Abstract
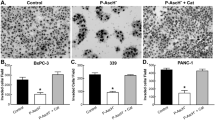
HIF-1α is a transcriptional regulator that functions in the adaptation of cells to hypoxic conditions; it strongly impacts the prognosis of patients with cancer. High-dose, intravenous, pharmacological ascorbate (P-AscH−), induces cytotoxicity and oxidative stress selectively in cancer cells by acting as a pro-drug for the delivery of hydrogen peroxide (H2O2); early clinical data suggest improved survival and inhibition of metastasis in patients being actively treated with P-AscH−. Previous studies have demonstrated that activation of HIF-1α is necessary for P-AscH− sensitivity. We hypothesized that pancreatic cancer (PDAC) progression and metastasis could be be targeted by P-AscH− via H2O2-mediated inhibition of HIF-1α stabilization. Our study demonstrates an oxygen- and prolyl hydroxylase-independent regulation of HIF-1α by P-AscH−. Additionally, P-AscH− decreased VEGF secretion in a dose-dependent manner that was reversible with catalase, consistent with an H2O2-mediated mechanism. Pharmacological and genetic manipulations of HIF-1α did not alter P-AscH−-induced cytotoxicity. In vivo, P-AscH− inhibited tumor growth and VEGF expression. We conclude that P-AscH− suppresses the levels of HIF-1α protein in hypoxic conditions through a post-translational mechanism. These findings suggest potential new therapies specifically designed to inhibit the mechanisms that drive metastases as a part of PDAC treatment.







Similar content being viewed by others
References
Matsuo Y, Ding Q, Desaki R et al (2014) Hypoxia inducible factor-1 alpha plays a pivotal role in hepatic metastasis of pancreatic cancer: an immunohistochemical study. J Hepatobiliary Pancreat Sci 21(2):105–112
Sun HC, Qiu ZJ, Liu J et al (2007) Expression of hypoxia-inducible factor-1 alpha and associated proteins in pancreatic ductal adenocarcinoma and their impact on prognosis. Int J Oncol 30(6):1359–1367
Hoffmann AC, Mori R, Vallbohmer D et al (2008) High expression of HIF1a is a predictor of clinical outcome in patients with pancreatic ductal adenocarcinomas and correlated to PDGFA, VEGF, and bFGF. Neoplasia 10(7):674–679
Shibaji T, Nagao M, Ikeda N et al (2003) Prognostic significance of HIF-1 alpha overexpression in human pancreatic cancer. Anticancer Res 23(6C):4721–4727
Buchler P, Reber HA, Buchler M et al (2003) Hypoxia-inducible factor 1 regulates vascular endothelial growth factor expression in human pancreatic cancer. Pancreas 26(1):56–64
Cheng ZX, Sun B, Wang SJ et al (2011) Nuclear factor-kappaB-dependent epithelial to mesenchymal transition induced by HIF-1alpha activation in pancreatic cancer cells under hypoxic conditions. PloS ONE 6(8):e23752
Keith B, Johnson RS, Simon MC (2012) HIF1alpha and HIF2alpha: sibling rivalry in hypoxic tumour growth and progression. Nat Rev Cancer 12(1):9–22
Wang M, Chen MY, Guo XJ, Jiang JX (2015) Expression and significance of HIF-1alpha and HIF-2alpha in pancreatic cancer. J Huazhong Univ Sci Technol Med Sci 35(6):874–879
Yang J, Zhang X, Zhang Y et al (2016) HIF-2alpha promotes epithelial-mesenchymal transition through regulating Twist2 binding to the promoter of E-cadherin in pancreatic cancer. J Exp Clin Cancer Res 26:35
Sendoel A, Kohler I, Fellmann C, Lowe SW, Hengartner MO (2010) HIF-1 antagonizes p53-mediated apoptosis through a secreted neuronal tyrosinase. Nature 465(7298):577–583
Ma WW, Adjei AA (2009) Novel agents on the horizon for cancer therapy. CA Cancer J Clin 59(2):111–137
Wang M, Kirk JS, Venkataraman S et al (2005) Manganese superoxide dismutase suppresses hypoxic induction of hypoxia-inducible factor-1alpha and vascular endothelial growth factor. Oncogene 24(55):8154–8166
Schofield CJ, Ratcliffe PJ (2004) Oxygen sensing by HIF hydroxylases. Nat Rev Mol Cell Biol 5(5):343–354
Dupuy F, Tabaries S, Andrzejewski S et al (2015) PDK1-dependent metabolic reprogramming dictates metastatic potential in breast cancer. Cell Metab 22(4):577–589
