signals in mammalian tissues. Physiol. Rev. 60:143–189.
Hawkins, R. A., Williamson, D. H. and Krebs, H. A. 1971. Ketone body utilization by adult and suckling rat brain in vivo. Biochem. J. 122:13–18.
Cremer, J. E. Nutrients for the brain. Problems in supply. 1982. Early Hum. Develop. 5:117–132.
Page, M. A., Krebs, H. A., and Williamson, D. H. 1971. Activities of enzymes of ketone-body utilization in brain and other tissues of suckling rats. Biochem. J. 121:49–53.
Middleton, B. 1973. The acetoacetyl CoA thiolases of rat brain and their relative activities during postnatal development. Biochem. J. 132:731–737.
Tildon, J. T., Cone, A. L., and Cornblath, M. 1971. Coenzyme A transferase activity in rat brain. Biochem. Biophys. Res. Commun. 148:382–390.
Roeder, L. M., Tildon, J. T., and Holman, D. C. 1984. Competition among oxidizable substrates in brains of young and adult rats: dissociated cells. Biochem. J. 219:131–135.
Roeder, L. M., Tildon, J. T., and Stevenson, Jr., J. H. 1984. Competition among oxidizable substrates in brains of young and adult rats: whole homogenates. Biochem. J. 219:125–130.
Daniel, P. M., Love, E. R., Moorhouse, S. R., and Pratt, O. E. 1977. The transport of ketone bodies into the brain of the rat (in vivo). J. Neurol. Sci. 34:1–13.
Gjedde, A., and Crone, C. 1975. Induction processes in blood brain transfer of ketone bodies during starvation. Am. J. Physiol. 229:1165–1169.
Pollay, M., and Steven, F. A. 1980. Starvation induced changes in transport of ketone bodies across the blood brain barrier. J. Neurosci. Res. 5:163–172.
Tildon, J. T., and Roeder, L. M. 1988. Transport of 3-hydroxy[3-14C]butyrate by dissociated cells from rat brain. Am. J. Physiol. 255 (Cell Physiol. 24) C133–139.
Shank, R. P., and Campbell, G. LeM. 1984. α-ketoglutarate and malate uptake and metabolism by synaptosomes: Further evidence for an astrocyte-to-neuron metabolic shuttle. J. Neurochem. 42: 1153–1161.
Shank, R. P., and Campbell, G. LeM. 1984. Amino acid uptake, content and metabolism by neuronal and glial enriched cellular fractions from mouse cerebellum. J. Neurosci. 4:58–69.
Yu, A. C., Hertz, E., Schousboe, A., and Hertz, L. 1984. Uptake and metabolism of malate in cultures of astrocytes and neurons. Trans. Am. Soc. Neurochem. 15:A216.
Auestad, N., Korsak, R. A., Marrow, J. W. and Edmond, J. 1991. Fatty acid oxidation and ketogenesis by astrocytes in primary culture. J. Neurochem. 56:1376–1386.
Edmond, J., Robbins, R. A., Bergstron, J. D., Cole, R. A., and deVellis, J. 1987. Capacity for substrate utilization in oxidative metabolism by neurons, astrocytes and oligodendrocytes from developing brain in primary culture. J. Neurosci. Res. 18:551–561.
McKenna, M. C., Tildon, J. T., Couto, R., Stevenson, J. H., and Caprio, F. J. 1990. The metabolism of malate by cultured rat brain astrocytes. Neurochemical Research 15:1211–1220.
Tildon, J. T., McKenna, M. C., Stevenson, J. H., and Couto, R. 1993. Transport of L-lactate by cultured rat brain astrocytes. Neurochemical Research 18:177–184.
Roeder, L. M., Tildon, J. T., and Williams, I. B. 1985. Transport of 2-deoxy-D-glucose by dissociated brain cells. Brain Res. 345: 289–305.
Zielke, H. R., Tildon, J. T., Landry, M. E. and Max, S. R. 1990. Effect of 8-Bromo-cAMP and dexamethasone on glutamate metabolism in rat astrocytes. Neurochem. Res. 15:1115–1122.
Smith, P. K., Krohn, R. I., Hermanson, G. T., Mallia, A. K., Gartner, F. H., Provenzano, M. D., Fujimoto, E. K., Goeke, N. M., Olson, B. J., and Klenk, D. C. 1985. Measurement of protein using bicinchoninic acid. Anal. Biochem. 150:76–85.
Spencer, T. L., and Lehninger, A. L. 1976. L-lactate transport in Ehrlich ascites-tumor cells. Biochem. J. 154:405–414.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tildon, J.T., McKenna, M.C. & Stevenson, J.H. Transport of 3-hydroxybutyrate by cultured rat brain astrocytes. Neurochem Res 19, 1237–1242 (1994). https://doi.org/10.1007/BF01006812
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01006812