Graumlich JF, Ludden TM, Conry-Cantilena C, Cantilena LR Jr, Wang Y, Levine M (1997) Pharmacokinetic model of ascorbic acid in healthy male volunteers during depletion and repletion. Pharm Res 14(9):1133–1139
Levine M, Conry-Cantilena C, Wang Y et al (1996) Vitamin C pharmacokinetics in healthy volunteers: evidence for a recommended dietary allowance. Proc Natl Acad Sci USA 93(8):3704–3709
Welsh JL, Wagner BA, van’t Erve TJ et al (2013) Pharmacological ascorbate with gemcitabine for the control of metastatic and node-positive pancreatic cancer (PACMAN): results from a phase I clinical trial. Cancer Chemother Pharmacol 71(3):765–775
Du J, Martin SM, Levine M et al (2010) Mechanisms of ascorbate-induced cytotoxicity in pancreatic cancer. Clin Cancer Res 16(2):509–520
Chen Q, Espey MG, Sun AY et al (2007) Ascorbate in pharmacologic concentrations selectively generates ascorbate radical and hydrogen peroxide in extracellular fluid in vivo. Proc Natl Acad Sci USA 104(21):8749–8754
Tian W, Wang Y, Xu Y et al (2014) The hypoxia-inducible factor renders cancer cells more sensitive to vitamin C-induced toxicity. J Biol Chem 289(6):3339–3351
Roy I, Zimmerman NP, Mackinnon AC, Tsai S, Evans DB, Dwinell MB (2014) CXCL12 chemokine expression suppresses human pancreatic cancer growth and metastasis. PloS ONE 9(3):e90400
Kim MP, Evans DB, Wang H, Abbruzzese JL, Fleming JB, Gallick GE (2009) Generation of orthotopic and heterotopic human pancreatic cancer xenografts in immunodeficient mice. Nat Protoc 4(11):1670–1680
Buettner GR (1988) In the absence of catalytic metals ascorbate does not autoxidize at pH 7: ascorbate as a test for catalytic metals. J Biochem Biophys Methods 16(1):27–40
Du J, Daniels DH, Asbury C et al (2006) Mitochondrial production of reactive oxygen species mediate dicumarol-induced cytotoxicity in cancer cells. J Biol Chem 281(49):37416–37426
Matsuura H, Ichiki T, Ikeda J et al (2011) Inhibition of prolyl hydroxylase domain-containing protein downregulates vascular angiotensin II type 1 receptor. Hypertension (Dallas Tex: 1979) Sep 58(3):386–393
Epstein AC, Gleadle JM, McNeill LA et al (2001) C. elegans EGL-9 and mammalian homologs define a family of dioxygenases that regulate HIF by prolyl hydroxylation. Cell 05(1):43–54 107
Euhus DM, Hudd C, LaRegina MC, Johnson FE (1986) Tumor measurement in the nude mouse. J Surg Oncol 31(4):229–234
Doskey CM, van ‘t Erve TJ, Wagner BA, Buettner GR (2015) Moles of a substance per cell is a highly informative dosing metric in cell culture. PloS ONE 10(7):e0132572
Doskey CM, Buranasudja V, Wagner BA et al (2016) Tumor cells have decreased ability to metabolize H2O2: implications for pharmacological ascorbate in cancer therapy. Redox Biol 10:274–284
Brown LF, Detmar M, Claffey K et al (1997) Vascular permeability factor/vascular endothelial growth factor: a multifunctional angiogenic cytokine. EXS 79:233–269
Nagy JA, Vasile E, Feng D et al (2002) Vascular permeability factor/vascular endothelial growth factor induces lymphangiogenesis as well as angiogenesis. J Exp Med 196(11):1497–1506
Dvorak HF, Detmar M, Claffey KP, Nagy JA, van de Water L, Senger DR (1995) Vascular permeability factor/vascular endothelial growth factor: an important mediator of angiogenesis in malignancy and inflammation. Int Arch Allergy Immunol 107(1–3):233–235
Choi KS, Bae MK, Jeong JW, Moon HE, Kim KW (2003) Hypoxia-induced angiogenesis during carcinogenesis. J Biochem Mol Biol 36(1):120–127
Erudaitius D, Huang A, Kazmi S, Buettner GR, Rodgers VG (2017) Peroxiporin expression is an important factor for cancer cell susceptibility to therapeutic H2O2: implications for pharmacological ascorbate therapy. PloS ONE 12(1):e0170442
Liu YV, Baek JH, Zhang H, Diez R, Cole RN, Semenza GL (2007) RACK1 competes with HSP90 for binding to HIF-1alpha and is required for O(2)-independent and HSP90 inhibitor-induced degradation of HIF-1alpha. Mol Cell 25(2):207–217
Kaewpila S, Venkataraman S, Buettner GR, Oberley LW (2008) Manganese superoxide dismutase modulates hypoxia-inducible factor-1 alpha induction via superoxide. Cancer Res 68(8):2781–2788
Maes C, Carmeliet G, Schipani E (2012) Hypoxia-driven pathways in bone development, regeneration and disease. Nat Rev Rheumatol 8(6):358–366
Du J, Cieslak JA 3rd, Welsh JL et al (2015) Pharmacological ascorbate radiosensitizes pancreatic cancer. Cancer Res 75(16):3314–3326
Serrano OK, Parrow NL, Violet PC et al (2015) Antitumor effect of pharmacologic ascorbate in the B16 murine melanoma model. Free Radic Biol Med 87:193–203
Ma Y, Chapman J, Levine M, Polireddy K, Drisko J, Chen Q (2014) High-dose parenteral ascorbate enhanced chemosensitivity of ovarian cancer and reduced toxicity of chemotherapy. Sci Transl Med 6(222):222ra218
Hoffer LJ, Robitaille L, Zakarian R et al (2015) High-dose intravenous vitamin C combined with cytotoxic chemotherapy in patients with advanced cancer: a phase I-II clinical trial. PloS ONE 10(4):e0120228
Rouleau L, Antony AN, Bisetto S et al (2016) Synergistic effects of ascorbate and sorafenib in hepatocellular carcinoma: New insights into ascorbate cytotoxicity. Free Radic Biol Med 95:308–322
Malik NK, May KS, Chandrasekhar R et al (2012) Treatment of locally advanced unresectable pancreatic cancer: a 10-year experience. J Gastrointest Oncol 3(4):326–334
Rhim AD, Mirek ET, Aiello NM et al (2012) EMT and dissemination precede pancreatic tumor formation. Cell 148(1–2):349–361
Campbell EJ, Vissers MC, Wohlrab C et al (2016) Pharmacokinetic and anti-cancer properties of high dose ascorbate in solid tumours of ascorbate-dependent mice. Free Radic Biol Med 99:451–462
Montagner M, Enzo E, Forcato M et al (2012) SHARP1 suppresses breast cancer metastasis by promoting degradation of hypoxia-inducible factors. Nature 487(7407):380–384
Briggs KJ, Koivunen P, Cao S et al (2016) Paracrine induction of HIF by glutamate in breast cancer: EglN1 senses cysteine. Cell 166(1):126–139
Gerald D, Berra E, Frapart YM et al (2004) JunD reduces tumor angiogenesis by protecting cells from oxidative stress. Cell 118(6):781–794
Daijo H, Hoshino Y, Kai S et al (2016) Cigarette smoke reversibly activates hypoxia-inducible factor 1 in a reactive oxygen species-dependent manner. Sci Rep 6:34424
Rawal M, Schroeder SR, Wagner BA et al (2013) Manganoporphyrins increase ascorbate-induced cytotoxicity by enhancing H2O2 generation. Cancer Res 73(16):5232–5241
Lee DC, Sohn HA, Park ZY et al (2015) A lactate-induced response to hypoxia. Cell 161(3):595–609
Acknowledgements
HIF-1α Adenovirus construct obtained from Patrick H Maxwell, FMedSciRegius Professor of Physic & Head of the School of Clinical Medicine, University of Cambridge. Supported by NIH Grants CA184051, CA148062, CA169046, CA086862, and a Merit Review grant from the Medical Research Service, Department of Veterans Affairs 1I01BX001318-01A2.
Author information
Authors and Affiliations
Contributions
JC and JW conceived the hypothesis, designed the study, and performed the majority of the experiments. JW, BO, JD and KGC, performed experiments. JW, JC, and BO wrote the manuscript. JD, ZS, AK, KGC, and ST contributed reagents and tools for the study. JW, BO, GB, and JC contributed to discussion and edited the manuscript. All authors reviewed the results and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The Authors declare that they have no conflicts of interest with the contents of this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wilkes, J.G., O’Leary, B.R., Du, J. et al. Pharmacologic ascorbate (P-AscH−) suppresses hypoxia-inducible Factor-1α (HIF-1α) in pancreatic adenocarcinoma. Clin Exp Metastasis 35, 37–51 (2018). https://doi.org/10.1007/s10585-018-9876-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-018-9876-z